-
PDF
- Split View
-
Views
-
Cite
Cite
Kim Ryun Drasbek, Kimmo Jensen, THIP, a Hypnotic and Antinociceptive Drug, Enhances an Extrasynaptic GABAA Receptor-mediated Conductance in Mouse Neocortex, Cerebral Cortex, Volume 16, Issue 8, August 2006, Pages 1134–1141, https://doi.org/10.1093/cercor/bhj055
Close - Share Icon Share
Abstract
THIP (4,5,6,7-tetrahydroisoxazolo[5,4-c]pyridin-3-ol) is a selective GABAA receptor agonist with a preference for δ-subunit containing GABAA receptors. THIP is currently being tested in human trials for its hypnotic effects, displaying advantageous tolerance and addiction properties. Since its cellular actions in the neocortex are uncertain, we studied the effects of THIP on neurons in slices of frontoparietal neocortex of 13- to 19-day-old (P13–19) mice. Using whole-cell patch-clamp recordings, we found that the clinically relevant THIP concentration of 1 μM induced a robust tonic GABAA-mediated current in layer 2/3 neurons. In comparison, only a minute tonic current was induced by mimicking in vivo endogenous GABA levels. Miniature IPSCs were not affected by 1 μM THIP suggesting an extrasynaptic site of action. The EC50 for THIP was 44 μM. In accordance with the stronger expression of δ-containing receptors in superficial neocortical layers, THIP induced a 44% larger tonic current in layer 2/3 than in layer 5 neurons. Finally, monitoring spontaneously active neocortical neurons, THIP caused an overall depression of inhibitory activity, while enhancing excitatory activity prominently. Our studies suggest that THIP activates an extrasynaptic GABAA receptor-mediated conductance in the neocortex, which may alter the cortical network activity.
Introduction
THIP (4,5,6,7-tetrahydroisoxazolo[5,4-c]pyridin-3-ol) is a metabolically stable GABAA receptor agonist, which crosses the blood–brain barrier and induces analgesia, anxiolysis and hypnosis in humans, rats and dogs (Krogsgaard-Larsen et al., 2004). THIP has received renewed attention lately, since it is going through clinical trials for the treatment of sleep disorders and marketed under the name Gaboxadol. THIP appears mainly to exert its action in the midbrain and forebrain (Krogsgaard-Larsen et al., 2004) and serum concentrations of 1 μM reportedly enhances the quality of sleep (Madsen et al., 1983; Faulhaber et al., 1997). Although many of the behavioral effects have been studied, it is still unclear how THIP acts at the level of single neurons in the neocortex. Therefore, studies of brain slice preparations are warranted where the cellular actions of THIP can be analyzed in detail.
GABAA receptors are ligand-gated chloride channels, composed as pentamers of different combinations of α1–6-, β1–3-, γ1–3-, ρ1–2-, δ-, ε-, π- and θ-subunits. At native GABAA receptors in cultured cortical and cerebellar neurons, THIP is a directly acting agonist, displaying partial agonism and eliciting 50–70% of the response of GABA (Hansen et al., 2001). Similarly, at expressed α1β2γ2S receptors, THIP is a partial agonist, with an Emax of 70% and a >10-fold increase in EC50 compared with GABA, indicating considerably less potency (Ebert et al., 1997; Mortensen et al., 2004).
A recent study showed that THIP efficiently activates δ-subunit containing GABAA receptors, and is more potent at δ-containing receptors compared with γ2-containing receptors (Adkins et al., 2001; Brown et al., 2002). Furthermore, THIP displayed superagonist behaviour (compared with GABA) at δ-containing receptors with an Emax of about 160%.
GABAA receptors containing the δ-subunit are located extrasynaptically (Nusser et al., 1998) and are therefore interesting in terms of understanding extrasynaptic GABAergic ‘volume’ transmission. These GABAA receptors also display a high affinity for GABA and are weakly desensitizing (Saxena and Macdonald, 1994, 1996). Thus, these receptors react to low micromolar concentrations of ambient GABA, which leads to a tonic inhibitory conductance in the postsynaptic neurons. In the cerebellum, this conductance is thought to affect both the slope and the gain or the input–output relationship of granule cells, which has been examined by blocking the tonic GABAA conductance pharmacologically (Brickley et al., 2001) or by inducing a tonic current using dynamic clamp (Mitchell and Silver, 2003). Outside the cerebellum, a tonic GABAA conductance has also been studied in dentate gyrus granule cells (Stell and Mody, 2002; Stell et al., 2003), hippocampal interneurons (Semyanov et al., 2003) and CA1 pyramidal cells (Caraiscos et al., 2004a), all supporting the notion that tonic currents are important for the cellular excitability.
Tonic GABAA receptor-mediated conductances have not been characterized in the neocortex. Since THIP is a δ-preferring agonist, and δ-subunits are expressed in the superficial layers of the neocortex (Peng et al., 2002), it was relevant to test THIP in this area.
To study the actions of THIP in the neocortex at the cellular level, we performed whole-cell recordings from neurons in several neocortical layers in mouse brain slices. We recorded tonic GABAA receptor-mediated currents induced by THIP and determined the concentration-response relationship for THIP. Even low micromolar THIP concentrations easily superseded the tonic currents induced by elevated endogenous GABA levels. Furthermore, since expression of the δ-receptor is more prominent in the superficial neocortical layers than in the deeper layers (Peng et al., 2002), we measured the differential THIP currents in layer 2/3 versus layer 5 neurons to determine whether THIP responses were correlated to the expression of the δ-subunit. Finally, we tested how THIP affected the spontaneous inhibitory and excitatory activity in the tissue. Our studies indicate that THIP primarily activates extrasynaptic GABAA receptors to alter the cortical activity in a manner correlated to the δ-subunit expression.
Materials and Methods
Brain Slice Preparation
Male postnatal day 13–19 (P13–19) C57BL/6 mice were deeply anesthetized using isoflurane before decapitation in accordance with the guidelines of the University of Aarhus, and with Danish and European law regarding laboratory animals. The brain was removed and placed in ice-cold artificial cerebrospinal fluid (ACSF) consisting of (in mM): 126 NaCl, 2.5 KCl, 2 CaCl2, 2 MgCl2, 1.25 NaH2PO4, 26 NaHCO3, 10 D-glucose (osmolality 305–315 mosmol/kg), pH 7.4 when bubbled with 5% CO2 and 95% O2. To improve slice quality 3 mM kynurenic acid, 0.2 mM ascorbic acid and 0.2 mM pyruvic acid was present during slicing and storage. The brain was glued onto the stage of a Vibratome 3000 Plus (Vibratome Company, St Louis, MO) and 350-μm-thick coronal slices were prepared in ice-cold bubbled ACSF. The slices were stored for at least 1 h in bubbled ACSF before they were individually transferred to a recording chamber, which was continuously perfused (1.5–2 ml/min) with 33–34°C bubbled ACSF.
Brain Slice Electrophysiology
Whole-cell patch-clamp recordings were done using a MultiClamp 700B amplifier (Axon Instruments, Union City, CA) in voltage-clamp mode. A custom-built infrared video microscope (Versascope, E. Marton, CA) equipped with a ×40 water immersion objective (Olympus, Ballerup, Denmark) and a CCD100 camera (DAGE-MTI, Michigan City, IN) was employed to visualize the neurons. Patch pipettes were pulled from borosilicate glass (OD = 1.5 mm, ID = 0.8 mm; Garner Glass Company, Claremont, CA) using a DMZ Universal Puller (Zeitz Instruments, Munich, Germany) and backfilled with intracellular solution (in mM): 140 CsCl, 2 MgCl2, 10 HEPES, 0.05 EGTA, adjusted to pH 7.2 with CsOH (280–290 mosmol/kg). For recordings of inhibitory and excitatory postsynaptic currents (I/EPSCs) in the same neuron, an intracellular Cs-gluconate solution was used (in mM): 135 CsOH, 10 CsCl, 5 TEACl, 0.05 EGTA, 15 HEPES, adjusted to pH 7.2 with gluconic acid (280–290 mosmol/kg). Pipette resistances were 3–5 MΩ. Voltage-clamp recordings were carried out at Vhold = −70 mV unless otherwise stated. Whole-cell capacitances and series resistances were repeatedly monitored throughout the experiment, and series resistances were compensated by ∼70% (lag 10 μs). GABAA receptor-mediated currents were isolated by 3 mM kynurenic acid, while miniature IPSCs (mIPSCs) were recorded in kynurenic acid and TTX (1 μM).
Data Acquisition and Analysis
Currents were low-pass filtered (8-pole Bessel) at 3 kHz, and digitized at 20 kHz using a DA converter (BNC-2110) and a PCI acquisition board (PCI-6014, National Instruments, Austin, TX). A custom-written LabView 6.1 (National Instruments) based program containing an acquisition interface and analysis module was used (EVAN v. 1.4, courtesy of Istvan Mody).
GABAA receptor-mediated tonic currents were measured by injecting the selective GABAA receptor antagonist SR95531 into the slice chamber in a final concentration of 100–150 μM. The resulting outward shift in the holding current was taken as the tonic GABAA-mediated current. For quantification, 5 ms long samples of the holding current was taken every 100 ms and plotted against time. Those samples that contained contaminating IPSCs were discarded (Nusser and Mody, 2002). From these plots, the mean current was calculated in 3 s long regions at three time points (denoted a, b and c): just before SR95531 application (b); and at equal time points (normally 20 s) before (a) and after (c) SR95531. The tonic GABAA-mediated tonic current was calculated as b – c, while the spontaneous baseline fluctuations before the SR95531 injection (‘No Treatment’) was taken as b – a.
Spontaneous IPSCs (sIPSCs), sEPSCs and mIPSCs were detected and analyzed using EVAN v. 1.4. Detection thresholds of 7–9 pA were used for mIPSCs. When sEPSCs and sIPSCs were recorded simultaneously, detection thresholds were raised to 10–15 and 15–20 pA, respectively. The latter approach would remove the bulk of miniature events, which are unaffected by THIP (Fig. 4). The analysis would thereby be favored towards action potential-driven events.
All events were inspected and accepted visually, before the rise-time, frequency and amplitude were measured. Weighted decay time constants were measured from double exponential fitting of IPSC decays. Dose-response curves were generated using GraphPad Prism v. 4.0 (San Diego, CA). SPSS v. 11.0 (Chicago, IL) was used to compare means (paired and unpaired t-test) with a significance level of P < 0.05. Data are expressed as means ± SEM, with n indicating the number of cells.
Solutions and Drugs
Kynurenic acid, ascorbic acid, SR95531, GABA and NO-711 were purchased from Sigma (St Louis, MO). Pyruvic acid was obtained from MP Biomedicals (Irvine, CA), while tetrodotoxin (TTX) was from Alomone Labs (Jerusalem, Israel). THIP (Gaboxadol) was a gift from Bjarke Ebert (Lundbeck, Copenhagen, Denmark). The SR95531 stock solutions (6–8 mM) were prepared in ACSF containing 50% DMSO.
Results
THIP Induces a Tonic GABAA Receptor Current in the Mouse Neocortex
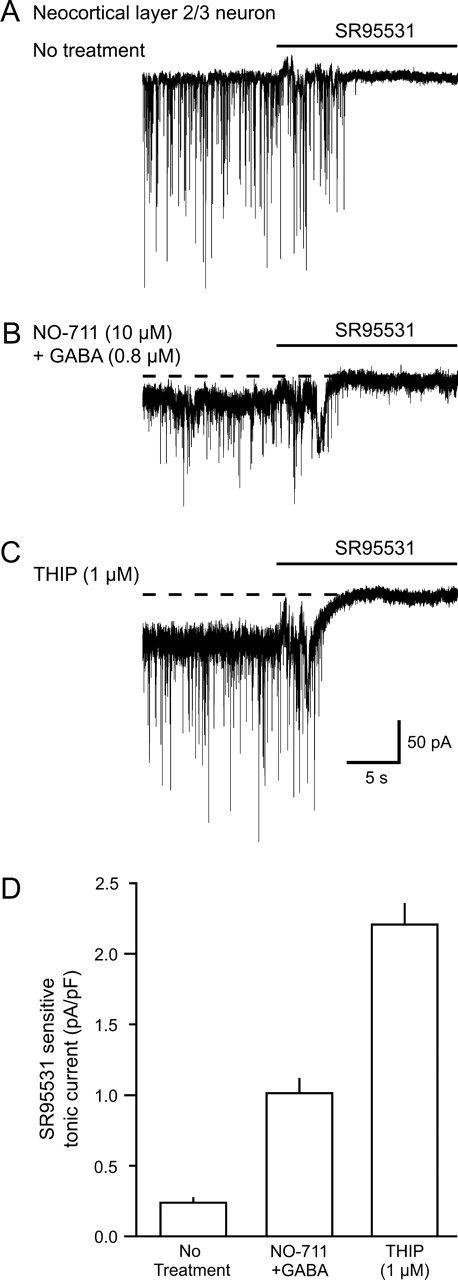
Since important parts of the hypnotic effects of THIP are likely mediated in the forebrain (Krogsgaard-Larsen et al., 2004), we wanted to analyze its actions in the cerebral cortex. For this purpose, we chose layer 2/3 neurons, since these are involved in cortical information processing and also these neurons can be visualized for electrophysiological recordings. To measure GABAA receptor-mediated tonic currents induced by THIP, whole-cell patch clamp recordings were performed from somata of neocortical layer 2/3 neurons in mouse brain slices (Jensen and Mody, 2001). Neurons were selected if they displayed a pyramidal shaped cell body and a prominent apical dendrite projecting towards layer 1. Using CsCl intracellular solutions (ECl ∼ 0 mV), GABAA-mediated sIPSCs appeared as fast inward currents (Vhold = −70 mV) in the presence of kynurenic acid (3 mM), which blocks ionotropic glutamate receptors. Mean sIPSCs amplitudes were 53.1 ± 5.3 pA, frequencies were 13.2 ± 1.5 Hz, while 10–90% rise times were 1074 ± 74 μs and weighted decay constants were 8.9 ± 0.7 ms (n = 8). Injecting the GABAA antagonist SR95531 (>100 μM) into the chamber blocked sIPSCs and in untreated slices revealed no tonic current (Fig. 1A). In contrast to the lack of tonic GABAA currents in controls, when mimicking the in vivo ambient extracellular GABA levels by perfusing 0.8 μM GABA plus the GABA uptake inhibitor NO-711 (10 μM) (Jensen et al., 2003) a tonic GABAA current of 26.6 ± 3.6 pA (n = 5) was revealed (Fig. 1B). On the other hand, 1 μM THIP induced a tonic current of 43.2 ± 4.7 pA (n = 9) (Fig. 1C). In Figure 1D the mean tonic currents are normalized to the cell capacitance (pA/pF) and pooled. The capacitance showed little variability between individual layer 2/3 neurons, and was on average 29.5 ± 1.1 pF (n = 31).
Tonic currents in neocortical layer 2/3 neurons induced by GABA or THIP. (A) Whole cell patch clamp recordings of GABAA receptor-mediated spontaneous IPSCs. sIPSCs were blocked by direct injection of the GABAA receptor antagonist SR95531 (>100 μM) into the slice chamber. (B) Elevated extracellular GABA (10 μM NO-711 + 0.8 μM GABA) and (C) THIP (1 μM) induced a SR95531-sensitive tonic current. (D) Histogram showing the GABAA receptor-mediated tonic current normalized to the capacitance for neurons without treatment (n = 31), in 0.8 μM NO-711 + 10 μM GABA (n = 5) and in 1 μM THIP (n = 9, error bars show SEM).
The Concentration Dependency of THIP-induced tonic GABAA currents
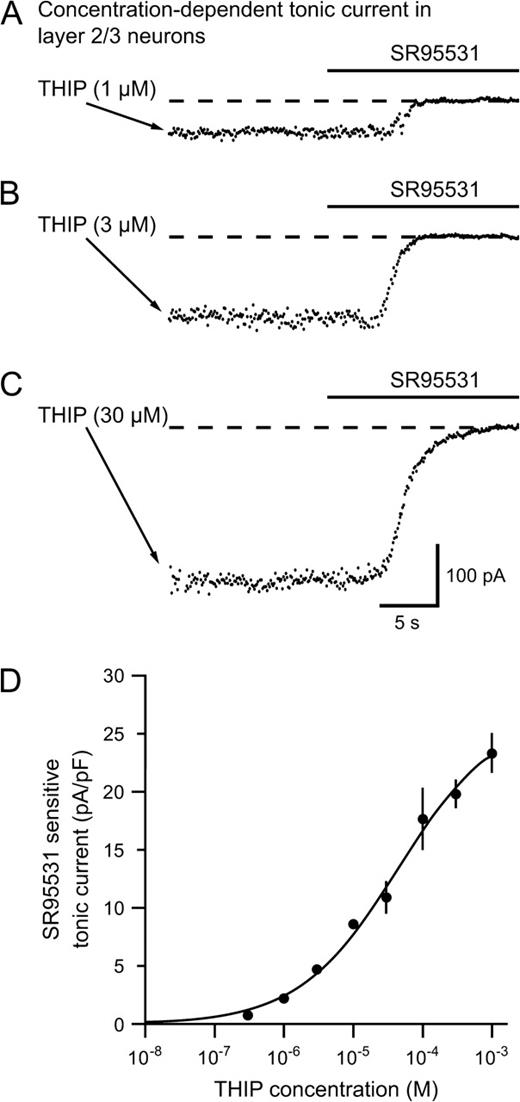
Sedation occurs at plasma concentrations of THIP in the low micromolar range, so we wanted to examine the concentration dependency of THIP on GABAA receptor currents in layer 2/3 neurons. Figure 2A–C shows plots of the holding current before and after an SR95531 application (>100 μM). THIP concentrations >0.3 μM induced robust tonic currents, while >1 mM THIP would be needed to reach the maximum response. From the graph in Figure 2D, an EC50 of 44 μM was determined (n = 4– 9 per THIP concentration).
THIP induces a concentration dependent tonic current in neocortical layer 2/3 neurons. (A–C) Plots of the holding current in neurons showing SR95531 sensitive tonic currents following increasing concentrations of THIP (1, 3 and 30 μM respectively). (D) Dose–response curve showing the tonic current mediated by increasing bath concentrations of THIP. The EC50 value was 44 μM. Several neurons were tested at each THIP concentration (n = 4–9 neurons per point). At the lower concentrations, the response to THIP was very consistent between neurons.
The Tonic THIP Current Correlates with δ-Subunit Expression in Mouse Neocortex
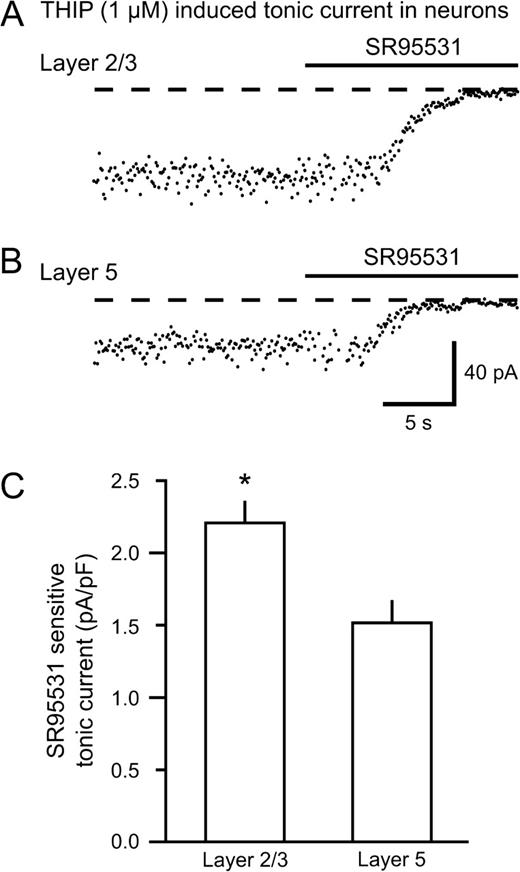
As shown by Peng and coworkers, the expression of the δ-subunit of GABAA receptors displays a region-specific pattern in the mouse brain (Peng et al., 2002). For the neocortex, there is a clear difference in the δ-expression between the superficial and deep layers, being strongest in layers 1–3. Since THIP is a putative δ-subunit preferring agonist, we tested the effects of THIP in layer 2/3 versus layer 5 neurons. Holding currents recorded from a layer 2/3 and layer 5 neuron during SR95531 injection are shown in Figure 3A and 3B, respectively. The THIP-induced current was 44% larger in layer 2/3 (43.2 ± 4.7 pA, n = 9) compared with layer 5 (30.1 ± 3.5 pA, n = 10, P < 0.05) neurons (Fig. 3C), consistent with the hypothesis that the actions of THIP are related to the δ-subunit expresison.
THIP (1 μM) mediates larger tonic currents in neurons from layer 2/3 than from layer 5 in the neocortex. Recordings from neurons in layer 2/3 (A) and layer 5 (B) treated with 1 μM THIP. (C) Average tonic currents in neurons from either layer 2/3 (n = 9) or layer 5 (n = 10) normalized to the cell capacitance. The tonic current induced by THIP was ∼45% larger in layer 2/3 neurons than in layer 5 neurons (*P < 0.05).
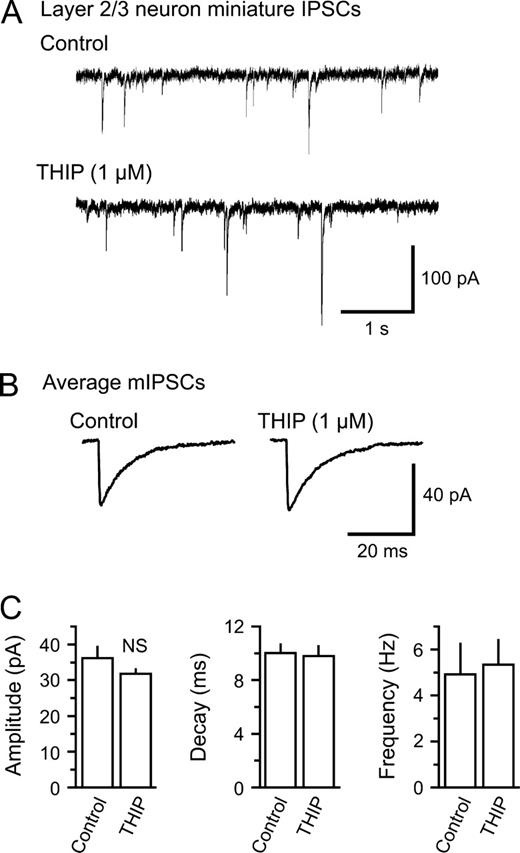
THIP Does not Affect Miniature IPSCs in Layer 2/3 Neurons
To test whether THIP binds to intrasynaptic GABAA receptors and thereby affects the postsynaptic response to single GABA quanta, we recorded miniature IPSCs (mIPSCs) in layer 2/3 neurons in the presence of TTX (1 μM). Current traces before and after bath application of THIP are presented in Figure 4A. The analysis of averages of mIPSCs with and without THIP (Fig. 4B) showed that THIP at 1 μM did not significantly alter the mIPSC frequency (4.9 ± 1.3 versus 5.3 ± 1.0 Hz, control and THIP), amplitude (36.2 ± 3.2 versus 31.8 ± 1.4 pA), 10–90% rise time (1.1 ± 0.3 versus 1.4 ± 0.1 ms) or weighted decay constant (10.0 ± 0.7 versus 9.8 ± 0.8 ms) (P > 0.05, n = 6, for all parameters) (Fig. 4C), indicating that THIP has little or no affinity for intrasynaptic GABAA receptor at this concentration.
THIP does not affect miniature IPSCs in layer 2/3 neurons. (A) Miniature IPSCs (mIPSCs) recorded in the presence of 1 μM TTX in a layer 2/3 neuron. Following the perfusion of 1 μM THIP, there were only minor changes in the amplitude and frequency of mIPSCs. (B) Average of 100 mIPSCs before and after perfusion of THIP in the same neuron. There were no major changes in the mIPSC waveform. (C) Pooled results of mIPSC properties from layer 2/3 neurons (n = 6). Neither the peak, the amplitude, the weighted decay constant nor the event frequency was significantly altered by THIP (NS: non-significant).
Effects of THIP on Spontaneous Neuronal Activity in Mouse Neocortex
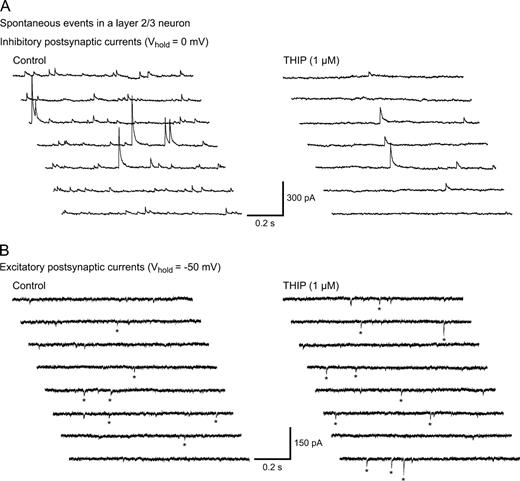
Our results indicated that GABAA agonism by THIP caused a tonic current mediated by extrasynaptic GABAA receptors, with minimal influence on intrasynaptic receptors. What would then be the effect of THIP on spontaneously active neocortical neurons? Given the findings above, we wanted to assess how THIP affected spontaneous inhibitory and excitatory activity in the neocortex. We chose to test 1 μM THIP, since this concentration induces a robust tonic GABAA current in at least two neocortical cell types and is a clinically relevant concentration for sedation and analgesia (Madsen et al., 1983). We recorded from layer 2/3 neurons in slices perfused with plain ACSF. Using Cs-gluconate rich pipettes, sIPSCs and sEPSCs could be isolated by alternating Vhold between EIPSC (∼ −50 mV) and EEPSC (∼ 0 mV) in 30–60 s epochs. Traces are shown in Figure 5.
THIP induces opposite changes in sIPSC and sEPSC frequency in layer 2/3 neurons. Multiple sweeps (1 s duration) showing recordings of sIPSCs (A) and sEPSCs (B) in the same neuron. Recordings were performed in a plain Ringer solution using a Cs-gluconate patch solution at the indicated Vhold. The sIPSC frequency decreased while sEPSCs increased upon THIP treatment. For EPSCs, the stars indicate which events the analysis software detected.
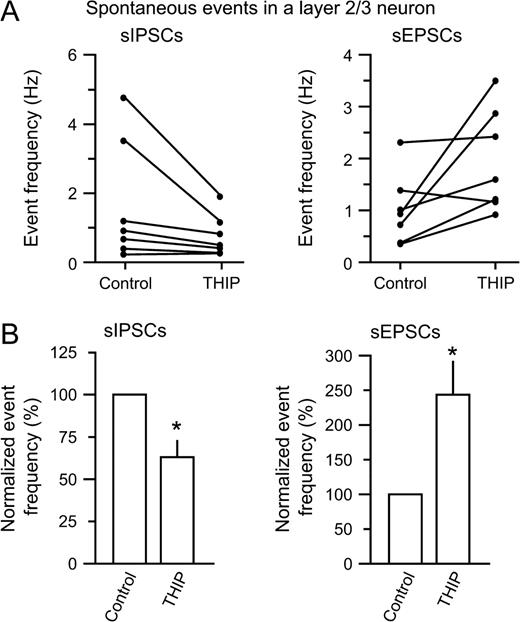
Before THIP was added, detected sIPSCs occurred at 1.7 ± 0.7 Hz, while sEPSCs occurred at 1.0 ± 0.3 Hz. The addition of THIP caused a decrease in sIPSCs by 37 ± 10% (n = 6, P < 0.05), while the sEPSCs increased by 144 ± 49% (n = 6, P < 0.05) (Fig. 6A,B). THIP did not significantly alter the amplitude of sIPSCs (96 ± 20.8 versus 70 ± 9.5 pA for control and THIP) (P > 0.05) or sEPSCs (27 ± 3.6 versus 30 ± 4.6 pA for control and THIP) (P > 0.05). Since our analysis favored large action-potential driven IPSCs (see Materials and Methods), the decrease in inhibitory event frequency indicates that interneuron firing was depressed by THIP. A local shunting of IPSCs in the postsynaptic cell by THIP is unlikely, since neither the amplitude nor the frequency of miniature IPSCs were affected by 1 μM THIP (Fig. 4). Concomitantly, the increase in sEPSCs indicates an increased pyramidal cell firing. The results show that although THIP is a GABAmimetic drug, a surprising effect of reduced inhibition and increased excitation is observed in the neocortical tissue.
Summary of THIP-induced changes in sIPSCs and sEPSCs. (A) Plots from individual neurons showing changes in sIPSC (left) and sEPSC frequency (right) (n = 7). (B) The normalized event frequency showed a significant reduction in sIPSC frequency (left, by 37%, *P < 0.05), while the sEPSC frequency increased strongly (right, by 144%, *P < 0.05) following THIP application. This could indicate that interneuron firing decreased, with a concomitant increase in pyramidal cell firing.
Discussion
We have studied the actions of the δ-subunit preferring hypnotic drug THIP on neuronal GABAA receptors in the mouse neocortex. We found that THIP, at clinically relevant concentrations, induced a significant GABAA-mediated tonic current in neocortical neurons, without affecting intrasynaptic GABAA receptors. The magnitude of the tonic current correlated qualitatively with the δ-subunit expression in neocortical layers. Surprisingly, THIP caused a pronounced decrease in the frequency of activity driven sIPSCs in layer 2/3 neurons, while the frequency of sEPSCs increased. These results indicate that THIP primarily acts at extrasynaptic GABAA receptors in the neocortex, and may particularly depress interneuron activity to diminish inhibitory synaptic input. This may explain why THIP alters the cortical oscillatory activity (Vyazovskiy et al., 2005), since the spike timing, in part, depends on the networks of extensively connected interneurons (Tamas et al., 2000; Buhl et al., 1998).
GABAA Receptor Subunits in the Neocortex
Neocortical neurons express a variety of GABAA receptor subunits. The combinations of α1, β1–3- and γ2-subunits are prototypic intrasynaptic receptors, since the essential γ2-subunits link up with various clustering proteins, such as gephyrin and GABA-RAP in the postsynaptic membrane opposing the inhibitory nerve terminals (Essrich et al., 1998). In the neocortex, α1, β2/3 and γ2 are among the most abundant subunits, consistent with the high concentration of GABA synapses in this region.
On the contrary, at peri- or extrasynaptic sites, α4- and δ-subunits are thought to link up in brain regions such as neocortex, dentate gyrus and thalamus (Sur et al., 1999). The partnership between δ and α4 was recently substantiated, because the deletion of δ-subunits also led to a decrease in α4 expression in brain regions where δ is normally found (Peng et al., 2002). Analogously, in the cerebellum the δ-subunit is carried by α6 to extrasynaptic sites (Jones et al., 1997). Furthermore, γ- and δ-subunits appear to be complementary subunits (Araujo et al., 1998), consistent with the δ-subunit being excluded from intrasynaptic sites. The δ-subunit confers a high affinity for GABA and minimal desensitization, which make them ideal for reacting to low ambient levels of extracellular GABA (Brown et al., 2002). Their presence in the extrasynaptic membrane is thought to have significant consequences for neuronal properties (Farrant and Nusser, 2005).
The Pharmacological Profile of THIP at GABAA Receptors
Studies in expression systems have shown that THIP is a partial agonist at α1β3γ2 and α4β3γ2 receptors (Ebert et al., 1997). In contrast, at receptors containing α4- and β3-subunits in combination with δ-subunits, THIP displays a ∼10-fold higher potency and a superagonist behaviour with a maximal response of 160% compared with GABA (Brown et al., 2002). This means that THIP, at clinically relevant concentrations in the low micromolar range, will preferentially activate δ- over γ-containing receptors. We found in brain slices that 1 μM THIP induced a robust tonic GABAA-mediated conductance in neocortical neurons, with little or no effect on miniature IPSCs. These findings are in accordance with the hypothesis that extrasynaptic receptors contain δ-subunits, while intrasynaptic receptors contain γ-subunits. In support of our findings, THIP induced the most prominent response in superficial cortical layers, where the expression of the δ-subunit is strongest (Peng et al., 2002).
The Functional Role of Tonic/Extrasynaptic GABAA Receptor Conductances
Most knowledge about tonic GABAA conductances stems from studies in the hippocampus and cerebellum, while data from the neocortex are sparse. This is why we performed studies using THIP as a putative extrasynaptic agonist. Indeed, THIP induces a strong tonic extrasynaptic conductance in layer 2/3 neurons, but the role of this conductance in vivo remains to be fully elucidated. Previous studies have found that a tonic inhibitory conductance controls the gain and sets the input–output characteristics of cerebellar neurons (Mitchell and Silver, 2003). Another study found that hippocampal interneurons were strongly controlled by a tonic conductance, since its removal resulted in robust interneuron firing (Semyanov et al., 2003). This is in accordance with our results in the neocortex that THIP depressed interneuron firing and thereby the sIPSC frequency. At the protein level, a clear expression of δ-subunits is found in interneurons in CA1/CA3 regions, while principal cells display a very weak δ-subunit expression under normal conditions (Peng et al., 2002).
We found recently at the behavioural level that enhancing a tonic GABAA conductance via the knock-down of GABA reuptake (Jensen et al., 2003) leads to altered body temperature, ataxia, tremor and increased nervousness (Chiu et al., 2005). The alterations in motor function may in part be due to altered activity in cortical and cerebellar circuits, and in fact both the cerebellar Purkinje and granule cell function was changed. This strengthens the hypothesis that tonic GABAA conductances indeed play an important role in neuronal processing. This implies that THIP affects an important regulatory system in the brain.
Clinical Relevance of the THIP-induced Tonic GABAA-mediated Conductance
The extrasynaptic δ-subunits appear to be quite plastic, and their expression is regulated in several neuropathological states. In a mouse model of temporal lobe epilepsy, δ-subunits were down-regulated in principal cells, while they increased in interneurons (Peng et al., 2004). This indicates that these cell types react differently to such insults. Recently, in a model of catamenial epilepsy, δ-subunit activity was found to cycle with the sex hormones, being lowest when seizure susceptibility and anxiety were highest. Also, the response to THIP in dentate gyrus granule cells was abolished in δ-subunit knockout mice (Maguire et al., 2005), again supporting the notion that THIP is δ-subunit preferring (Shen et al., 2005). Interestingly, THIP has very limited antiepileptic effects (Petersen et al., 1983; Holland et al., 1992; Hansen et al., 2004). However, if δ-subunits are usually down-regulated in principal cells in epileptic conditions, this may not be surprising after all.
Compounds which potentiate the function of δ-subunit containing GABAA receptors, such as propofol, act as potent anaesthetics (Feng and Macdonald, 2004). Similarly, it was found that extrasynaptic GABAA receptors are the major target in the cerebral cortex for volatile anaesthetics, although this primarily seemed to involve α5-containing receptors on hippocampal neurons (Caraiscos et al., 2004a,b).
The Effects of THIP on Sleep
In humans, THIP is an advantageous sleep aid and increases sleep consolidation and intensity without depressing REM sleep (Faulhaber et al., 1997). Subjects report an improved intensity and quality of sleep, as well as next-day performance (Mathias et al., 2005). Additionally, since THIP displays little tolerance and addiction potential, it may have many advantages over traditional benzodiazepine substances and other positive GABAA modulators. In vivo experiments in rats showed that non-REM slow wave activity was increased by THIP (Lancel and Faulhaber, 1996). On the other hand, in mice THIP increases certain types of asymmetric low frequency EEG activity, although the significance of this still has to be determined (Vyazovskiy et al., 2005). The difference between rats and mice could of course have a variety of explanations, including a species specific wiring, connectivity and physiology of the cerebral cortex. However, it is known that tonic hippocampal GABAA currents are remarkably larger in rats (Bai et al., 2001; Nusser and Mody, 2002) than in mice (Stell and Mody, 2002), which indicates a higher density of extrasynaptic GABAA receptors containing δ-subunits in rats.
While we found prominent effects in the neocortex of clinically relevant concentrations of THIP, it is also possible that THIP promotes changes in sleep via actions in thalamic nuclei (Steriade, 2005), since these have a particular strong δ-subunit expression (Peng et al., 2002). Other putative targets include the locus coeruleus, the raphe nucleus and areas of the hypothalamus such as the tuberomammaliary nucleus (Nelson et al., 2002), although the δ-subunit expression is variable in these areas. In the future, it will be relevant to systematically study the cellular effects of THIP in subcortical brain regions, and finally to resolve its influence on the dynamic cortical–subcortical interactions.
We thank Bjarke Ebert (Lundbeck, Copenhagen) for the gift of THIP. We appreciate the technical support of Lene Steffensen, Dianna Haun Nielsen and Brita Holst Jensen. This research was supported by The Danish Medical Research Council, Lundbeck Foundation, Carlsberg Foundation, Novo Nordisk Foundation, and University of Aarhus Research Foundation.
References
Adkins CE, Pillai GV, Kerby J, Bonnert TP, Haldon C, McKernan RM, Gonzalez JE, Oades K, Whiting PJ, Simpson PB (
Araujo F, Ruano D, Vitorica J (
Bai D, Zhu G, Pennefather P, Jackson MF, MacDonald JF, Orser BA (
Brickley SG, Revilla V, Cull-Candy SG, Wisden W, Farrant M (
Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA (
Buhl EH, Tamas G, Fisahn A (
Caraiscos VB, Elliott EM, You T, Cheng VY, Belelli D, Newell JG, Jackson MF, Lambert JJ, Rosahl TW, Wafford KA, MacDonald JF, Orser BA (
Caraiscos VB, Newell JG, You T, Elliott EM, Rosahl TW, Wafford KA, MacDonald JF, Orser BA (
Chiu CS, Brickley S, Jensen K, Southwell A, Mckinney S, Cull-Candy S, Mody I, Lester HA (
Ebert B, Thompson SA, Saounatsou K, McKernan R, Krogsgaard-Larsen P, Wafford KA (
Essrich C, Lorez M, Benson JA, Fritschy JM, Luscher B (
Farrant M, Nusser Z (
Faulhaber J, Steiger A, Lancel M (
Feng HJ, Macdonald RL (
Hansen SL, Ebert B, Fjalland B, Kristiansen U (
Hansen SL, Sperling BB, Sanchez C (
Holland KD, McKeon AC, Canney DJ, Covey DF, Ferrendelli JA (
Jensen K, Mody I (
Jensen K, Chiu CS, Sokolova I, Lester HA, Mody I (
Jones A, Korpi ER, McKernan RM, Pelz R, Nusser Z, Makela R, Mellor JR, Pollard S, Bahn S, Stephenson FA, Randall AD, Sieghart W, Somogyi P, Smith AJ, Wisden W (
Krogsgaard-Larsen P, Frolund B, Liljefors T, Ebert B (
Lancel M, Faulhaber J (
Madsen SM, Lindeburg T, Folsgard S, Jacobsen E, Sillesen H (
Maguire JL, Stell BM, Rafizadeh M, Mody I (
Mathias S, Zihl J, Steiger A, Lancel M (
Mitchell SJ, Silver RA (
Mortensen M, Kristiansen U, Ebert B, Frolund B, Krogsgaard-Larsen P, Smart TG (
Nelson LE, Guo TZ, Lu J, Saper CB, Franks NP, Maze M (
Nusser Z, Mody I (
Nusser Z, Sieghart W, Somogyi P (
Peng Z, Hauer B, Mihalek RM, Homanics GE, Sieghart W, Olsen RW, Houser CR (
Peng Z, Huang CS, Stell BM, Mody I, Houser CR (
Petersen HR, Jensen I, Dam M (
Saxena NC, Macdonald RL (
Saxena NC, Macdonald RL (
Semyanov A, Walker MC, Kullmann DM (
Shen H, Gong QH, Yuan M, Smith SS (
Stell BM, Brickley SG, Tang CY, Farrant M, Mody I (
Stell BM, Mody I (
Steriade M (
Sur C, Farrar SJ, Kerby J, Whiting PJ, Atack JR, McKernan RM (
Tamas G, Buhl EH, Lorincz A, Somogyi P (