Abstract
Background: There is evidence to suggest that obsessive–compulsive disorder (OCD) is associated with structural abnormalities in cortico–striato–thalamic circuits, yet the extent of white matter abnormalities is not well established. In this study, we used diffusion tensor imaging (DTI) to examine white matter integrity in specific regions of interest (ROIs) in patients with OCD.
Methods: Patients with OCD and sex-, age- and IQ-matched healthy controls underwent DTI. The primary objective was to explore whether patients with OCD had white matter abnormalities in the anterior limb of the internal capsule (ALIC), the uncinate fasciculus, the genu of the corpus callosum and the cingulum. The secondary objective was to evaluate the relation between fractional anisotropy and mean diffusivity in these ROIs and other clinical variables (including age at onset of OCD, OCD severity and levels of depressive and anxiety symptomatology) in patients with OCD.
Results: There were 15 patients and 17 controls enrolled in our study. Compared with healthy controls, patients with OCD showed increased fractional anisotropy in bilateral regions of the ALIC adjacent to the body of the caudate, as well as decreased fractional anisotropy in the right anterior limb near the head of the caudate. Patients also had decreased mean diffusivity in the body of the right cingulum and the left anterior cingulum compared with controls. Correlational analyses revealed significant associations of fractional anisotropy and mean diffusivity in select circuits with OCD, depression and anxiety severity scores.
Limitations: Inclusion of patients with OCD receiving pharmacotherapy may have been a limitation. In addition, the patients were heterogeneous in terms of their obsessive–compulsive symptom profiles; we did not distinguish between different obsessive–compulsive symptom dimensions.
Conclusion: The study results provide further evidence for OCD-related white matter abnormalities in the ALIC and cingulum, consistent with a corticostriatal model of OCD.
Introduction
In the DSM-IV,1 obsessive–compulsive disorder (OCD) is classified as an anxiety disorder that is characterized by recurrent unwanted ideas, thoughts or impulses that cause anxiety (obsessions); and/or repetitive, irresistible and often ritualized behaviour aimed at reducing anxiety or performed according to rigid rules (compulsions). Although prevalence estimates have varied considerably across surveys,2 most studies have found a lifetime rate of 2%–3% in the general population, suggesting that a substantial number of individuals worldwide have this disabling condition.
Neuroimaging studies investigating the pathophysiology of OCD have generally supported the hypothesis that frontostriatal circuitry is key. Specifically, findings from structural, functional and positron emission tomography (PET) studies3,4 have demonstrated that regions in the prefrontal cortex and the connecting striatal areas are implicated in the psychobiology of OCD.
Although most magnetic resonance imaging (MRI) work has focused on grey matter abnormalities in patients with OCD, there are several tiers of evidence that white matter abnormalities are also associated with the condition. First, voxel-based morphometry (VBM) studies have pointed to the involvement of the anterior limb of the internal capsule (ALIC) and cingulum bundle (for example, see Cannistraro and colleagues5). Second, neurosurgical reports have emphasized the importance of the ALIC and the cingulum in OCD by implicating these as target sites for the neurosurgical treatment of OCD using ablative procedures (see the review by Rauch6). Third, studies with diffusion tensor imaging (DTI), an MRI technique that is useful in assessing the directionality and integrity of white matter tracts by quantifying water diffusion in brain tissue, suggest dysfunction of corticostriatal circuits in patients with OCD. Whole-brain and selected regions-of-interest (ROIs) analyses found variable fractional anisotrophy values in the anterior cingulate and the internal capsule, the bilateral semioval centre extending to the medial frontal white matter, the subinsular white matter, the corpus callosum and the parietal white matter in patients with OCD.7
In a recent literature review, Fontenelle and colleagues7 described recent DTI studies of patients with OCD. This review included findings from 8 studies on the anatomic localization of fractional anisotropy changes, the heritability of these white matter abnormalities and associations with treatment response to serotonin reuptake inhibitors (SRIs) and with the severity of various obsessive–compulsive symptom dimensions. Further investigation is, however, warranted. First, reports from studies of OCD have not been consistent in terms of the nature, extent or regions of white matter abnormalities or in terms of the direction (i.e., increase/decrease) of fractional anisotropy changes. Tract-based spatial statistics (TBSS), a DTI analysis pipeline that forms part of the FMRIB Software Library (FSL), overcomes some of the shortcomings of other DTI analyses based on VBM, offering the advantage of improved sensitivity and interpretability of analysis of multisubject diffusion imaging studies, and may therefore be able to address some of the inconsistencies.8 Second, there have been few reports on the correlations between DTI findings and clinical variables, such as OCD severity,9,10 major symptom dimensions of OCD,11 neuropsychologic functioning12 and treatment response.13
Our primary objective was to investigate whether adult patients with OCD have white matter abnormalities in selected ROIs that have been implicated in OCD pathology (i.e., the ALIC, the cingulum bundle, the corpus callosum and the uncinate fasciculus). Our secondary objective was to investigate the relation between fractional anisotropy and mean diffusivity findings in these ROIs and selected clinical variables in patients with OCD, including age at onset of OCD, OCD severity, levels of depressive and anxiety symptoms and treatment status. The fractional anisotropy metric is sensitive to the colinearity of the axonal fibres and gives a representation of white matter integrity, whereas mean diffusivity represents local diffusivity and generally indicates the presence of diffusion barriers.
Methods
Participants
Participants for this study were recruited for a pharmaco-imaging (functional MRI; fMRI), crossover study of single-dose escitalopram and placebo in patients with OCD. Patients were referred to our unit from a wide range of sources, including specialist psychiatrists, community-based primary care practitioners, consumer advocacy organizations, as well as through media advertisements and the assistance of the Mental Health Information Centre of Southern Africa. Controls were recruited from the community and our university campus through media advertisements. All participants were first screened for inclusion over the telephone by a postgraduate research assistant.
After we obtained their voluntary informed consent, participants who passed the telephone screening were personally interviewed by a clinical psychologist to screen for DSM-IV psychiatric disorders using the Mini International Neuropsychiatric Interview Plus (MINI Plus), version 5.14,15 All interviews took place at the MRC Unit on Anxiety and Stress Disorders, Stellenbosch University. We included adult patients with OCD and age-, sex- and IQ-matched healthy controls if they were right-handed, had no history of clinically significant substance or alcohol abuse and were white South Africans. Patients were included if they had a primary diagnosis of OCD and no other comorbid psychiatric conditions. Patients with OCD who had specific comorbid phobias were allowed to participate if, according to clinical impression, the phobia was not the primary diagnosis and did not substantially interfere with their daily functioning. Patients were also included if they were free of psychotropic medications or on a stabilized treatment regimen (i.e., medication limited to a single agent from the selective SRI class, administered at a steady dose and taken for at least 8 weeks).
The study protocol and patient information and consent forms were approved by the Health Research Ethics Committee of the Faculty of Health Sciences, University of Stellenbosch; by the Ethics Committee of the University of Cape Town; and the Medicines Control Council of South Africa. Written informed consent was obtained from all participants before study procedures began.
Interviewing material
Scales that were administered included the Yale–Brown Obsessive–Compulsive Scale (Y-BOCS)16 to assess the severity of OCD, the Montgomery–Åsberg Depression Scale (MADRS)17 to assess the presence and severity of depressive symptoms, the Hamilton Anxiety Scale (HAM-A)18 to assess anxiety levels and the Edinburgh Handedness Inventory19 to determine hand preference. The Wechsler Abbreviated Scale of Intelligence (WASI),20 an abbreviated form of the South African Wechsler Adult Intelligence Scale (SAWAIS-III), was used to obtain an IQ for participants.
The Y-BOCS16 comprises 10 items, with each item rated on a 5-point scale. Patients with OCD were included if they were at least moderately symptomatic on the Y-BOCS (i.e., a Y-BOCS total score greater than 16). The MADRS17 rates 10 depression symptoms on a scale from 0 to 6, with higher scores reflecting more severe depression. Participants with MADRS scores greater than 20, indicating possible comorbid depressive syndromes, were excluded. The HAM-A is a 14-item rating scale designed to assess the presence and quantify the severity of physical and psychologic symptoms of anxiety.18 Each item is rated on a 5-point scale, with each item contributing 0–4 points to the total HAM-A score. The WASI applied here was a 4-test estimation that included vocabulary and performance quotients as well as global IQ.
Procedures
After screening and clinical assessments, participants underwent DTI.
Image acquisition
Diffusion tensor images were acquired at the Cape Universities Brain Imaging Centre on a Siemens Magnetom 3-T Allegra scanner with the following parameters: repetition time 8800 ms, echo time 88 ms, diffusion weighted volumes in 30 directions with b = 1000 s/mm2 and a single unweighted volume (b = 0 s/mm2). The in-plane resolution was 2 × 2 mm2, and the slice thickness was 2.2 mm. The sequence was repeated 3 times.
Image analysis
We performed DTI analyses in FSL, using TBSS8 as well as custom tools in MATLAB (Mathworks Inc.). Eddy current correction was performed in FSL on each of the 3 acquisitions separately. Subsequent preprocessing was then performed in MATLAB as follows. The 3 acquisitions were coregistered by applying affine transformations to all volumes using the un-weighted (b = 0 mm/s2) volume of the first acquisition as a reference. For each of the 3 coregistered acquisitions, outliers were determined relative to the tensor estimate by calculating the Z value at the 25th and 75th percentiles and ignoring values 3 standard deviations (SD) away from the mean. The 3 acquisitions were then averaged, carefully taking the removed outliers into account.
Returning to FSL, fractional anisotropy images were created by fitting a tensor model to the raw diffusion data. Here it was confirmed that for fractional anisotropy > 0.2, the number of outliers rejected were insufficient to remove data points after the averaging. Brain extraction was subsequently performed with the BET utility in FSL.21 The participants’ fractional anisotropy data were then aligned into a common space, using the nonlinear registration tool FNIRT.22,23 The mean fractional anisotropy image was created next and then “thinned” to create a mean fractional anisotropy skeleton that represents the centre of the white matter tracts for each participant in the group. Each participant’s aligned fractional anisotropy data were then projected onto this skeleton. Following this, mean diffusivity images were calculated and subjected to the identical processing pipeline as the fractional anisotropy images, resulting in corresponding mean diffusivity skeleton representations. The 4 ROIs (ALIC, cingulum bundle, corpus callosum and uncinate fasciculus) were defined by using the ICBM-81 white matter atlas as a template to mask those regions.24
The TBSS pipeline we described is a robust method for limiting the impact of residual misalignment and partial voluming — shortcomings typical of voxel-based analysis in DTI25 — by only considering white matter voxels that are part of white matter tract centres common to the study cohort.8 In addition, TBSS reduces the problem of multiple comparisons and increases statistical power owing to a smaller search volume for significant voxels. Thus this technique is well suited for small cohort studies26,27 such as ours.
Statistical analysis
Demographic characteristics and aspects of the clinical data were analyzed using SPSS 17.0 for Windows (SPSS Inc.). Statistical analyses using χ2 tests (for categorical data) and Student t tests (for interval and ratio-level data) were undertaken to investigate whether the groups significantly differed in terms of demographic characteristics (sex and age) and clinical scores (HAM-A and MADRS), respectively. Nonparametric linear correlation analyses were performed for the OCD subset using FSL randomize.
Fractional anisotropy and mean diffusivity data (masked for the 4 tracts) were analyzed using permutation testing and threshold-free cluster enhancement by applying an unpaired 2-sample t test to the data for the between-group comparison (patients with OCD compared with healthy controls). The significance level was Bonferroni-corrected for 4 clusters. This lowered the threshold of significance from p < 0.05 to p < 0.0125 after correction. Permutation-based linear correlation analyses were performed on mean fractional anisotropy and mean diffusivity data in each ROI in FSL randomize. Demographic data (sex, age) and clinical variables (age at onset of OCD; Y-BOCS, HAM-A and MADRS scores; and treatment status) were included as independent variables in the relevant analyses.
Results
Fifteen patients with OCD and 17 sex-, age- and IQ-matched controls were included in the study. Demographic and clinical data for both groups are summarized in Table 1.
Demographic and clinical characteristics of patients with obsessive–compulsive disorder versus healthy controls
Scores on the Y-BOCS ranged from 17 to 32 (mean 22.87, SD 3.64), indicating moderately severe obsessive–compulsive symptomatology. Patients with OCD had significantly higher levels of depression and anxiety (both p < 0.001) than controls.
Nine of 15 patients with OCD were receiving pharmacotherapy at the time of the study: 3 were on sertraline (150 mg, 125 mg, 300 mg), 3 on fluoxetine (40 mg, 40 mg, 60 mg), 2 on escitalopram (10 mg and 5 mg) and 1 on paroxetine (40 mg). Treatment was stabilized for all of these participants. Of note, however, is that all patients were symptomatic enough in terms of their OCD to be included in the study (i.e., Y-BOCS total score > 16).
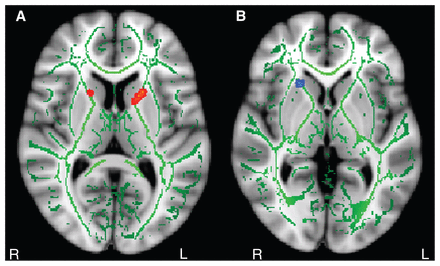
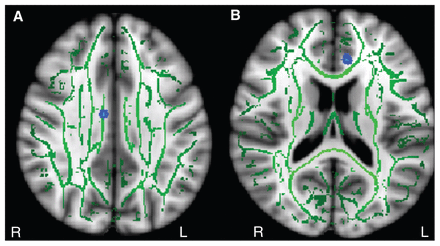
Compared with controls, patients with OCD showed increased fractional anisotropy in bilateral regions of the ALIC closer to the posterior limbs and the body of the caudate, as well as decreased fractional anisotropy in the right anterior limb (near the head) of the caudate structure (Fig. 1, Table 2). Patients with OCD also displayed decreased mean diffusivity in the right cingulum body region and left anterior cingulum compared with controls (Fig. 2, Table 2). There were no significant differences between patients with OCD and controls in terms of fractional anisotropy and mean diffusivity in the other ROIs (i.e., the genu of the corpus callosum and the uncinate fasciculus).
An axial representation of the fractional anisotropy changes in the anterior limb of the internal capsule (ALIC) in patients with obsessive–compulsive disorder (OCD) versus controls. (A) The red regions indicate increased fractional anisotropy bilaterally in the ALIC of patients with OCD compared with controls, and (B) the blue region indicates decreased fractional anisotropy on the right-hand side of the ALIC in patients with OCD compared with controls.
An axial representation of mean diffusivity changes in the cingulum of patients with obsessive–compulsive disorder (OCD). The blue regions indicate decreased mean diffusivity in the patients with OCD compared with controls for the (A) right cingulum body region and (B) left anterior cingulum.
Regions of fractional anisotropy and mean diffusivity abnormalities in patients with obsessive–compulsive disorder and healthy controls (p < 0.01, corrected)
The findings revealed no significant correlations between the total Y-BOCS score and fractional anisotropy in any of the ROIs. However, the MADRS (r = 0.4, p = 0.024) and HAM-A (r = 0.47, p = 0.006) scores correlated significantly with fractional anisotropy in the bilateral regions of the ALIC. There was a significant positive correlation between the total Y-BOCS score and mean diffusivity in the left anterior cingulum (r = 0.33, p = 0.049), and there was a significant negative correlation between MADRS (r = −0.47, p = 0.006) and HAM-A (r = −0.43, p = 0.015) scores and mean diffusivity in the right cingulum body region. No significant associations were demonstrated between fractional anisotropy and sex, age at onset of OCD and treatment status. Similarly, no significant associations were found between mean diffusivity and these 3 variables.
Discussion
Our findings indicate that, first, white matter abnormalities occur in the neuronal circuitry connecting striatal and cortical regions (ALIC, cingulum) in patients with OCD and, second, that DTI findings (fractional anisotropy and mean diffusivity) correlate significantly with clinical severity scores (i.e., OCD, depressive and anxiety symptoms).
We applied an ROI approach to examine specific white matter regions that have been implicated in the pathology of OCD. In patients with OCD, we found evidence of white matter abnormalities in the ALIC and the cingulum, with increased fractional anisotropy in bilateral regions of the ALIC closer to the posterior limbs and the body of the caudate, as well as decreased fractional anisotropy in the right anterior limb (near the head) of the caudate nucleus in comparison with controls. These findings are consistent with those of previous DTI studies that have demonstrated increased fractional anisotropy in the left cingulate bundle and internal capsule,5 and reduced fractional anisotropy in certain cingulate regions9 and the right cingulate bundle5 in patients with OCD. They are also consistent with a range of findings implicating cortico–striato–thalamic pathways in the pathophysiology of OCD; the ALIC connects the frontal cortices with the thalamus, and the cingulum connects the cingulate cortex to limbic cortices28 as well as interconnecting a range of other regions, such as the prefrontal cortex, parahippocampal gyrus, striatum and a number of thalamic and limbic nuclei.
Whereas findings that fractional anisotropy is both increased and decreased in patients with OCD compared with controls may seem contradictory, they are consistent with previous fMRI research. Increased fractional anisotropy in bilateral anterior regions of the internal capsule in patients with OCD may reflect increased connectivity in regions closer to the thalamus, whereas decreased fractional anisotropy in the right anterior internal capsule closer to prefrontal projections may indicate disrupted or decreased demyelination in those regions or decreased fibre density or coherence. Although, unlike other groups,10 we did not find a correlation between increased fractional anisotropy and OCD severity, we did find an association between increased fractional anisotropy in the ALIC and depression and anxiety scores, consistent with increased activity/connectivity in this area.
Maps of parallel diffusivity (which are thought to represent myelination or axon packing density) may be used in future investigations to assess this hypothesis. Indeed, a recent study demonstrated that radial diffusivity is increased and axial diffusivity unchanged in the corpus callosum of patients with OCD compared with controls.29 These findings suggest a possible demyelination process in the commissural fibres in patients with OCD.
The lateralization of findings noted here may simply represent a false-positive result, but it is also consistent with previous work demonstrating lateralization of imaging findings in patients with OCD. For example, a PET study comparing patients with OCD and controls found enhanced activity in the left amygdala in response to contamination stimuli as well as sensitization effects in the right amygdala in the OCD group.30
We also found decreased mean diffusivity in the left anterior cingulum and the body of the right cingulum in patients with OCD compared with controls. Increased mean diffusivity is associated with increased volume of the extracellular space or with decreased barriers to diffusion in white matter;31 thus it is usually a consequence of neuroinflammation or edema. Whereas the significance of decreased mean diffusivity, as found here, remains uncertain, it may again reflect the increased activity and connectivity in this region, which is thought to be present in patients with OCD. Such putative increased activity may be primary (as perhaps suggested by the correlation of decreased mean diffusivity with depression and anxiety scores) or compensatory (as perhaps suggested by the correlation of increased mean diffusivity with milder obsessive–compulsive symptoms).
There were no significant differences between patients with OCD and controls in terms of fractional anisotropy and mean diffusivity in the genu of the corpus callosum and the uncinate fasciculus. In an earlier study, greater white matter concentration was found in the anterior genu of the corpus callosum in medication-naive pediatric patients with OCD.32 Possibly, changes in the corpus callosum and uncinate fasciculus are particularly apparent in pediatric patients with OCD. Nevertheless, we did not find an association between age at onset of OCD and DTI measures. Recently, decreased fractional anisotropy values in the uncinate fasciculus in healthy women were found to correlate with increased expression of low-expressing alleles of the serotonin transporter gene promoter region polymorphism;33 thus, differences across studies may also reflect variability in genotype.
There were a few methodologic differences between the present study and other recent DTI studies using TBSS in patients with OCD. To our knowledge, 3 other studies have performed a TBSS analysis in patients with OCD. One was a whole brain investigation in a Japanese population that examined fractional anisotropy changes and their relation to OCD severity.34 This investigation also included a post hoc tractography analysis. Another whole brain investigation was performed in an Australian population and investigated fractional anisotropy as well as axial and radial diffusivity in patients with OCD.29 The third study that used TBSS employed a whole brain analysis as well as an ROI-based approach to investigate fractional anisotropy, and axial and radial diffusivity in patients with OCD.35 Our findings and the findings across these studies show overlap and are consistent with a corticostriatal model of OCD.
Further work is needed to investigate fully the clinical implications of DTI changes in patients with OCD. Such work would need to establish precise predictors of DTI alterations at baseline and the extent to which these predict response to treatment. In the interim, a corticostriatal model of OCD remains useful in providing a clinical rationale for intervention with both psychopharmacologic and psychotherapeutic treatments that are able to normalize the relevant functional neuroanatomy.
Limitations
Limitations of the study are the inclusion of patients with OCD who were receiving treatment. However, we found no evidence that treatment status affected the findings, with no differences in fractional anisotropy or mean diffusivity in participants on or off pharmacotherapy. Another possible limitation is that the OCD group was heterogeneous in terms of their obsessive–compulsive symptom profiles; the study was not powered to distinguish between different obsessive–compulsive symptom dimensions (e.g., washers/checkers/ hoarders). Our study population was nevertheless relatively homogeneous compared with other OCD groups in the literature; we focused exclusively on participants who were right-handed and white and who had no current psychiatric comorbidities. Inclusion of people with comorbidities may in part account for the inconsistencies in previous imaging studies.
Conclusion
To our knowledge, this is one of the few published DTI studies of patients with OCD that focuses on the ALIC, the uncinate fasciculus, the genu of the corpus callosum and the cingulum as specific ROIs. This also is one of the first studies to include the uncinate fasciculus as an ROI and to implement the TBSS technique. Moreover, very few studies have included correlational analyses of the imaging data with clinical features. As such, our findings contribute to the limited literature on white matter abnormalities in patients with OCD. Our findings indicate white matter abnormalities in brain areas connecting striatal and cortical regions of patients with OCD, specifically the anterior limbs of the internal capsule and the cingulum. The clinical relevance of these findings is supported by significant correlations of fractional anisotropy and mean diffusivity with clinical severity scores in these specific ROIs.
Acknowledgements
We are grateful to Lundbeck Pharmaceuticals and Siemens Medical Solutions South Africa for their support.
Footnotes
Contributors: C. Lochner, N. Fineberg and D. Stein designed the study. C. Lochner acquired the data, which all authors analyzed. C. Lochner wrote the article, which J.-P. Fouché, S. du Plessis, B. Spottiswoode, S. Seedat, N. Fineberg, S. Chamberlain and D. Stein reviewed. All authors approved its publication.
Competing interests: C. Lochner declares that her work has received institutional grant support from the National Research Council (competitive funding for rated researchers) and the Harry and Doris Crossley Foundation. J.P. Fouché declares having received grant support from the Departments of Psychiatry, Universities of Stellenbosch and Cape Town, and travel support from the European and South African Research Network in Anxiety Disorders. None declared for S. du Plessis. None declared for B. Spottiswoode. S. Seedat declares having received institutional grant support from the National Research Foundation and the National Institutes of Health, as well as lecture fees from Servier, AstraZeneca, Lundbeck and Dr. Reddy’s. N. Fineberg declares having consulted for Servier, Lundbeck, GlaxoSmithKline and Bristol Myers Squibb; and having received grant support from Lundbeck, Glaxo-SmithKline and Servier; lecture fees from Lundbeck, Bristol Myers Squibb and Servier; royalties from Oxford University Press; payment for educational presentations from Lundbeck and travel support from Wyeth and Cephalon. S. Chamberlain declares having consulted for Cambridge Cognition, P1Vital, Lucinda Ellery and Shire, and having received lecture fees from Lilly. D. Stein declares membership of the advisory boards of Eli Lilly, Lundbeck and Pfizer, membership of the speakers’ bureaus of GlaxoSmithKline, Lundbeck, Pfizer, Solvay and Servier, and having received consultant fees from Servier, Wyeth and Biocodex.
- Received June 29, 2011.
- Revision received October 17, 2011.
- Accepted October 21, 2011.