Abstract
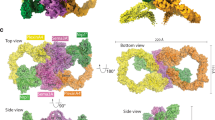
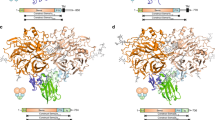
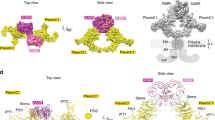
Co-receptors add complexity to cell-cell signaling systems. The secreted semaphorin 3s (Sema3s) require a co-receptor, neuropilin (Nrp), to signal through plexin As (PlxnAs) in functions ranging from axon guidance to bone homeostasis, but the role of the co-receptor is obscure. Here we present the low-resolution crystal structure of a mouse semaphorin–plexin–Nrp complex alongside unliganded component structures. Dimeric semaphorin, two copies of plexin and two copies of Nrp are arranged as a dimer of heterotrimers. In each heterotrimer subcomplex, semaphorin contacts plexin, similar to in co-receptor–independent signaling complexes. The Nrp1s cross brace the assembly, bridging between sema domains of the Sema3A and PlxnA2 subunits from the two heterotrimers. Biophysical and cellular analyses confirm that this Nrp binding mode stabilizes a canonical, but weakened, Sema3–PlxnA interaction, adding co-receptor control over the mechanism by which receptor dimerization and/or oligomerization triggers signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kolodkin, A.L. & Tessier-Lavigne, M. Mechanisms and molecules of neuronal wiring: a primer. Cold Spring Harb. Perspect. Biol. 3, a001727 (2011).
Tran, T.S., Kolodkin, A.L. & Bharadwaj, R. Semaphorin regulation of cellular morphology. Annu. Rev. Cell Dev. Biol. 23, 263–292 (2007).
Zhou, Y., Gunput, R.A. & Pasterkamp, R.J. Semaphorin signaling: progress made and promises ahead. Trends Biochem. Sci. 33, 161–170 (2008).
Takamatsu, H. & Kumanogoh, A. Diverse roles for semaphorin-plexin signaling in the immune system. Trends Immunol. 33, 127–135 (2012).
Muratori, C. & Tamagnone, L. Semaphorin signals tweaking the tumor microenvironment. Adv. Cancer Res. 114, 59–85 (2012).
Janssen, B.J. et al. Structural basis of semaphorin-plexin signalling. Nature 467, 1118–1122 (2010).
Nogi, T. et al. Structural basis for semaphorin signalling through the plexin receptor. Nature 467, 1123–1127 (2010).
He, Z. & Tessier-Lavigne, M. Neuropilin is a receptor for the axonal chemorepellent Semaphorin III. Cell 90, 739–751 (1997).
Kolodkin, A.L. et al. Neuropilin is a semaphorin III receptor. Cell 90, 753–762 (1997).
Tamagnone, L. et al. Plexins are a large family of receptors for transmembrane, secreted, and GPI-anchored semaphorins in vertebrates. Cell 99, 71–80 (1999).
Takahashi, T. et al. Plexin–neuropilin-1 complexes form functional semaphorin-3A receptors. Cell 99, 59–69 (1999).
Soker, S., Takashima, S., Miao, H.Q., Neufeld, G. & Klagsbrun, M. Neuropilin-1 is expressed by endothelial and tumor cells as an isoform-specific receptor for vascular endothelial growth factor. Cell 92, 735–745 (1998).
Antipenko, A. et al. Structure of the semaphorin-3A receptor binding module. Neuron 39, 589–598 (2003).
Love, C.A. et al. The ligand-binding face of the semaphorins revealed by the high-resolution crystal structure of SEMA4D. Nat. Struct. Biol. 10, 843–848 (2003).
Liu, H. et al. Structural basis of semaphorin-plexin recognition and viral mimicry from Sema7A and A39R complexes with PlexinC1. Cell 142, 749–761 (2010).
Tong, Y. et al. Structure and function of the intracellular region of the plexin-b1 transmembrane receptor. J. Biol. Chem. 284, 35962–35972 (2009).
He, H., Yang, T., Terman, J.R. & Zhang, X. Crystal structure of the plexin A3 intracellular region reveals an autoinhibited conformation through active site sequestration. Proc. Natl. Acad. Sci. USA 106, 15610–15615 (2009).
Bell, C.H., Aricescu, A.R., Jones, E.Y. & Siebold, C. A dual binding mode for RhoGTPases in plexin signalling. PLoS Biol. 9, e1001134 (2011).
Wang, Y. et al. Plexins are GTPase-activating proteins for Rap and are activated by induced dimerization. Sci. Signal. 5, ra6 (2012).
Chen, H., He, Z., Bagri, A. & Tessier-Lavigne, M. Semaphorin-neuropilin interactions underlying sympathetic axon responses to class III semaphorins. Neuron 21, 1283–1290 (1998).
Giger, R.J. et al. Neuropilin-2 is a receptor for semaphorin IV: insight into the structural basis of receptor function and specificity. Neuron 21, 1079–1092 (1998).
Nakamura, F., Tanaka, M., Takahashi, T., Kalb, R.G. & Strittmatter, S.M. Neuropilin-1 extracellular domains mediate semaphorin D/III-induced growth cone collapse. Neuron 21, 1093–1100 (1998).
Appleton, B.A. et al. Structural studies of neuropilin/antibody complexes provide insights into semaphorin and VEGF binding. EMBO J. 26, 4902–4912 (2007).
Gaur, P. et al. Role of class 3 semaphorins and their receptors in tumor growth and angiogenesis. Clin. Cancer Res. 15, 6763–6770 (2009).
Rohm, B., Ottemeyer, A., Lohrum, M. & Puschel, A.W. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. Mech. Dev. 93, 95–104 (2000).
Chen, G. et al. Semaphorin-3A guides radial migration of cortical neurons during development. Nat. Neurosci. 11, 36–44 (2008).
Takahashi, T. & Strittmatter, S.M. PlexinA1 autoinhibition by the plexin sema domain. Neuron 29, 429–439 (2001).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Toyofuku, T. et al. Repulsive and attractive semaphorins cooperate to direct the navigation of cardiac neural crest cells. Dev. Biol. 321, 251–262 (2008).
Adams, R.H., Lohrum, M., Klostermann, A., Betz, H. & Puschel, A.W. The chemorepulsive activity of secreted semaphorins is regulated by furin-dependent proteolytic processing. EMBO J. 16, 6077–6086 (1997).
Parker, M.W., Hellman, L.M., Xu, P., Fried, M.G. & Vander Kooi, C.W. Furin processing of semaphorin 3F determines its anti-angiogenic activity by regulating direct binding and competition for neuropilin. Biochemistry 49, 4068–4075 (2010).
Merte, J. et al. A forward genetic screen in mice identifies Sema3A(K108N), which binds to neuropilin-1 but cannot signal. J. Neurosci. 30, 5767–5775 (2010).
Ueyama, H. et al. Semaphorin 3A lytic hybrid peptide binding to neuropilin-1 as a novel anti-cancer agent in pancreatic cancer. Biochem. Biophys. Res. Commun. 414, 60–66 (2011).
Shirvan, A. et al. Semaphorins as mediators of neuronal apoptosis. J. Neurochem. 73, 961–971 (1999).
Shirvan, A. et al. Anti-semaphorin 3A antibodies rescue retinal ganglion cells from cell death following optic nerve axotomy. J. Biol. Chem. 277, 49799–49807 (2002).
Gu, C. et al. Characterization of neuropilin-1 structural features that confer binding to semaphorin 3A and vascular endothelial growth factor 165. J. Biol. Chem. 277, 18069–18076 (2002).
Hayashi, M. et al. Osteoprotection by semaphorin 3A. Nature 485, 69–74 (2012).
Roth, L. et al. Transmembrane domain interactions control biological functions of neuropilin-1. Mol. Biol. Cell 19, 646–654 (2008).
Aricescu, A.R., Lu, W. & Jones, E.Y. A time- and cost-efficient system for high-level protein production in mammalian cells. Acta Crystallogr. D Biol. Crystallogr. 62, 1243–1250 (2006).
O'Callaghan, C.A. et al. BirA enzyme: production and application in the study of membrane receptor-ligand interactions by site-specific biotinylation. Anal. Biochem. 266, 9–15 (1999).
Koppel, A.M., Feiner, L., Kobayashi, H. & Raper, J.A. A 70 amino acid region within the semaphorin domain activates specific cellular response of semaphorin family members. Neuron 19, 531–537 (1997).
Reeves, P.J., Callewaert, N., Contreras, R. & Khorana, H.G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl. Acad. Sci. USA 99, 13419–13424 (2002).
Chang, V.T. et al. Glycoprotein structural genomics: solving the glycosylation problem. Structure 15, 267–273 (2007).
Walter, T.S. et al. A procedure for setting up high-throughput nanolitre crystallization experiments. Crystallization workflow for initial screening, automated storage, imaging and optimization. Acta Crystallogr. D Biol. Crystallogr. 61, 651–657 (2005).
Mayo, C.J. et al. Benefits of automated crystallization plate tracking, imaging, and analysis. Structure 13, 175–182 (2005).
Leslie, A.G.W. Recent changes to the MOSFLM package for processing film and image plate data. Joint CCP4 + ESF-EAMCB Newsletter on Protein Crystallography 26 (1992).
CCP4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D Biol. Crystallogr. 50, 760–763 (1994).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Adams, P.D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D Biol. Crystallogr. 58, 1948–1954 (2002).
Davis, I.W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007).
Dolinsky, T.J., Nielsen, J.E., McCammon, J.A. & Baker, N.A. PDB2PQR: an automated pipeline for the setup of Poisson-Boltzmann electrostatics calculations. Nucleic Acids Res. 32, W665–W667 (2004).
Baker, N.A., Sept, D., Joseph, S., Holst, M.J. & McCammon, J.A. Electrostatics of nanosystems: application to microtubules and the ribosome. Proc. Natl. Acad. Sci. USA 98, 10037–10041 (2001).
Larkin, M.A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Landau, M. et al. ConSurf 2005: the projection of evolutionary conservation scores of residues on protein structures. Nucleic Acids Res. 33, W299–W302 (2005).
Krissinel, E. & Henrick, K. Inference of macromolecular assemblies from crystalline state. J. Mol. Biol. 372, 774–797 (2007).
Gasteiger, E. et al. ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res. 31, 3784–3788 (2003).
Malin, S.A., Davis, B.M. & Molliver, D.C. Production of dissociated sensory neuron cultures and considerations for their use in studying neuronal function and plasticity. Nat. Protoc. 2, 152–160 (2007).
Kapfhammer, J.P., Xu, H. & Raper, J.A. The detection and quantification of growth cone collapsing activities. Nat. Protoc. 2, 2005–2011 (2007).
Acknowledgements
We thank the staff of Diamond beamlines I02, I03 and I04-1 for assistance with diffraction data collection, Y. Zhao and W. Lu for help with protein expression, T.S. Walter for help with crystallization, G. Sutton for help with multiangle light scattering (MALS) experiments and A.R. Aricescu and D.I. Stuart for critical reading of the manuscript. This work was funded by Cancer Research UK (A10976) and the Medical Research Council (G9900061) to E.Y.J. B.J.C.J. is funded by the Human Frontier Science Program, G.A.W. and M.Z.C. are funded by the Medical Research Council and the John Fell Fund, C.S. is funded by the Wellcome Trust and E.Y.J. is funded by Cancer Research UK (A5261).
Author information
Authors and Affiliations
Contributions
B.J.C.J., T.M., C.S. and E.Y.J. designed the project. B.J.C.J. and E.Y.J. wrote the manuscript with input from all authors. B.J.C.J. produced the constructs for crystallization and performed MALS, crystallization, diffraction data collection, structure solution and refinement. T.M. and B.J.C.J. cloned and produced constructs for and performed SPR experiments. T.M. did the western blot analysis. T.M. produced proteins for growth cone–collapse assays, which G.A.W. performed under the supervision of M.Z.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 5023 kb)
Rights and permissions
About this article
Cite this article
Janssen, B., Malinauskas, T., Weir, G. et al. Neuropilins lock secreted semaphorins onto plexins in a ternary signaling complex. Nat Struct Mol Biol 19, 1293–1299 (2012). https://doi.org/10.1038/nsmb.2416
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2416
This article is cited by
-
Cerebral Semaphorin3D is a novel risk factor for age-associated cognitive impairment
Cell Communication and Signaling (2023)
-
Plexin-A2 enables the proliferation and the development of tumors from glioblastoma derived cells
Cell Death & Disease (2023)
-
Semaphorin3F Drives Dendritic Spine Pruning Through Rho-GTPase Signaling
Molecular Neurobiology (2021)
-
Cryo-EM structure of the PlexinC1/A39R complex reveals inter-domain interactions critical for ligand-induced activation
Nature Communications (2020)
-
Diversity of oligomerization in Drosophila semaphorins suggests a mechanism of functional fine-tuning
Nature Communications (2019)