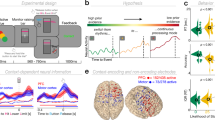
Abstract
Cognitive control allows to flexibly guide behaviour in a complex and ever-changing environment. It is supported by theta band (4–7 Hz) neural oscillations that coordinate distant neural populations. However, little is known about the precise neural mechanisms permitting such flexible control. Most research has focused on theta amplitude, showing that it increases when control is needed, but a second essential aspect of theta oscillations, their peak frequency, has mostly been overlooked. Here, using computational modelling and behavioural and electrophysiological recordings, in three independent datasets, we show that theta oscillations adaptively shift towards optimal frequency depending on task demands. We provide evidence that theta frequency balances reliable set-up of task representation and gating of task-relevant sensory and motor information and that this frequency shift predicts behavioural performance. Our study presents a mechanism supporting flexible control and calls for a reevaluation of the mechanistic role of theta oscillations in adaptive behaviour.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Raw behavioural, eye-tracking and EEG data can be found on the Open Science Framework repository at https://osf.io/nwh87/?view_only=b11ee1f860804da582c816fe8acdecad.
Code availability
Code of the model, the behavioural experiment and analysis scripts to reproduce all results and figures from the study can be found on Github at https://github.com/mehdisenoussi/theta_shift_cog_control.
References
Cavanagh, J. F. & Frank, M. J. Frontal theta as a mechanism for cognitive control. Trends Cogn. Sci. 18, 414–421 (2014).
Sauseng, P., Tschentscher, N. & Biel, A. L. Be prepared: tune to FM-theta for cognitive control. Trends Neurosci. 42, 307–309 (2019).
Voloh, B. & Womelsdorf, T. A role of phase-resetting in coordinating large scale neural networks during attention and goal-directed behavior. Front. Syst. Neurosci. 10, 18 (2016).
Canolty, R. T. et al. High gamma power is phase-locked to theta oscillations in human neocortex. Science 313, 1626–1628 (2006).
Varela, F., Lachaux, J.-P., Rodriguez, E. & Martinerie, J. The brainweb: phase synchronization and large-scale integration. Nat. Rev. Neurosci. 2, 229–239 (2001).
Bressler, S. L., Coppola, R. & Nakamura, R. Episodic multiregional cortical coherence at multiple frequencies during visual task performance. Nature 366, 153–156 (1993).
Palva, J. M., Palva, S. & Kaila, K. Phase synchrony among neuronal oscillations in the human cortex. J. Neurosci. 25, 3962–3972 (2005).
Wallis, J. D. & Miller, E. K. From rule to response: neuronal processes in the premotor and prefrontal cortex. J. Neurophysiol. 90, 1790–1806 (2003).
Stokes, M. G. et al. Dynamic coding for cognitive control in prefrontal cortex. Neuron 78, 364–375 (2013).
Mansouri, F. A., Freedman, D. J. & Buckley, M. J. Emergence of abstract rules in the primate brain. Nat. Rev. Neurosci. 21, 595–610 (2020).
Voloh, B., Valiante, T. A., Everling, S. & Womelsdorf, T. Theta–gamma coordination between anterior cingulate and prefrontal cortex indexes correct attention shifts. Proc. Natl Acad. Sci. USA 112, 8457–8462 (2015).
Fries, P. Rhythms for cognition: communication through coherence. Neuron 88, 220–235 (2015).
Klimesch, W., Schack, B. & Sauseng, P. The functional significance of theta and upper alpha oscillations. Exp. Psychol. 52, 99–108 (2005).
Cooper, P. S. et al. Frontal theta predicts specific cognitive control-induced behavioural changes beyond general reaction time slowing. NeuroImage 189, 130–140 (2019).
Nigbur, R., Cohen, M. X., Ridderinkhof, K. R. & Stürmer, B. Theta dynamics reveal domain-specific control over stimulus and response conflict. J. Cogn. Neurosci. 24, 1264–1274 (2011).
Donoghue, T. et al. Parameterizing neural power spectra into periodic and aperiodic components. Nat. Neurosci. 23, 1655–1665 (2020).
Desimone, R. & Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 18, 193–222 (1995).
Usher, M. & McClelland, J. L. The time course of perceptual choice: the leaky, competing accumulator model. Psychol. Rev. 108, 550–592 (2001).
Verguts, T. Binding by random bursts: a computational model of cognitive control. J. Cogn. Neurosci. 29, 1103–1118 (2017).
Senoussi, M., Moreland, J. C., Busch, N. A. & Dugué, L. Attention explores space periodically at the theta frequency. J. Vis. 19, 22–22 (2019).
Kienitz, R., Schmid, M. C. & Dugué, L. Rhythmic sampling revisited: experimental paradigms and neural mechanisms. Eur. J. Neurosci. Advance online publication https://doi.org/10.1111/ejn.15489 (2021).
De Loof, E. et al. Preparing for hard times: scalp and intracranial physiological signatures of proactive cognitive control. Psychophysiology 56, e13417 (2019).
Cavanagh, J. F., Cohen, M. X. & Allen, J. J. B. Prelude to and resolution of an error: EEG phase synchrony reveals cognitive control dynamics during action monitoring. J. Neurosci. 29, 98–105 (2009).
Luu, P., Tucker, D. M. & Makeig, S. Frontal midline theta and the error-related negativity: neurophysiological mechanisms of action regulation. Clin. Neurophysiol. 115, 1821–1835 (2004).
Senoussi, M. et al. Pre-stimulus antero-posterior EEG connectivity predicts performance in a UAV monitoring task. In Proceedings of 2016 International Conference on Systems, Man, and Cybernetics (Canada): IEEE SMC, 1167–1172 (2017).
Kaiser, J. & Schütz-Bosbach, S. Proactive control without midfrontal control signals? The role of midfrontal oscillations in preparatory conflict adjustments. Biol. Psychol. 148, 107747 (2019).
Nelli, S., Itthipuripat, S., Srinivasan, R. & Serences, J. T. Fluctuations in instantaneous frequency predict alpha amplitude during visual perception. Nat. Commun. 8, 1–12 (2017).
Lopes da Silva, F. H., Vos, J. E., Mooibroek, J. & van Rotterdam, A. Relative contributions of intracortical and thalamo-cortical processes in the generation of alpha rhythms, revealed by partial coherence analysis. Electroencephalogr. Clin. Neurophysiol. 50, 449–456 (1980).
Wutz, A., Melcher, D. & Samaha, J. Frequency modulation of neural oscillations according to visual task demands. Proc. Natl Acad. Sci. USA 115, 1346–1351 (2018).
Samaha, J., Bauer, P., Cimaroli, S. & Postle, B. R. Top-down control of the phase of alpha-band oscillations as a mechanism for temporal prediction. Proc. Natl Acad. Sci. USA 112, 8439–8444 (2015).
Lisman, J. E. & Jensen, O. The theta–gamma neural code. Neuron 77, 1002–1016 (2013).
Siebenhühner, F., Wang, S. H., Palva, J. M. & Palva, S. Cross-frequency synchronization connects networks of fast and slow oscillations during visual working memory maintenance. eLife 5, e13451 (2016).
Senoussi, M., Verbeke, P. & Verguts, T. Time-based binding as a solution to and a limitation for flexible cognition. Front. Psychol. 12, 798061 (2022).
Axmacher, N. et al. Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proc. Natl Acad. Sci. USA 107, 3228–3233 (2010).
Kosciessa, J. Q., Grandy, T. H., Garrett, D. D. & Werkle-Bergner, M. Single-trial characterization of neural rhythms: potential and challenges. NeuroImage 206, 116331 (2020).
Wolinski, N., Cooper, N. R., Sauseng, P. & Romei, V. The speed of parietal theta frequency drives visuospatial working memory capacity. PLoS Biol. 16, e2005348 (2018).
Riddle, J., Scimeca, J. M., Cellier, D., Dhanani, S. & D’Esposito, M. Causal evidence for a role of theta and alpha oscillations in the control of working memory. Curr. Biol. 30, 1748–1754 (2020).
Itthipuripat, S., Wessel, J. R. & Aron, A. R. Frontal theta is a signature of successful working memory manipulation. Exp. Brain Res. 224, 255–262 (2013).
Mitchell, D. J., McNaughton, N., Flanagan, D. & Kirk, I. J. Frontal-midline theta from the perspective of hippocampal “theta”. Prog. Neurobiol. 86, 156–185 (2008).
Siapas, A. G., Lubenov, E. V. & Wilson, M. A. Prefrontal phase locking to hippocampal theta oscillations. Neuron 46, 141–151 (2005).
Shenhav, A., Botvinick, M. M. & Cohen, J. D. The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron 79, 217–240 (2013).
Shenhav, A. et al. Toward a rational and mechanistic account of mental effort. Annu. Rev. Neurosci. 40, 99–124 (2017).
Holroyd, C. B. & Yeung, N. Motivation of extended behaviors by anterior cingulate cortex. Trends Cogn. Sci. 16, 122–128 (2012).
Pastötter, B., Dreisbach, G. & Bäuml, K.-H. T. Dynamic adjustments of cognitive control: oscillatory correlates of the conflict adaptation effect. J. Cogn. Neurosci. 25, 2167–2178 (2013).
Verbeke, P. & Verguts, T. Neural synchrony for adaptive control. J. Cogn. Neurosci. 33, 2394–2412 (2021).
Holroyd, C. B. & McClure, S. M. Hierarchical control over effortful behavior by rodent medial frontal cortex: a computational model. Psychol. Rev. 122, 54–83 (2015).
Holroyd, C. B. & Verguts, T. The best laid plans: computational principles of anterior cingulate cortex. Trends Cogn. Sci. 25, 316–329 (2021).
Holroyd, C. B., Ribas-Fernandes, J. J. F., Shahnazian, D., Silvetti, M. & Verguts, T. Human midcingulate cortex encodes distributed representations of task progress. Proc. Natl Acad. Sci. USA 115, 6398–6403 (2018).
Womelsdorf, T., Johnston, K., Vinck, M. & Everling, S. Theta-activity in anterior cingulate cortex predicts task rules and their adjustments following errors. Proc. Natl Acad. Sci. USA 107, 5248–5253 (2010).
Haynes, J.-D. et al. Reading hidden intentions in the human brain. Curr. Biol. 17, 323–328 (2007).
Smith, E. H. et al. Widespread temporal coding of cognitive control in the human prefrontal cortex. Nat. Neurosci. 22, 1883–1891 (2019).
Helfrich, R. F. et al. Neural mechanisms of sustained attention are rhythmic. Neuron 99, 854–865 (2018).
Dugué, L., Roberts, M. & Carrasco, M. Attention reorients periodically. Curr. Biol. 26, 1595–1601 (2016).
Dugué, L., McLelland, D., Lajous, M. & VanRullen, R. Attention searches nonuniformly in space and in time. Proc. Natl Acad. Sci. USA 112, 15214–15219 (2015).
Fiebelkorn, I. C., Saalmann, Y. B. & Kastner, S. Rhythmic sampling within and between objects despite sustained attention at a cued location. Curr. Biol. 23, 2553–2558 (2013).
Landau, A. N. & Fries, P. Attention samples stimuli rhythmically. Curr. Biol. 22, 1000–1004 (2012).
Dugué, L., Xue, A. M. & Carrasco, M. Distinct perceptual rhythms for feature and conjunction searches. J. Vis. 17(3), 22 (2017).
Holcombe, A. O. & Chen, W.-Y. Splitting attention reduces temporal resolution from 7 Hz for tracking one object to <3 Hz when tracking three. J. Vis. 13(1), 12 (2013).
VanRullen, R. Perceptual cycles. Trends Cogn. Sci. 20, 723–735 (2016).
Fiebelkorn, I. C. & Kastner, S. A rhythmic theory of attention. Trends Cogn. Sci. https://doi.org/10.1016/j.tics.2018.11.009 (2018).
Kienitz, R. et al. Theta rhythmic neuronal activity and reaction times arising from cortical receptive field interactions during distributed attention. Curr. Biol. 28, 2377–2387 (2018).
Fiebelkorn, I. C. & Kastner, S. A rhythmic theory of attention. Trends Cogn. Sci. 23, 87–101 (2019).
Helfrich, R. F. & Knight, R. T. Oscillatory dynamics of prefrontal cognitive control. Trends Cogn. Sci. 20, 916–930 (2016).
de Vries, I. E. J., Slagter, H. A. & Olivers, C. N. L. Oscillatory control over representational states in working memory. Trends Cogn. Sci. 24, 150–162 (2020).
Riddle, J., Vogelsang, D. A., Hwang, K., Cellier, D. & D’Esposito, M. Distinct oscillatory dynamics underlie different components of hierarchical cognitive control. J. Neurosci. 40, 4945–4953 (2020).
Formica, S., González-García, C., Senoussi, M. & Brass, M. Neural oscillations track the maintenance and proceduralization of novel instructions. NeuroImage 232, 117870 (2021).
Formica, S., González-García, C., Senoussi, M., Marinazzo, D. & Brass, M. Theta-phase connectivity between medial prefrontal and posterior areas underlies novel instructions implementation. Preprint at bioRxiv https://doi.org/10.1101/2022.02.23.481594 (2022).
Cavanagh, J. F., Zambrano-Vazquez, L. & Allen, J. J. B. Theta lingua franca: a common mid-frontal substrate for action monitoring processes. Psychophysiology 49, 220–238 (2012).
Cohen, M. X. Midfrontal theta tracks action monitoring over multiple interactive time scales. NeuroImage 141, 262–272 (2016).
Nee, D. E. & D’Esposito, M. The hierarchical organization of the lateral prefrontal cortex. eLife 5, e12112 (2016).
Voytek, B. et al. Oscillatory dynamics coordinating human frontal networks in support of goal maintenance. Nat. Neurosci. 18, 1318–1324 (2015).
Badre, D. & D’Esposito, M. Is the rostro-caudal axis of the frontal lobe hierarchical? Nat. Rev. Neurosci. 10, 659–669 (2009).
Shahnazian, D., Senoussi, M., Krebs, R. M., Verguts, T. & Holroyd, C. B. Neural representations of task context and temporal order during action sequence execution. Top. Cogn. Sci. Advance online publication https://doi.org/10.1111/tops.12533 (2021).
Balaguer, J., Spiers, H., Hassabis, D. & Summerfield, C. Neural mechanisms of hierarchical planning in a virtual subway network. Neuron 90, 893–903 (2016).
Driel, J., van, Sligte, I. G., Linders, J., Elport, D. & Cohen, M. X. Frequency band-specific electrical brain stimulation modulates cognitive control processes. PLoS ONE 10, e0138984 (2015).
Lehr, A., Henneberg, N., Nigam, T., Paulus, W. & Antal, A. Modulation of conflict processing by theta-range tACS over the dorsolateral prefrontal cortex. Neural Plast. 2019, e6747049 (2019).
Riddle, J. & Frohlich, F. Targeting neural oscillations with transcranial alternating current stimulation. Brain Res. 1765, 147491 (2021).
O’Connell, R. G., Dockree, P. M. & Kelly, S. P. A supramodal accumulation-to-bound signal that determines perceptual decisions in humans. Nat. Neurosci. 15, 1729–1735 (2012).
Wyart, V., de Gardelle, V., Scholl, J. & Summerfield, C. Rhythmic fluctuations in evidence accumulation during decision making in the human brain. Neuron 76, 847–858 (2012).
Hagemann, D., Hewig, J., Walter, C. & Naumann, E. Skull thickness and magnitude of EEG alpha activity. Clin. Neurophysiol. 119, 1271–1280 (2008).
Voytek, B. et al. Hemicraniectomy: a new model for human electrophysiology with high spatio-temporal resolution. J. Cogn. Neurosci. 22, 2491–2502 (2010).
Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: a review and analysis. Brain Res. Rev. 29, 169–195 (1999).
Mierau, A., Klimesch, W. & Lefebvre, J. State-dependent alpha peak frequency shifts: experimental evidence, potential mechanisms and functional implications. Neuroscience 360, 146–154 (2017).
Haegens, S., Cousijn, H., Wallis, G., Harrison, P. J. & Nobre, A. C. Inter- and intra-individual variability in alpha peak frequency. NeuroImage 92, 46–55 (2014).
Minami, S., Oishi, H., Takemura, H. & Amano, K. Inter-individual differences in occipital alpha oscillations correlate with white matter tissue properties of the optic radiation. eNeuro 7, 2 (2020).
Grandy, T. H. et al. Peak individual alpha frequency qualifies as a stable neurophysiological trait marker in healthy younger and older adults. Psychophysiology 50, 570–582 (2013).
Jafari, Z., Kolb, B. E. & Mohajerani, M. H. Neural oscillations and brain stimulation in Alzheimer’s disease. Prog. Neurobiol. 194, 101878 (2020).
Pistono, A. et al. Language network connectivity increases in early Alzheimer’s disease. J. Alzheimers Dis. 82, 447–460 (2021).
Brem, A.-K. & Sensi, S. L. Towards combinatorial approaches for preserving cognitive fitness in aging. Trends Neurosci. 41, 885–897 (2018).
Chen, L., Chung, S. W., Hoy, K. E. & Fitzgerald, P. B. Is theta burst stimulation ready as a clinical treatment for depression? Expert Rev. Neurother. 19, 1089–1102 (2019).
Slobodskoy-Plusnin, J. Behavioral and brain oscillatory correlates of affective processing in subclinical depression. J. Clin. Exp. Neuropsychol. 40, 437–448 (2018).
Cohen, M. X. A neural microcircuit for cognitive conflict detection and signaling. Trends Neurosci. 37, 480–490 (2014).
Silvetti, M., Vassena, E., Abrahamse, E. & Verguts, T. Dorsal anterior cingulate–brainstem ensemble as a reinforcement meta-learner. PLoS Comput. Biol. 14, e1006370 (2018).
Sara, S. J. Locus Coeruleus in time with the making of memories. Curr. Opin. Neurobiol. 35, 87–94 (2015).
Silvetti, M., Wiersema, J. R., Sonuga-Barke, E. & Verguts, T. Deficient reinforcement learning in medial frontal cortex as a model of dopamine-related motivational deficits in ADHD. Neural Netw. 46, 199–209 (2013).
Bonnefond, M., Kastner, S. & Jensen, O. Communication between brain areas based on nested oscillations. eNeuro 4, 2 (2017).
Verbeke, P. & Verguts, T. Learning to synchronize: how biological agents can couple neural task modules for dealing with the stability–plasticity dilemma. PLoS Comput. Biol. 15, e1006604 (2019).
Bartos, M., Vida, I. & Jonas, P. Synaptic mechanisms of synchronized gamma oscillations in inhibitory interneuron networks. Nat. Rev. Neurosci. 8, 45–56 (2007).
Whittington, M. A., Cunningham, M. O., LeBeau, F. E. N., Racca, C. & Traub, R. D. Multiple origins of the cortical gamma rhythm. Dev. Neurobiol. 71, 92–106 (2011).
Tiesinga, P. & Sejnowski, T. J. Cortical enlightenment: are attentional gamma oscillations driven by ING or PING? Neuron 63, 727–732 (2009).
Wang, X. J., Golomb, D. & Rinzel, J. Emergent spindle oscillations and intermittent burst firing in a thalamic model: specific neuronal mechanisms. Proc. Natl Acad. Sci. USA 92, 5577–5581 (1995).
Gips, B., Eerden, J. P. J. Mvander & Jensen, O. A biologically plausible mechanism for neuronal coding organized by the phase of alpha oscillations. Eur. J. Neurosci. 44, 2147–2161 (2016).
Wong, K.-F. & Wang, X.-J. A recurrent network mechanism of time integration in perceptual decisions. J. Neurosci. 26, 1314–1328 (2006).
Jia, X., Xing, D. & Kohn, A. No consistent relationship between gamma power and peak frequency in macaque primary visual cortex. J. Neurosci. 33, 17–25 (2013).
Siegel, M., Warden, M. R. & Miller, E. K. Phase-dependent neuronal coding of objects in short-term memory. Proc. Natl Acad. Sci. USA 106, 21341–21346 (2009).
Atallah, B. V. & Scanziani, M. Instantaneous modulation of gamma oscillation frequency by balancing excitation with inhibition. Neuron 62, 566–577 (2009).
Palestro, J. J., Weichart, E., Sederberg, P. B. & Turner, B. M. Some task demands induce collapsing bounds: evidence from a behavioral analysis. Psychon. Bull. Rev. 25, 1225–1248 (2018).
Botvinick, M. M. & Cohen, J. D. The computational and neural basis of cognitive control: charted territory and new frontiers. Cogn. Sci. 38, 1249–1285 (2014).
Müller, M. G., Papadimitriou, C. H., Maass, W. & Legenstein, R. A model for structured information representation in neural networks. eNeuro 7, 3 (2020).
Peirce, J. et al. PsychoPy2: experiments in behavior made easy. Behav. Res. Methods 51, 195–203 (2019).
Dalmaijer, E. S., Mathôt, S. & Van der Stigchel, S. PyGaze: an open-source, cross-platform toolbox for minimal-effort programming of eyetracking experiments. Behav. Res. Methods 46, 913–921 (2014).
Gramfort, A. et al. MEG and EEG data analysis with MNE-Python. Front. Neurosci. 7, 267 (2013).
Virtanen, P. et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat. Methods 17, 261–272 (2020).
Manning, J. R., Jacobs, J., Fried, I. & Kahana, M. J. Broadband shifts in local field potential power spectra are correlated with single-neuron spiking in humans. J. Neurosci. 29, 13613–13620 (2009).
He, B. J. Scale-free brain activity: past, present, and future. Trends Cogn. Sci. 18, 480–487 (2014).
Voytek, B. et al. Age-related changes in 1/f neural electrophysiological noise. J. Neurosci. 35, 13257–13265 (2015).
Maris, E. & Oostenveld, R. Nonparametric statistical testing of EEG- and MEG-data. J. Neurosci. Methods 164, 177–190 (2007).
Gerster, M. et al. Separating neural oscillations from aperiodic 1/f activity: challenges and recommendations. Preprint at https://www.biorxiv.org/content/10.1101/2021.10.15.464483v1 (2021).
Kerby, D. S. The simple difference formula: an approach to teaching nonparametric correlation. Compr. Psychol. 3, 11.IT.3.1 (2014).
Vallat, R. Pingouin: statistics in Python. J. Open Source Softw. 3, 1026 (2018).
Rousselet, G. A. & Pernet, C. R. Improving standards in brain–behavior correlation analyses. Front. Hum. Neurosci. 6, 119 (2012).
Acknowledgements
The authors thank C. Buc Calderon for fruitful discussions and comments on the manuscript. M.S. and T.V. were supported by grant G012816 from Research Foundation Flanders. M.S., T.V. and E.D.L. were supported by grant BOF17-GOA-004 from the Research Council of Ghent University. P.V. was supported by grant 1102519N from Research Foundation Flanders. K.D. was supported by FWO [PEGASUS]² Marie Skłodowska-Curie fellowship 12T9717N. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.S., D.T. and T.V. designed the study. M.S., P.V. and T.V. developed the model. M.S. and E.D.L. collected the data. M.S. analysed model simulations, and behavioural and EEG data. M.S. and T.V. wrote the manuscript. All of the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks James Cavanagh, Sirawaj Itthipuripat and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Rule node competition and effect of MFC theta amplitude.
a Number of time points the correct rule node won the competition in the LFC unit depending on the instructed rule. Black bars represent the 95% Confidence Interval (smaller than dot size). The colour of each dot represents the rule, as represented in the rule names on the x-axis. b Time course of proportion of correct rule node winning the competition across a sample of the ISD. Only two rule nodes are shown for clarity, one easy (green curve) and one difficult (orange curve). Shaded area represents the 95% Confidence Interval. c Effect of MFC theta amplitude and frequency on competition time window. Average competition window length, in milliseconds, for one theta cycle, that is one competition window, as a function of theta frequency for different theta amplitude. Varying the amplitude (the different lines) shows that although the competition window increases with amplitude, this effect reaches a ceiling around amplitude values of 2-3. Each line represents simulations at different levels of MFC theta frequency with a fixed theta amplitude. Line color represents MFC theta amplitude as represented in the legend. d Varying MFC theta amplitude from 0.8 to 2.0 yielded similar results concerning what was the optimal theta frequency depending on rule difficulty (n = 34 simulations per theta frequency and for each theta amplitude, two-sided Wilcoxon sign-rank test: all W > 33, all ps < 0.002). Data are presented as violin plots, left- and right-most bars represent extrema, middle bar represent the median. Distribution density is represented by violin plot width.
Extended Data Fig. 2 Reaction times and control analyses with DDM.
Reaction times and DDM parameters (bound, drift rate and non-decision time) estimated in model and participant data with the EZ-Diffusion model (see Methods). a Reaction time and DDM parameters of model performance by rule difficulty (same-side, different-side) and theta frequency (4 to 7Hz, steps of 0.5Hz), n = 34 simulations per theta frequency. We ran a repeated-measure 2x7 ANOVA with factors rule difficulty (2 levels) and theta frequency (7 levels). There was a main effect of rule difficulty in all measures, that is reaction times, bound, drift rate and non-decision time, (all Fs(1, 33) > 98.96, all ps < 10-10, all η2 > 0.20). There was a main effect of theta frequency in reaction times, drift rate and nondecision time (all Fs(6, 33) > 3.20, all ps < 0.006, all η 2 > 0.01). There was a significant rule-difficulty – theta-frequency interaction in drift rate (F(6, 33) = 6.69, p < 0.001, η2 = 0.021). Error bars represent standard deviation across simulations. b Reaction time and DDM parameters (bound, drift rate and nondecision time) estimated on participants’ data grouped by rule (n = 34 participants). Data were collapsed across ISD to avoid data sparsity. We ran a repeated-measure 2x2 ANOVA with factors target-location (2 levels: Left, Right) and hand (2 levels: Left, Right). Only drift rate showed a significant interaction between hand and target-location (F(1, 33) = 31.86, p < 0.001, η2 = 0.21), as well as a main effect of hand (F(1, 33) = 6.65, p = 0.014, η2 = 0.02). Reaction time and bound showed a significant effect of hand (Reaction time: F(1, 33) = 4.62, p = 0.039, η2 = 0.04; bound: F(1, 33) = 4.84, p = 0.034, η2 = 0.03). All other effects were not significant (all Fs(1, 33) < 3.29, all ps > 0.079). Data are presented as mean values, error bars represent s.e.m. Smaller grey dots represent individual participants’ data.
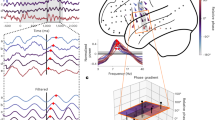
Extended Data Fig. 3 Raw spectra of individual participants per rule in Dataset 1.
The grey area represents the theta frequency band.
Extended Data Fig. 4 Control analyses on the effect of peak frequency and amplitude of nearby frequency bands.
Panels a to f: delta frequency band (1-3Hz). Panels g to l: alpha frequency band (8-12Hz). Two-way repeated-measure ANOVAs were used for all Dataset 1 data. Two-sided Wilcoxon sign-rank tests were used for all Dataset 2 and 3 data. a Peak frequency of delta band oscillations in Dataset 1 (all Fs < 0.78, all ps > 0.384). b Peak amplitude of delta band oscillations in Dataset 1 (all Fs < 0.76, all ps > 0.390). c Peak frequency of delta band oscillations in Dataset 2 (W = 26, p = 0.104, r = 0.50, 95% CI = (−0.00, 0.04)). d Peak amplitude of delta band oscillations in Dataset 2 (W = 49, p = 0.855, r = 0.07, 95% CI = (−0.02, 0.03)). e Peak frequency of delta band oscillations in Dataset 3 (W = 173, p = 0.091, r = − 0.34, 95% CI = (−0.07, 0.01)). f Peak amplitude of delta band oscillations in Dataset 3 (W = 195, p = 0.200, r = −0.26, 95% CI = (−0.04, 0.01)). g Peak frequency of alpha band oscillations in Dataset 1 (all Fs < 1.71, all ps > 0.199). h Peak amplitude of alpha band oscillations in Dataset 1. There was a main effect of target-location (F(1, 33) = 5.32, p = 0.027, η 2 = 0.051; uncorrected for multiple comparisons). i Peak frequency of alpha band oscillations in Dataset 2 (W = 29, p = 0.153, r = 0.44, 95% CI = (−0.05, 0.13)). j Peak amplitude of alpha band oscillations in Dataset 2 (W = 51, p = 0.951, r = 0.03, 95% CI = (−0.02, 0.02)). k Peak frequency of alpha band oscillations in Dataset 3 (W = 244, p = 0.715, r = − 0.07, 95% CI = (−0.05, 0.03)). l Peak amplitude of alpha band oscillations in Dataset 3 (W = 218, p = 0.394, r = −0.17, 95% CI = (−0.03, 0.01)). Data are presented as mean values, error bars represent s.e.m. computed over n = 34, 14 and 33 participants for Dataset 1, 2 and 3, respectively. Smaller grey dots represent individual participants’ data.
Supplementary information
Supplementary information
Supplementary Figs. 1–8 and methods (model equations),
Rights and permissions
About this article
Cite this article
Senoussi, M., Verbeke, P., Desender, K. et al. Theta oscillations shift towards optimal frequency for cognitive control. Nat Hum Behav 6, 1000–1013 (2022). https://doi.org/10.1038/s41562-022-01335-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-022-01335-5