Abstract
Background: Impulsivity is understood as a range of behaviours, but the association between these behaviours is not well understood. Although high motor impulsivity is a key symptom of disorders like pathological gambling and addiction, in which decision-making on laboratory tasks is compromised, there have been no clear demonstrations that choice and motor impulsivity are associated in the general population. We examined this association in a large population of rodents.
Methods: We performed a meta-analysis on behavioural data from 211 manipulation-naive male animals that performed a rodent gambling task in our laboratory between 2008 and 2012. The task measures an aspect of both impulsive decision-making and impulsive action, making it possible to evaluate whether these 2 forms of maladaptive behaviour are related.
Results: Our meta-analysis revealed that motor impulsivity was positively correlated with poor decision-making under risk. Highly motor impulsive rats were slower to adopt an advantageous choice strategy and quicker to make a choice on individual trials.
Limitations: The data analyzed were limited to that produced by our laboratory and did not include data of other researchers who have used the task.
Conclusion: This work may represent the first demonstration of a clear association between choice and motor impulsivity in a nonclinical population. This lends support to the common practice of studying impulsivity in nonclinical populations to gain insight into impulse control disorders and suggests that differences in impulsive behaviours between clinical and nonclinical populations may be ones of magnitude rather than ones of quality.
Introduction
Impulsivity, loosely defined as action without foresight, is a component of many psychiatric disorders, including attention-deficit/hyperactivity disorder, mania, substance use and addictive disorders, and impulse control disorders (ICDs).1 The term “impulsivity” describes a breadth of behaviours and may be better understood as a spectrum of loosely related constructs rather than a uniform pathology.2,3 These various forms can be broadly classified as motor impulsivity and choice impulsivity.1,3,4 Motor impulsivity is often characterized by the inability to withhold a response, whereas choice impulsivity relates to disadvantageous decision-making that favours short-term gains without consideration of the associated long-term losses.1–6
Laboratory paradigms, such as the Iowa Gambling Task (IGT), wherein participants must choose between decks of cards with varying probabilities of monetary reward and punishment, provide a reliable measure of one aspect of choice impulsivity in humans, namely preference for risky (disadvantageous) over conservative (advantageous) options.7 As an analogue of the IGT, the rodent gambling task (rGT) allows animals to choose among 4 options, signalled by illumination of 4 response apertures, each with a unique probability of sucrose reward or time-out punishment.8 As in the IGT, the best strategy is to favour options associated with smaller incremental gain but also smaller penalties, resulting in maximization of sugar pellet profits. In contrast, a preference for the tempting “high-risk, high-reward” outcomes is ultimately disadvantageous: although such options can yield greater rewards per trial, the disproportionately larger punishments result in considerably less benefit over time.
Whereas such a preference reflects an aspect of choice impulsivity, the task also incorporates a measure of motor impulsivity: after initiation of each trial, the choice phase is preceded by a 5-second interval during which animals must withhold from responding at any of the apertures.8 Such premature responding has been extensively validated as an index of impulsive action through the 5-choice serial reaction time task (5CSRT) — a rodent analogue of the continuous performance task recently back-translated into humans.6,9,10 This concurrent measurement provides the unique opportunity to examine the association between discrete forms of impulsivity within individual animals.
In the absence of robust animal models for psychiatric disorders in which impulsivity is elevated, researchers assume that manipulations that reduce high levels of impulsivity in healthy individuals may have potential as treatments for clinically significant impulse control deficits. This assumption may be unjustified; high impulsivity is a key symptom of disorders like pathological gambling and addiction in which decision-making is compromised,11,12 but there have been no clear demonstrations that choice and motor impulsivity are likewise related in the general population. In fact, there are many findings that suggest that there is no association between various types of impulsivity.6,13–18 This is potentially problematic; the lack of a similar association between different forms of impulsivity in clinical and nonclinical samples may indicate that impulsivity is regulated by fundamentally different mechanisms in psychiatric versus healthy populations, raising serious concerns over the ability of preclinical models to generate data translatable to the clinical conditions they hope to address.
However, many animal studies that have attempted to determine whether various forms of impulsivity co-occur have necessarily used relatively small samples and may therefore have been underpowered. The recurrent use of the rGT in our laboratory has allowed us to collect data from hundreds of rats, an experimental cohort large enough to perform a reliable meta-analysis that provides population-level insight into the association between choice and motor impulsivity. We believe this work marks the first reliable population-level examination of the association between decision-making and motor impulsivity using a behavioural preclinical model. This meta-analysis demonstrates that animals that make more disadvantageous economic choices also exhibit greater motor impulsivity. These highly motor impulsive (H-MI) rats are also slower to learn the task and faster to make a decision.
Methods
Animals
All animals were male Long-Evans rats (Charles River Laboratories) weighing 250–300 g at the beginning of the experiments. They were food-restricted to 85% free-feeding weight, fed 14 g of standard rat chow per day and given access to water ad libitum. Rats were pair-housed and maintained under a 12-hour reverse-light cycle (lights off at 8 am). All housing and procedures were monitored and approved by the Canadian Council of Animal Care and the Animal Care Committee of the University of British Columbia.
All animals trained on the rGT in C.A.W.’s laboratory were considered for inclusion in this analysis. Any animals that received pretraining manipulations (i.e., lesions), that were not pair-housed, or that were fed special diets were excluded, resulting in a population naive to experimental manipulation. Animals from 13 experimental cohorts were included (8–32 rats per cohort), amounting to 240 rats in all. Animals were excluded if choice behaviour or premature response data were incomplete from any session. We excluded 29 animals on the basis of these criteria, leaving a final sample of 211 rats.
The rGT
Testing was performed in standard Med Associates 5-hole operant chambers, each enclosed in a ventilated, sound-attenuating cabinet (Med Associates Inc.). Chambers were controlled by software written in Med-PC by C.A.W.8 Animals were first trained to nosepoke an illuminated aperture within 10 seconds for a sucrose pellet reward (45 mg; Bioserv) as per 5-CSRT training.9 Animals were trained for up to 10 sessions on a forced-choice variant of the rGT before testing on the full rGT began in order to ensure equal exposure to all contingencies associated with the task and minimize any potential primacy effects.
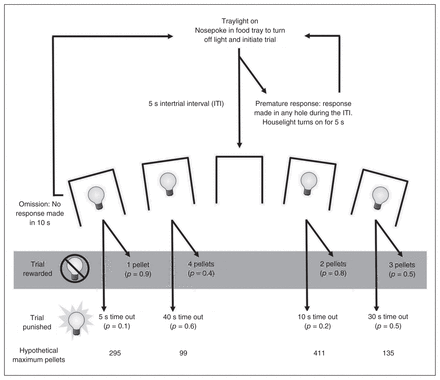
The structure and training of the rGT has been extensively discussed in previous papers.8,19–21 A task schematic is provided in Figure 1. Each session lasted 30 minutes, with animals initiating individual trials at will. At the beginning of the session, the light in the food tray was illuminated, indicating the opportunity to begin a trial. Animals were required to nosepoke in the food tray, thus initiating a new trial and extinguishing the tray light, and a 5-second intertrial interval (ITI) began. After the ITI, stimulus lights in holes 1, 2, 4 and 5 were illuminated for 10 seconds. The trial was scored as an omission if animals failed to nosepoke in an illuminated aperture, at which point the tray light was reilluminated and animals could start a new trial. A response in any illuminated hole turned off all stimulus lights and led to either onset of the tray light and delivery of reward, or the start of a time-out punishment period during which the aperture’s stimulus light flashed at 0.5 Hz and reward could not be earned.
Schematic of the rodent gambling task (rGT). Each trial began with the illumination of the tray light. A nosepoke in the tray extinguished the tray light and initiated a 5-second intertrial interval (ITI), during which all lights in the chamber were off. After the ITI, stimulus lights were illuminated in apertures 1, 2, 4 and 5, each of which was associated with a different schedule of reward/punishment. If the animal nosepoked one of the apertures within 10 seconds, the animal was rewarded or punished according to the schedule associated with that aperture. The size of reward and duration of punishment for each option are indicated on the schematic; the p value in brackets beneath each of those indicates the probability of a win or loss on any given trial. On a rewarded trial, the tray light was illuminated and the requisite pellets dispensed. A response at the tray then initiated a new trial. On a punished trial, the light in the chosen aperture flashed at a frequency of 0.5 Hz for the duration of the time-out period; all other lights were extinguished. At the end of the time out, the tray light was once again illuminated and the animal could initiate a new trial. A nosepoke at an aperture during the ITI was scored as a premature response and initiated a 5-second time-out period during which the house light was illuminated. Failure to make a response at an aperture within 10 seconds of the stimulus lights being illuminated was scored as an omission; the stimulus lights were extinguished, the tray light was once again illuminated, and the animal was able to initiate a new trial. Adapted with permission from Zeeb and colleagues.8
Each hole was associated with a different reinforcement schedule. Rewards ranged from 1–4 sucrose pellets (P1–P4), with probability and duration of time-out punishment increasing as potential reward size increased (Fig. 1). In contrast to the original IGT design, rewarding outcomes were not more prevalent at the start of each test period. Instead, the probability of reward or punishment for a given option did not change across the session; the schedule was pseudorandom throughout to ensure a fair distribution of rewards and punishments that accurately reflected the contingencies in force. As such, P2 was consistently the most economically advantageous option, with 2 sugar pellets upon reward and a relatively small probability of a shorter punishment. Despite larger rewards, P3 and P4 were accompanied by lengthy time-outs with a high probability of occurrence, and thus choice of these holes was disadvantageous. Choice of P1 may reflect a risk-averse strategy,8 but the low reward size still made this a suboptimal strategy compared with P2. To control for side biases, reward/punishment contingencies were counterbalanced across the aperture array, resulting in 2 arrangements of the options (from left to right, P1–P4–P2–P3 v. P4–P1–P3–P2).
In parallel to the 5-CSRT, any nosepoke response made at the array during the ITI was scored as a premature response and punished by a 5-second time out during which the house light was illuminated and the animal was unable to earn reward. The number of premature responses made per session provided a measure of motor impulsivity on the task. At the end of the time-out period, the tray light was illuminated and animals could initiate a new trial.
Behavioural measurements and data analysis
The rGT allows a number of performance measures to be recorded for each session, of which the following were included in the present meta-analysis. Choice of each option (P1–P4) was calculated as a percentage ((number of choices of a particular option ÷ total choices made) × 100), thus minimizing the effects of variation in total number of completed trials on preference. As for analysis of data from the 5-CSRT, premature responses were also calculated as a percentage ((number of premature responses ÷ number of completed trials) × 100). Choice latency (in seconds) was calculated as the delay between the onset of the stimulus lights and a nosepoke response in one of the holes. Collection latency (in seconds) was defined as the time taken to collect the reward once it had been delivered. Both choice latency and collection latency were averaged across the session for each option.
Statistical analysis
All data analysis was performed using SPSS version 20.0 for Mac (IBM). Any percentage variables were arcsine transformed to minimize artificial ceiling effects.8,22 Data were analyzed using repeated-measures analysis of variance (ANOVA), with choice (P1–P4) and session (1–5) as within-subjects factors. Data were compiled from the first free-choice rGT session to the last naive (i.e., pre-experimental) session. Prior to experimental manipulation each cohort was trained to behavioural stability, as demonstrated by a lack of significant session and choice × session terms in a repeated-measures ANOVA. As individual animals/cohorts differed in the number of sessions needed to reach stability, comparisons of baseline behaviour used the 5 sessions before experimental manipulation rather than from a fixed time point (e.g., sessions 20–25). Task acquisition was defined as occurring during the first 20 free-choice sessions, following protocol established by other studies using the rGT,8,19–21 and analyzed in bins of 5 sessions.
Animals were subdivided into quartiles based on premature response rates at baseline, and the quartile with the highest rate of premature responding was considered the H-MI group (n = 52, mean 25.52 ± 3.08), whereas the quartile with the lowest rate of premature responding was considered the least motor impulsive group (L-MI; n = 52, mean 7.00 ± 2.18). This variable was added to all ANOVAs as a between-subjects factor (group: 2 levels, L-MI or H-MI) to determine whether group differences existed at baseline or during task acquisition. If any analyses produced significant group × choice interactions, we conducted further ANOVAs comparing choice of individual options between groups. To determine whether an association existed between motor impulsivity and choice behaviour across the population at baseline, a single score was needed that conveyed the level to which choice was optimal. This score variable was calculated as choice of P2 (most advantageous) – P4 (least advantageous), with higher scores indicating a more optimal choice strategy. Whether the degree of premature responding exhibited correlated with this variable, as well as with the average choice and collection latency, was then determined. We considered results ot be significant at p < 0.05 for all analyses.
Results
Choice behaviour at baseline
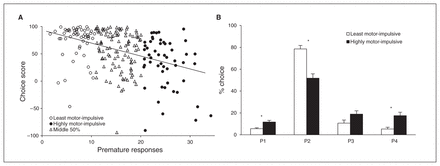
As has been previously demonstrated, rats were able to discriminate between options on the rGT and learned to choose P2, the most advantageous option.8 Animals showed clear choice preferences at baseline, favouring P2 over P3, P4 and P1, in order of descending preference (choice: F3, 630 = 280.33, p < 0.001). Although previous studies suggested an inverse association between optimal choice and premature responding, sample sizes were insufficient to be conclusive.21 When baseline data from all animal cohorts were combined for analyses, there was indeed a significant negative correlation between advantageous choice and premature responding: animals with a higher score exhibited less motor impulsivity (Fig. 2A, r211 = −0.41, p < 0.001). Similarly, choice of P2 at baseline correlated with lower rates of premature responding, while choice of P4, the most disadvantageous option, correlated with higher rates of premature responding (P2: r211 = −0.37, p < 0.001; P4: r211 = 0.36, p < 0.001; data not shown). Partial correlations were performed to control for any confounding effects of using difference scores in computing these statistics. These analyses also returned significant correlations between choice of P4 and premature responding when controlling for P2 (r211 = −0.134, p = 0.05) as well as between choice of P2 and premature responding when controlling for P4 (r211 = 0.183, p = 0.007).
Analysis of the population revealed a negative correlation between task performance and rate of premature responses; animals that made more premature responses also adopted a more disadvantageous choice strategy, preferring the riskier options that were associated with larger rewards but also disproportionately large punishments (A). This association between motor impulsivity and choice strategy was consistent across all 4 options but was most pronounced when the most (P2) and least (P4) economically advantageous choices were considered (B). Data are means ± standard errors of the mean. *p < 0.05.
When animals were divided into L-MI and H-MI groups based on their rate of premature responding, clear differences were observed in choice preferences (Fig. 2B, choice × group: F3, 306 = 14.91, p < 0.001). These group differences extended across most options, with L-MI animals choosing P2 more than H-MI animals (P2, group: F1, 102 = 26.49, p < 0.001) and H-MI animals choosing P1 and P4 more than L-MI animals (P1, group: F1,102 = 16.98, p < 0.001; P4, group: F1,102 = 23.20, p < 0.001). Choice of P3 did not differ significantly between the groups (P3, group: F1, 102 = 3.13, p = 0.08).
Choice behaviour during acquisition
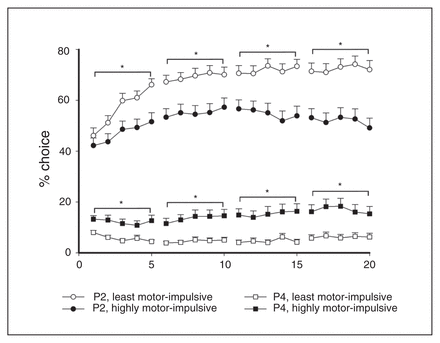
Significant between-group contrasts were also revealed in choice behaviour during task acquisition. The L-MI animals chose P2 more than H-MI animals almost immediately (P2, sessions 1–5, group: F1,102 = 6.91, p = 0.010; Fig. 3) and maintained that advantage for the rest of the acquisition period (P2, sessions 6–10, group: F1,102 = 11.23, p < 0.001; sessions 11–15, group: F1,102 = 15.04, p < 0.001, sessions 16–20, choice × group: F1,100 = 18.76, p < 0.001). This pattern was reversed with P4, as H-MI animals immediately showed a higher preference for the least advantageous option (P4, sessions 1–5, group: F1,102 = 16.71, p < 0.001; Fig. 3) and maintained this preference for the duration of acquisition (P4, sessions 6–10, group: F1,102 = 17.79, p < 0.001; sessions 11–15, group: F1,102 = 18.25, p < 0.001; sessions 16–20, group: F1,102 = 15.14, p < 0.001).
Differences in choice behaviour between the highly motor-impulsive (H-MI) and least motor-impulsive (L-MI) animals were apparent during acquisition, and significant group differences in choice of the most and least advantageous options emerged as early as the first 5 sessions. These differences became more pronounced throughout acquisition. Data are means ± standard errors of the mean. *p < 0.05.
Other behavioural measures at baseline
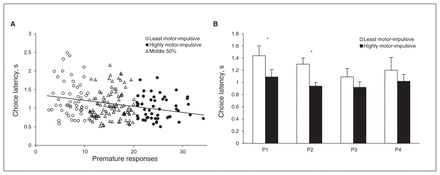
In general, H-MI animals were quicker to make a choice (group: F1, 97 = 9.51, p = 0.003; Fig. 4B), and there was a significant negative correlation between premature responses and choice latency (r211 = −0.25, p < 0.001; Fig. 4A). When latency to choose each individual option was considered separately, H-MI rats were significantly faster than L-MI rats only when choosing P1 and P2 (choice × group: F3, 291 = 3.80, p = 0.011; P1, group: F1,105 = 16.84, p < 0.001; P2, group: F1,106 = 18.78, p < 0.001; P3: F1,103 = 0.97, p = 0.33; P4: F1,101 = 0.98, p = 0.33). However, when data from L-MI and H-MI animals were analyzed independently, choice latency was equivalent across all options (L-MI, choice: F3,138 = 2.53, p = 0.06; H-MI, choice: F3,153 = 1.84, p = 0.16).
There was a negative correlation between the latency to make a choice and rate of premature responding across the entire population (A). Though the highly motor-impulsive (H-MI) animals were significantly quicker to make a choice overall, the differences in choice latency between H-MI and low motor-impulsive (L-MI) animals achieved statistical significance only on the options P1 and P2 when each option was considered individually (B). There were no within-group differences in choice latency across the 4 options. Data shown are means ± standard errors of the mean. *p < 0.05.
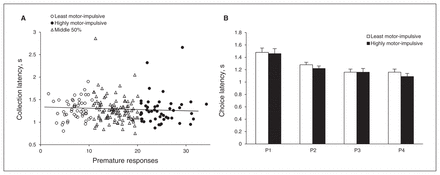
Unlike choice latency, there was no significant correlation between premature responses and collection latency (r211 = −0.049, p = 0.49; Fig. 5A). The L-MI and H-MI animals collected reward equally quickly (group: F1,71 = 2.247, p = 0.14; Fig. 5B). While collection latency differed across options, with larger rewards collected faster than smaller ones, this was equally true across both groups (choice: F3,213 = 29.99, p < 0.001, choice × group: F3,213 = 2.57, p = 0.06).
There was no association between motor impulsivity and speed of reward collection at the population level (A). Likewise, the highly motor-impulsive (H-MI) animals were no quicker to collect reward than the least motor-impulsive (L-MI) animals (or vice-versa), but animals in both groups were quicker to collect reward as reward size increased (B). Data shown are means ± standard errors of the mean. *p < 0.05.
Given that latency measures are often positively skewed and easily biased by extreme responses, these data were examined for outliers that may have been artificially driving the correlational analyses (mean ± 3 standard deviations;22 choice latency: n = 1; collection latency: n = 3). However, when these data points were excluded and the analyses recalculated, r values and statistical significance were unchanged.
Discussion
The present meta-analysis demonstrates that levels of risky decision-making — an aspect of impulsive choice — and motor impulsivity reliably correlate across a large population of rodents. When animals were split into subgroups based on rate of premature responding, H-MI rats clearly exhibited a reduced preference for the most economically advantageous option and increased choice of the disadvantageous options, especially the “high-risk, high-reward” choice. During task acquisition, significant choice differences between H-MI and L-MI animals emerged within the first few sessions and were maintained through behavioural stability. Our study, therefore, provides direct empirical evidence, which has until now been lacking, that different impulsive behaviours are comorbid in healthy controls. Furthermore, motor impulsivity correlated negatively with speed of decision-making, supporting the hypothesis that behavioural disinhibition is related to insufficient deliberation.2,23 While H-MI animals were quicker to make a selection from among the 4 options, they were no faster than L-MI animals to collect reward, indicating that H-MI animals were not simply hyperactive or more motivated. Collectively, these observations that high motor impulsivity, risky choice and fast decision-making are associated at the population level in rats support the study of the extremes of this distribution as a proxy for clinically significant ICDs in which multiple forms of impulsivity typically co-occur.
In the rGT, premature responses are punished by time-out periods, which decrease the amount of time within a session that the animal is able to initiate trials. We therefore considered the possibility that motor impulsivity could contribute to poor task performance by decreasing the time available to explore the task’s contingencies. A similar concern would be that favouring P4 early in training could impair learning in the same fashion; the long and frequent punishments associated with this choice would also limit the number of trials initiated by an animal that preferred P4. However, if the maladaptive choice strategy exhibited by H-MI animals was the product of inexperience due to shortened sessions, we would expect to see slower (relative to L-MI animals) but continuous improvements in task performance. Furthermore, H-MI animals should eventually reach the same baseline performance as L-MI animals if given sufficient time. In contrast, both L-MI and H-MI animals approached behavioural stability during the second week of training, and basic strategies and preferences appeared to be set at this time with little further improvement in either group. As noted previously, the primary shift in behaviour with regards to P2 occurs during the first 5 sessions.8,19–21 The H-MI animals do not increase their selection of P2 past this relatively early stage, suggesting that additional exposure to the various probabilities of reinforcement would not improve task performance. This observation suggests that disadvantageous choice is not an artifact of inexperience or an indication of a learning impairment, but something more integral to the animals’ existing decision-making preferences.
Previous experiments using the rGT found no consistent association between motor impulsivity and decision-making. Pharmacological and environmental manipulations often had dissociable effects on these 2 constructs,8,21,24 encouraging the conclusion that “a universal relationship between baseline levels of impulsive action and decision-making may not exist.”21 This conclusion is not unique to the rGT; numerous studies using both human and animal samples suggest that different types of impulsivity can be dissociated in terms of their neural and neurochemical basis,6,13–18 and there have previously been no clear demonstrations that choice and motor impulsivity are related in the general population. For example, an experiment using deep brain stimulation in humans found that stimulation did not affect the participants’ ability to learn a task, but did cause them to respond impulsively.25 Although animals screened for very high levels of motor impulsivity (top 7% of population) also exhibit high levels of preference for “smaller, sooner” over “larger, later“ rewards on a delay-discounting task,26 a choice preference also representative of impulsive decision-making,27 other studies have reported dissociations between the 2 measures.16,28,29 These and other work21 posited that different forms of impulsivity may be largely independent in the healthy population, but may co-occur in extreme conditions like pathological gambling. The results presented here contradict that conclusion, suggesting instead that an association between the 2 exists across the entire population, not simply in the most highly impulsive individuals.
Given that increases in all forms of impulsivity co-occur in psychiatric disorders, the lack of concurrence between measures of impulsive choice and action in nonclinical samples would be troubling, as it suggests that studying the regulation of impulsivity in normal populations cannot provide insight into the basis of ICDs. This consideration is particularly relevant for animal research in which models of psychiatric illnesses are often lacking and sample sizes are necessarily restricted. Hence, demonstrating that these forms co-occur in a nonclinical population lends crucial validity to the practice of studying high impulsivity as a proxy for ICDs and suggests that the behavioural differences between clinical and nonclinical populations could be ones of magnitude rather than type. While there may be cases in which clinically significant impulsivity is categorically different from high levels of impulsivity in the healthy population and, therefore, subject to distinct neurobiological regulation, it seems reasonable on the balance of these and other data to consider clinical impulsivity not as a sui generis syndrome, but as the extreme end of a normal distribution of behavioural expression.30
It is curious then that previous work was unable to delineate this association. It is possible that the sample sizes in the previous studies simply did not provide enough statistical power; the enormous variability of impulsivity exhibited in the present population suggests that possibility. It may also be that the nature of the association between motor and choice impulsivity deserves reconsideration. Perhaps impulsivity should be understood as a behavioural phenotype that arises from the interaction of a number of component processes that share related but distinct mechanisms rather than a unitary behavioural construct that has diverse and often unrelated manifestations. It seems at least possible that impulsivity may be better thought of not as a multifaceted top-down disorder, but rather as the interaction of bottom-up processes that potentiate each other. Disadvantageous decision-making may exist without being accompanied by motor impulsivity, but the presence of motor impulsivity may contribute to poor decision-making by hastening behaviours associated with that choice. Existing motor impulsivity may be further exaggerated in an individual with weak decision-making processes; weak executive control offers little counter to prepotent impulses. These are clearly speculative hypotheses, but the results presented here reinforce the fact that our understanding of impulsivity is far from complete.
Arguably both premature responding and choice of the disadvantageous options may hypothetically arise from insensitivity to negative feedback. The observation of stable preferences suggests that choice was not mediated by a learning impairment; were this the case, we would expect to see gradual improvements in decision-making over time as repeated exposure to the punishments accrued. It is nevertheless possible that differences between individuals’ responses to negative feedback could contribute to both motor and choice impulsivity. Recent work in human samples has suggested that extreme mood states can modulate risky decision-making and other forms of impulsive behaviour.31,32 Furthermore, highly salient stimuli can increase the excitability of the motor cortex.33 Hypothetically, the arousal induced by winning or losing on a given trial could then both influence risky choice via changes in mood while also encouraging premature responding on subsequent trials by priming the animal’s motor output. While these or similar mechanisms could theoretically underlie both behaviours, the present data do not provide any empirical insight into this matter and our consideration of this issue is necessarily speculative.
As is typical in most animal and human laboratory-based tasks, the expression of a choice preference in the rGT is indicated by responding to a cue. We have considered the possibility that both premature responding and impulsive decision-making in the rGT may be attributable to the degree to which animals tend to approach salient stimuli. This is an issue that has received considerable attention in the literature recently, in that “sign-tracking” (the preferential approach to a stimuli predictive of reward over the reward itself) has been suggested as a key marker of vulnerability to addiction,34,35 a disorder associated with high impulsivity.36–38 Furthermore, animals selectively bred to exhibit disinhibited behaviour show higher levels of sign-tracking and also higher levels of impulsive choice, as measured by increased preference for “smaller, sooner” over “larger, later” rewards in a delay-discounting paradigm.39,40 As such, sign-tracking may enhance impulsive tendencies in both animals and humans,41 a conclusion that may speak to the cognitive processes involved in impulse control rather than those reflective of methodological artifacts inherent within the design of animal paradigms, such as the rGT. However, sign-tracking was not found to correlate with either premature responding or delay-discounting in rats.16 Although the sample size in this latter study was small, the analysis was sufficiently well powered to detect correlations between sign-tracking and conditioned hyperactivity to food delivery. If the Pavlovian approach to reward-related stimuli was the key factor mediating an association between different forms of impulsivity, we would expect this correlation to have been as easily detected.
With respect to the latency data, the fact that H-MI and L-MI animals did not differ in reward collection latency and that they were uniformly faster to collect larger rewards indicates H-MI animals are not simply hyperactive in the presence of reward or differentially sensitive to changes in reward magnitude. Furthermore, the lack of effect of choice type on response latency within both H-MI and L-MI groups suggests that the economic utility of a choice and the decision speed can be dissociated. Nevertheless, H-MI rats were generally faster to make a choice than L-MI rats, indicating that their inferior decision-making could result, at least in part, from reduced deliberation. The influence of deliberation time on decision-making has been well documented in the literature; as early as 1966, researchers described insufficient deliberation as “reflection impulsivity,” a behaviour characterized by the failure to adequately collect and synthesize information before making a decision.42 More recent work has demonstrated that both current and abstinent substance abusers sampled less information than healthy controls before making a decision regarding probability, leading to poorer task outcomes.43 While this has been described as a separate manifestation of impulsivity,2,23,43,44 it is possible that such a processing style could exaggerate already suboptimal decision-making (like that demonstrated by H-MI animals). This presents a potential example of the bottom-up copotentiation theorized previously. Impulsivity elevates impulsivity; an animal’s already compromised decision-making abilities could be further hampered by insufficient reflection.
Exploring the neurologic underpinnings of ICDs, if they follow this theoretical model of bottom-up synergism, may perhaps benefit from a different research approach. While it has certainly been useful to delineate the distinct neurobiological mechanisms underlying different forms of impulse control, such an approach has not generated new treatments for ICDs. Rather than focusing on selective manipulations targeting individual brain regions or neurotransmitter receptor subtypes that typically affect only 1 aspect of impulsivity (as per studies by Sun and colleages,45 Winstanley and colleagues,46 Bari and Robbins,47 Murphy and colleagues,48 Fletcher and colleagues49 and Mendez and colleagues50), holistic experimental models that engage multiple neural loci and neurochemical systems may more accurately mimic the real-world neural processes behind ICDs (for a discussion, see the studies by Winstanley6 and Baarendse and colleagues24). While there is a well-documented causal relationship between traumatic brain injury (TBI) and major depression,51–57 mania and borderline personality disorder, in which high impulsivity is a core symptom, can also arise after TBI.41,58 Furthermore, mania has been documented following brain tumours, vascular disorders and multiple sclerosis.42,59 Hence, it is possible that ICDs could result from similar non-specific insults, and future research may benefit from the inclusion of such models.
Limitations
It is important to note that choice impulsivity is a multidimensional construct that can be subdivided into risky choice and intertemporal choice (often presented as a decision between “smaller, sooner” or “larger, later” rewards). The association between these subcategories of impulsivity is muddled and somewhat contradictory. Despite the distinction between them, the argument could be made that the tasks used to measure both processes demand a similar strategy of self-restraint defined by the avoidance of “superficially seductive options”60 in favour of options that confer more benefit over the long run. Some studies have found no association between delay-discounting and IGT scores in healthy controls,60 hospitalized adolescents61 and cocaine users,62 while other work has found a strong association between them in cocaine-dependent patients.63 The lack of association between these phenomena is surprising as certain risk factors decrease performance on both measures,62 but this could be indicative of a global impairment that is not specific to a capacity taxed by either paradigm. Hence, while it remains possible that these forms of impulsive choice are underpinned by similar neurobiological mechanisms, we nevertheless adopted a conservative approach and restricted our conclusions to choice impulsivity concerning risky decision-making and not intertemporal choice.
In addition, while most meta-analyses benefit from the inclusion of data from various researchers, the data examined herein was limited to that produced by our laboratory. We believe this has negligible impact on our findings; the research drawn from here was conducted by a number of individuals over the course of 4 years, introducing some measure of environmental variability.
Conclusion
The present meta-analysis has shown that motor and choice impulsivity co-occur across a rodent population, similar to the manner in which impulsive behaviours manifest in clinical populations. This novel finding is important as it validates the common practice of studying impulsivity in non-clinical populations to gain insight into ICDs. Accordingly, it may be practical to go beyond highly specific manipulations, like the modulation of single neurotransmitters or brain areas that affect only 1 measure of impulsivity, in order to better simulate the real-world conditions that give rise to ICDs.
Acknowledgements
This work was supported by operating grant funding to C.A. Winstanley from the Canadian Institutes of Health Research (CIHR), The National Sciences and Engineering Council of Canada, the Michael Smith Foundation for Health Research (MSFHR), Parkinson Society Canada, the Institute for Research into Pathological Gambling and Related Disorders (now the National Council for Responsible Gambling) and the Canadian Foundation for Innovation. C.A. Winstanley also receives salary support through the MSFHR and CIHR New Investigator Award program.
Footnotes
Competing interests: None declared.
Contributors: M. Barrus designed the review, performed the analysis and interpretation, prepared the article and can give final approval of the article as submitted. J. Hosking collected data, substantially assisted with the analysis and drafting of the article and can give final approval of the article as submitted. F. Zeeb designed the task, collected data, assisted with the preparation of the article and can give final approval of the article as submitted. M. Tremblay collected data, assisted with preparation of the manuscript and can give final approval of the article as submitted. C. Winstanley provided funding, conceived of the review, substantially assisted with the drafting of the article and can give final approval of the article as submitted.
- Received February 10, 2014.
- Revision received May 2, 2014.
- Accepted June 9, 2014.