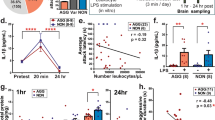
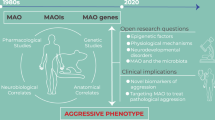
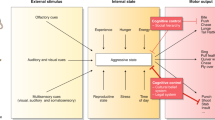
Key Points
-
Dysregulated aggressive behaviour has important negative consequences for human societies. A complicating factor is that aggression that is exhibited in different social contexts can be regulated by different neurobiological mechanisms.
-
Neurobiological studies have identified a subset of hypothalamic and limbic brain areas that tend to facilitate aggressive behaviour in rodents and primates. In contrast, neural activity in the frontal cortex generally acts to inhibit aggressive behaviour.
-
Aggressive behaviours in animal models and humans are known to be regulated by serotonin neurotransmission. Behaviour can be modified at several levels, including regulation of serotonin release, reuptake and sensitivity (via serotonin receptors).
-
Dopaminergic function appears to be necessary for aggressive behaviour, possibly by regulating arousal, learning and memory.
-
Neuronal nitric oxide (nNOS) synthase signalling (via nitric oxide gas) exerts inhibitory effects on male aggression in rodents. Several studies suggest that nNOS assists in the processing of salient social stimuli.
-
Mutations in the monoamine oxidase A (MAOA) enzyme are associated with increased aggressive behaviours in humans. MAOA knockout mice show increased aggression.
-
Steroid hormones have long been a focus of aggression research, but the relationship among androgens, oestrogens and behaviour is complex. These hormones do not function in isolation and their actions are affected by the environmental context.
-
Gene–environment interactions have important effects on aggressive behaviours. Mutations or hormones that increase aggression in one environment have no effect (or decrease aggression) in different environments.
-
Novel pharmacological treatments must target specific subtypes of aggression to have improved effectiveness. An appreciation of the contribution of environmental stressors to aggressive phenotypes is necessary for further advancements in the successful management of maladaptive aggression.
Abstract
Unchecked aggression and violence exact a significant toll on human societies. Aggression is an umbrella term for behaviours that are intended to inflict harm. These behaviours evolved as adaptations to deal with competition, but when expressed out of context, they can have destructive consequences. Uncontrolled aggression has several components, such as impaired recognition of social cues and enhanced impulsivity. Molecular approaches to the study of aggression have revealed biological signals that mediate the components of aggressive behaviour. These signals may provide targets for therapeutic intervention for individuals with extreme aggressive outbursts. This Review summarizes the complex interactions between genes, biological signals, neural circuits and the environment that influence the development and expression of aggressive behaviour.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Berkowitz, L. Aggression: Its Causes, Consequences, and Control (Temple Univ. Press, New York, 1993).
Moyer, K. E. The Physiology of Hostility, (Markham, Chicago, 1971).
Miczek, K. A., Fish, E. W., de Bold, J. F. & de Almeida, R. M. M. Social and neural determinants of aggressive behavior: pharmacotherapeutic targets at serotonin, dopamine and γ-aminobutyric acid systems. Psychopharmacology (Berl.) 163, 434–458 (2002).
Blair, R. J. R., Peschardt, K. S., Budhani, S. & Pine, D. S., in Biology of Aggression (ed. Nelson, R. J.) 351–368 (Oxford Univ. Press, New York, 2006).
Vitiello, B. & Stoff, D. M. Subtypes of aggression and their relevance to child psychiatry. J. Am. Acad. Child Adolesc. Psychiatry 36, 307–315 (1997). This paper emphasized that our understanding and treatment of human aggressive behaviour would benefit from research on specific subtypes of aggression. The authors cluster analyses indicated that a differentiation between the impulsive–affective and controlled–predatory subtypes as qualitatively different forms of aggressive behaviour is a promising construct.
Viding, E., Frick, P. J. & Plomin, R. Aetiology of the relationship between callous–unemotional traits and conduct problems in childhood. Br. J. Psychiatry 190, 33–38 (2007).
Raine, A. Annotation: the role of prefrontal deficits, low autonomic arousal, and early health factors in the development of antisocial and aggressive behavior in children. J. Child Psychol. Psychiatry 43, 417–737 (2002). This paper was the first to review the importance of reduced autonomic arousal in the development of antisocial and aggressive behaviour in children.
Connor, D. F., Boone, R. T., Steingard, R. J., Lopez, I. D. & Melloni, R. H. Psychopharmacology and aggression II: a meta-analysis of nonstimulant medication effects on overt aggression-related behaviors in youth with SED. J. Emot. Behav. Disord. 11, 157–168 (2003).
Swann, A. C. Neuroreceptor mechanisms of aggression and its treatment. J. Clin. Psychiatry 64, 26–35 (2003).
Newman, S. The medial extended amygdala in male reproductive behavior. A node in the mammalian social behavior network. Ann. NY Acad. Sci. 877, 242–257 (1999). This review summarizes studies that examined the social behaviour brain circuit and highlights how brain nuclei function in different contexts.
DaVanzo, J. P., Sydow, M. & Garris, D. R. Influence of isolation and training on fighting in mice with olfactory bulb lesions. Physiol. Behav. 31, 857–860 (1983).
Delville, Y., De Vries, G. J. & Ferris, C. F. Neural connections of the anterior hypothalamus and agonistic behavior in golden hamsters. Brain Behav. Evol. 55, 53–76 (2000).
Swanson, L. W. Cerebral hemisphere regulation of motivated behavior. Brain Res. 886, 113–164 (2000).
Kruk, M. R. Ethology and pharmacology of hypothalamic aggression in the rat. Neurosci. Biobehav. Rev. 15, 527–538 (1991).
de Bruin, J. P., van Oyen, H. G. & Van de Poll, N. Behavioural changes following lesions of the orbital prefrontal cortex in male rats. Behav. Brain Res. 10, 209–232 (1983).
Kruk, M. R. et al. Comparison of aggressive behaviour induced by electrical stimulation in the hypothalamus of male and female rats. Prog. Brain Res. 61, 303–314 (1984).
Ferris, C. F. & Potegal, M. Vasopressin receptor blockade in the anterior hypothalamus suppresses aggression in hamsters. Physiol. Behav. 44, 235–239 (1988).
Kollack-Walker, S. & Newman, S. W. Mating and agonistic behavior produce different patterns of FOS immunolabeling in the male Syrian hamster brain. Neuroscience 66, 721–736 (1995).
Davis, E. S. & Marler, C. A. C-fos changes following an aggressive encounter in female California mice: a synthesis of behavior, hormone changes and neural activity. Neuroscience 127, 611–624 (2004).
Hasen, N. S. & Gammie, S. C. Differential FOS activation in virgin and lactating mice in response to an intruder. Physiol. Behav. 84, 681–695 (2005).
Lipp, H. P. & Hunsperger, R. W. Threat, attack, and flight elicited by electrical stimulation of the ventromedial hypothalamus of the marmoset monkey Callithrix jacchus. Brain Behav. Evol. 15, 260–293 (1978).
Dixson, A. F. & Lloyd, S. A. C. Effects of hypothalamic lesions upon sexual and social behaviour of the male common marmoset (Callithrix jacchus). Brain Res. 463, 317–329 (1988).
Robinson, B. W. Vocalization evoked from forebrain in Macaca mulatta. Physiol. Behav. 2, 345–354 (1967).
Alexander, M. & Perachio, A. A. The influence of target sex and dominance on evoked attack in rhesus monkeys. Am. J. Phys. Anthropol. 38, 543–548 (1973).
Machado, C. J. & Bachevalier, J. The impact of selective amygdala, orbital frontal cortex, or hippocampal formation lesions on established social relationships in rhesus monkeys (Macaca mulatta). Behav. Neurosci. 120, 761–786 (2006).
Emery, N. J. et al. The effects of bilateral lesions of the amygdala on dyadic social interactions in rhesus monkeys (Macaca mulatta). Behav. Neurosci. 115, 515–544 (2001).
Butter, C. M. & Snyder, D. R. Alterations in aversive and aggressive behaviors following orbital frontal lesions in rhesus monkeys. Acta Neurobiol. Exp. 32, 525–565 (1972).
Gregg, T. R. & Siegel, A. Brain structures and neurotransmitters regulating aggression in cats: implications for human aggression. Prog. Neuropsychopharmacol. Biol. Psychiatry 25, 91–140 (2001).
Davidson, R. J., Putnam, K. M. & Larson, C. L. Dysfunction in the neural circuitry of emotion regulation — a possible prelude to violence. Science 289, 591–594 (2000).
Anderson, S. W., Bechara, A., Damasio, H., Tranel, D. & Damasio, A. R. Impairment of social and moral behavior related to early damage in human prefrontal cortex. Nature Neurosci. 2, 1032–1037 (1999).
Volkow, N. D. et al. Brain glucose metabolism in violent psychiatric patients: a preliminary study. Psychiatry Res. 61, 243–253 (1995).
Soloff, P. H. et al. Impulsivity and prefrontal hypometabolism in borderline personality disorder. Psychiatry Res. 123, 153–163 (2003).
Coccaro, E. F., McCloskey, M. S., Fitzgerald, D. A. & Phan, K. L. Amygdala and orbitofrontal reactivity to social threat in individuals with impulsive aggression. Biol. Psychiatry 8 January 2007 (doi: 10.1016/j.biopsych.2006.08.024).
Heimburger, R. F., Whillock, C. C. & Kalsbeck, J. E. Stereotaxic amygdalotomy for epilepsy with aggressive behavior. JAMA 198, 741–745 (1966).
Scarpa, A. & Raine, A. Psychophysiology of anger and violent behavior. Psychiatr. Clin. North Am. 20, 375–394 (1997).
Tonkonogy, J. M. & Geller, J. L. Hypothalamic lesions and intermittent explosive disorder. J. Neuropsychiatry Clin. Neurosci. 4, 45–50 (1992).
Coccaro, E. F. & Kavoussi, R. J. Fluoxetine and impulsive aggressive behavior in personality-disordered subjects. Arch. Gen. Psychiatry 54, 1081–1088 (1997).
New, A. S. et al. Fluoxetine increases relative metabolic rate in the prefrontal cortex in impulsive aggression. Psychopharmacology (Berl.) 176, 451–458 (2004).
Parsey, R. V. et al. Effects of sex, age, and aggressive traits in men on brain serotonin 5-HT1A receptor binding potential measured by PET using [C–11]WAY-100635. Brain Res. 954, 173–182 (2002).
Kirsch, P. et al. Oxytocin modulates neural circuitry for social cognition and fear in humans. J. Neurosci. 25, 11489–11493 (2005).
Winslow, J. T. & Insel, T. R. Social status in pairs of male squirrel monkeys determines the behavioral response to central oxytocin administration. J. Neurosci. 11, 2032–2038 (1991).
Winslow, J. T. et al. Infant vocalization, adult aggression, and fear behavior of an oxytocin null mutant mouse. Horm. Behav. 37, 145–155 (2000).
DeVries, A. C., Young, S. W. & Nelson, R. J. Reduced aggressive behavior in mice with targeted disruption of the oxytocin gene. J. Neuroendocrinol. 9, 363–368 (1997).
Insel, T. R. Oxytocin — a neuropeptide for affiliation: evidence from behavioral, receptor autoradiographic, and comparative studies. Psychoneuroendocrinology 17, 3–35 (1992).
Manuck, S. B., Kaplan, J. R. & Lotrich, F. E. in Biology of Aggression (ed. Nelson, R. J.) 65–113 (Oxford University Press, New York, 2006).
Chiavegatto, S. et al. Brain serotonin dysfunction accounts for aggression in male mice lacking neuronal nitric oxide synthase. Proc. Natl Acad. Sci. USA 98, 1277–1281 (2001).
Miczek, K. A., Maxson, S. C., Fish, E. W. & Faccidomo, S. Aggressive behavioral phenotypes in mice. Behav. Brain Res. 125, 167–181 (2001).
Nelson, R. J. & Chiavegatto, S. Molecular basis of aggression. Trends Neurosci. 24, 713–719 (2001).
Olivier, B. Serotonergic mechanisms in aggression. Novartis Found. Symp. 268, 171–183 (2005).
Saudou, F. et al. Enhanced aggressive behavior in mice lacking 5-HT1B receptor. Science 265, 1875–1878 (1994).
Ramboz, S. et al. 5-HT1B receptor knock out — behavioral consequences. Behav. Brain Res. 73, 305–312 (1996).
Korte, S. M. et al. Enhanced 5-HT1A receptor expression in forebrain regions of aggressive house mice. Brain Res. 736, 338–343 (1996).
Ase, A. R., Reader, T. A., Hen, R., Riad, M. & Descarries, L. Altered serotonin and dopamine metabolism in the CNS of serotonin 5-HT1A or 5-HT1B receptor knockout mice. J. Neurochem. 75, 2415–2426 (2000).
Holmes, A., Murphy, D. L. & Crawley, J. N. Reduced aggression in mice lacking the serotonin transporter. Psychopharmacology (Berl.) 161, 160–167 (2002).
Parsey, R. V. et al. Effects of sex, age, and aggressive traits in man on brain serotonin 5-HT1A receptor binding potential measured by PET using [C–11]WAY-100635. Brain Res. 954, 173–182 (2002).
de Boer, S. F. & Koolhaas, J. M. 5-HT1A and 5-HT1B receptor agonists and aggression: a pharmacological challenge of the serotonin deficiency hypothesis. Eur. J. Pharmacol. 526, 125–139 (2005).
de Almeida, R. M. M., Ferrari, P. M., Parmigiani, S. & Miczek, K. A. Escalated aggressive behavior: dopamine, serotonin and GABA. Eur. J. Pharmacol. 526, 51–64 (2005).
Ferrari, P. F., van Erp, A. M., Tornatzky, W. & Miczek, K. A. Accumbal dopamine and serotonin in anticipation of the next aggressive episode in rats. Eur. J. Neurosci. 17, 371–378 (2003). This study is unique because it measures dynamic changes in neurotransmitter levels during aggressive encounters in male rats. Interestingly, when aggression tests were regularly scheduled, increased dopamine and decreased 5-HT levels were observed in anticipation of aggressive encounters.
Vukhac, K. L., Sankoorikal, E. B. & Wang, Y. Dopamine D2L receptor- and age-related reduction in offensive aggression. Neuroreport 12, 1035–1038 (2001).
Rodriguiz, R. M., Chu, R., Caron, M. G. & Wetsel, W. C. Aberrant responses in social interaction of dopamine transporter knockout mice. Behav. Brain Res. 148, 185–198 (2004).
Miczek, K. A. & Fish, E. W. in Biology of Aggression (ed. Nelson, R. J.) 114–149 (Oxford Univ. Press, New York, 2006).
Clement, J. et al. Age-dependent changes of brain GABA levels, turnover rates and shock-induced aggressive behavior in inbred strains of mice. Pharmacol. Biochem. Behav. 26, 83–88 (1987).
Pfaff, D. W. Drive: Neurobiological and Molecular Mechanisms of Sexual Motivation. (MIT Press, Massachusetts, USA, 1999).
Haller, J. & Kruk, M. R. Normal and abnormal aggression: human disorders and novel laboratory models. Neurosci. Biobehav. Rev. 30, 292–303 (2006).
Marino, M. D., Bourdelat-Parks, B. N., Cameron, L. L. & Weinshenker, D. Genetic reduction of noradrenergic function alters social memory and reduces aggression in mice. Behav. Brain Res. 161, 197–203 (2005).
Snyder, S. H. Neuroscience at Johns Hopkins. Neuron 48, 201–211 (2005).
Nelson, R. J. et al. Behavioural abnormalities in male mice lacking neuronal nitric oxide synthase. Nature 378, 383–386 (1995). The first report to show a role for neuronal nitric oxide synthase in regulating behaviour. Nos1−/− mice were insensitive to social cues; they continued to attack other males that showed submissive displays and attempted to mate with females that were not sexually receptive.
Kriegsfeld, L. J., Dawson, T. M., Dawson, V. L., Nelson, R. J. & Snyder, S. H. Aggressive behavior in male mice lacking the gene for nNOS is testosterone-dependent. Brain Res. 769, 66–70 (1997).
Trainor, B. C., Workman, J. L., Jessen, R. & Nelson, R. J. Impaired nitric oxide synthase signaling dissociates social investigation and aggression. Behav. Neurosci. 121, 362–369 (2007).
Shih, J. C., Chen, K. & Ridd, M. J. Monoamine oxidase: from genes to behavior. Annu. Rev. Neurosci. 22, 197–217 (1999). This review summarizes converging evidence for the roles of MAOA and MAOB in the regulation of aggressive behaviour in mice.
Brunner, H. G., Nelen, M., Breakefield, X. O., Ropers, H. H. & van Oost, B. A. Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science 262, 578–580 (1993).
Cases, O. et al. Aggressive behavior and altered amounts of brain serotonin and norepinephrine in mice lacking MAOA. Science 268, 1763–1766 (1995).
Mossner, R. et al. Differential regulation of adenosine A1 and A2A receptors in serotonin transporter and monoamine oxidase A-deficient mice. Eur. Neuropsychopharmacol. 10, 489–493 (2000).
Chen, K. et al. Forebrain-specific expression of monoamine oxidase A reduces neurotransmitter levels, restores the brain structure, and rescues aggressive behavior in monoamine oxidase A-deficient mice. J. Biol. Chem. 282, 115–123 (2007).
Sabol, S. Z., Hu, S. & Hamer, D. A functional polymorphism in the monoamine oxidase A gene promoter. Hum. Genet. 103, 273–279 (1998).
Manuck, S. B., Flory, J. D., Ferrell, R. E., Mann, J. J. & Muldoon, M. F. A regulatory polymorphism of the monoamine oxidase-A gene may be associated with variability in aggression, impulsivity, and central nervous system responsivity. Psychiatry Res. 95, 9–23 (2000).
Beitchman, J. H., Mik, H. M., Ehtesham, S., Douglas, L. & Kennedy, J. L. MAOA and persistent, pervasive childhood aggression. Mol. Psychiatry 9, 546–547 (2004).
Demas, G. E., Moffatt, C. A., Drazen, D. L. & Nelson, R. J. Castration does not inhibit aggressive behavior in adult male prairie voles (Microtus ochrogaster). Physiol. Behav. 66, 59–62 (1999).
Trainor, B. C., Greiwe, K. M. & Nelson, R. J. Individual differences in estrogen receptor α in select brain nuclei are associated with individual differences in aggression. Horm. Behav. 50, 338–345 (2006).
Field, E. F., Whishaw, I. Q., Pellis, S. M. & Watson, N. V. Play fighting in androgen-insensitive tfm rats: evidence that androgen receptors are necessary for the development of adult playful attack and defense. Dev. Psychobiol. 48, 111–120 (2006).
Simon, N. G., Cologer-Clifford, A., Lu, S. F., McKenna, S. E. & Hu, S. Testosterone and its metabolites modulate 5HT1A and 5HT1B agonist effects on intermale aggression. Neurosci. Biobehav. Rev. 23, 325–336 (1998).
Siegel, A., Roeling, T. A., Gregg, T. R. & Kruk, M. R. Neuropharmacology of brain-stimulation-evoked aggression. Neurosci. Biobehav. Rev. 23, 359–389 (1999).
Trainor, B. C., Kyomen, H. H. & Marler, C. A. Estrogenic encounters: how interactions between aromatase and the environment modulate aggression. Front Neuroendocrinol. 27, 170–179 (2006).
Ogawa, S., Lubahn, D. B., Korach, K. S. & Pfaff, D. W. Behavioral effects of estrogen receptor gene disruption in male mice. Proc. Natl Acad. Sci. USA 94, 1476–1481 (1997).
Scordalakes, E. M. & Rissman, E. F. Aggression in male mice lacking functional estrogen receptor α. Behav. Neurosci. 117, 38–45 (2003).
Ogawa, S. et al. Survival of reproductive behaviors in estrogen receptor β gene-deficient (βERKO) male and female mice. Proc. Natl Acad. Sci. USA 96, 12887–12892 (1999).
Nomura, M. et al. Estrogen receptor-β gene disruption potentiates estrogen-inducible aggression but not sexual behaviour in male mice. Eur. J. Neurosci. 23, 1860–1868 (2006).
Nomura, M. et al. Genotype/age interactions on aggressive behavior in gonadally intact estrogen receptor β knockout (βERKO) male mice. Horm. Behav. 41, 288–296 (2002).
Scott, J. P. Genetic differences in the social behavior of inbred strains of mice. J. Hered. 33, 11–15 (1942).
Ginsburg, B. E. & Allee, W. C. Some effects of conditioning on social dominance and subordination in inbred strains of mice. Physiol. Zool. 15, 485–506 (1942).
Young, K. A. et al. Fierce: a new mouse deletion of Nr2e1; violent behaviour and ocular abnormalities are background-dependent. Behav. Brain Res. 132, 145–158 (2002).
Abrahams, B. S. et al. Pathological aggression in 'fierce' mice corrected by human nuclear receptor 2E1. J. Neurosci. 25, 6263–6270, (2005).
Caspi, A. et al. Role of genotype in the cycle of violence in maltreated children. Science 297, 851–854 (2003). This report helped spark renewed interest in studying the mechanisms of gene–environment interactions in humans. It shows that individuals that are homozygous for the short allele of the 5-HT transporter promoter are more likely to suffer mental disorders, but only if they were maltreated as children.
Kim-Cohen, J. et al. MAOA, maltreatment, and gene–environment interaction predicting children's mental health: new evidence and a meta-analysis. Mol. Psychiatry 11, 903–913 (2006).
Caspi, A. et al. Influence of life stress on depression: moderation by polymorphism in the 5-HTT gene. Science, 301, 386–389 (2003).
Greenberg, B. D. et al. Genetic variation in the serotonin transporter promoter region affects serotonin uptake in human blood platelets. Am. J. Med. Genet. 88, 83–87 (1999).
Barr, C. S. et al. Rearing condition and rh5-HTTLPR interact to influence limbic-hypothalamic-pituitary-adrenal axis response to stress in infant macaques. Biol. Psychiatry 55, 733–738 (2004).
Canli, T. et al. Neural correlates of epigenesis. Proc. Natl Acad. Sci. USA 103, 16033–16038 (2006). This study looked at how the interaction between the 5-HT transporter and the environment influences brain function. Life stressors were found to affect functional connectivity in the amygdala and hippocampus as a function of 5-HT transporter genotype.
Verona, E., Joiner, T. E., Johnson, F. & Bender, T. W. Gender specific gene–environment interactions on laboratory-assessed aggression. Biol. Psychol. 71, 33–41 (2006).
Hariri, A. R. et al. Serotonin transporter genetic variation and the response of the human amygdala. Science 297, 400–403 (2002).
Trainor, B. C., Lin, S., Finy, M. S., Rowland, M. R. & Nelson, R. J. Photoperiod reverses the effects of estrogen on male aggression via genomic and non-genomic pathways. Proc. Natl Acad. Sci. USA 5 June 2007 (doi: 10.1073/pnas.0701819104). This study used a precise environmental factor, day length, to probe gene function. When beach mice were exposed to 16 hours of light each day, oestrogen hormones decreased aggression, but when mice were exposed to only 8 hours of light each day, oestrogen hormones acted rapidly to increase aggression.
Vasudevan, N. & Pfaff, D. W. Membrane initiated actions of estrogens in neuroendocrinology: emerging principles. Endocr. Rev. 28, 1–19 (2006).
Earley, R. L. & Dugatkin, L. A. Eavesdropping on visual cues in green swordtail (Xiphophorus helleri) fights: a case for networking. Proc. R. Soc. Lond. B Biol. Sci. 269, 943–952 (2002).
Peake, T. M., Terry, A. M. R., McGregor, P. K. & Dabelsteen, T. Do great tits assess rivals by combining direct experience with information gathered by eavesdropping? Proc. R. Soc. Lond. B Biol. Sci. 269, 1925–1929 (2002).
Bernhardt, P. C., Dabbs, J. M. & Fielden, J. A. Testosterone changes during vicarious experiences of winning and losing among fans at sporting events. Physiol. Behav. 65, 59–62 (1998).
Mathiak, K. & Weber, R. Toward brain correlates of natural behavior: fMRI during violent video games. Hum. Brain Mapp. 27, 948–956 (2006).
Mathews, V. P. et al. Media violence exposure and frontal lobe activation measured by functional magnetic resonance imaging in aggressive and nonaggressive adolescents. J. Comput. Assist. Tomogr. 29, 287–292 (2005).
Cherek, D. R., Tcheremissine, O. V. & Lane, S. D. in Biology of Aggression (ed. Nelson, R. J.) 424–446 (Oxford Univ. Press, New York, 2006).
Darwin, C. The Expression of the Emotions in Man and Animals. (John Murray, London, 1872).
Berridge, K. C. in Handbook of Affective Sciences (eds Davidson, R. J., Scherer, K. R. & Goldsmith, H. H.) 25–51 (Oxford Univ. Press, New York, 2003).
Haller, J., Mikics, E., Halasz, J. & Toth, M. Mechanisms differentiating normal from abnormal aggression: glucocorticoids and serotonin. Eur. J. Pharmacol. 526, 89–100 (2005). This paper outlines a framework for classifying and studying aggressive behaviour in humans. The authors emphasize that aggression can occur in states of both hyperarousal and hypoarousal, and that different mechanisms are expected to be important in each affective context.
Simon, N. G. & Lu, S. in Biology of Aggression (ed. Nelson, R. J.) 211–230 (Oxford Univ. Press, New York, 2005).
Albert, D. J., Walsh, M. L. & Jonik, R. H. Aggression in humans: what is its biological foundation? Neurosci. Biobehav. Rev. 17, 405–425 (1993).
Wingfield, J. C., Hegner, R. E., Dufty, A. M. Jr & Ball, G. F. The 'challenge hypothesis': theoretical implications for patterns of testosterone secretion, mating systems, and breeding strategies. Am. Nat. 136, 829–846 (1990).
Mazur, A. & Booth, A. Testosterone and dominance in men. Behav. Brain Sci. 21, 353–397 (1998).
Acknowledgements
The authors acknowledge support by grants from the US National Institutes of Health and the US National Science Foundation. We thank J. Gusfa, K. Navarra and three anonymous reviewers for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
Molecules that Affect Aggression (PDF 583 kb)
Related links
Related links
DATABASES
Entrez Gene
Medscape Drug Info
FURTHER INFORMATION
Glossary
- Intermittent explosive disorder
-
A disorder characterized by repeated episodes of aggressive, violent behaviour that is grossly out of proportion to the situation; thought to affect as many as 7.3% of adults in the United States.
- Neuroleptic drug
-
An antipsychotic drug that is used to treat various psychiatric disorders, including schizophrenia. Although these drugs have stabilizing effects on mood, a major drawback is that they have potent sedative effects.
- Tardive dyskinesia
-
A disorder characterized by twitching of the face and tongue and involuntary motor movements of the trunk and limbs.
- Immediate early gene
-
A gene that is expressed rapidly and transiently in response to various cellular stimuli. Several of these genes (for example, Fos and Egr1) are used by neuroscientists as indirect markers of neuronal activity because they are expressed when neurons fire action potentials.
- Vasopressin
-
A neuropeptide that is present, among other regions, in the anterior hypothalamus and is known to affect aggression.
- Piloerection
-
The erection of hair on the skin, used as a threatening display by many animals.
- Lifetime History of Aggression (LHA) scale
-
An interview-based scale that is used by mental health workers to assess general aggressive tendencies in humans. Interviews can be supplemented with other sources such as clinical records.
- Selective serotonin-reuptake inhibitors
-
A class of antidepressants that inhibit the reuptake of serotonin (5-HT) by neural cells, thereby increasing the amount of 5-HT in the synapse.
- Borderline personality disorder
-
A disorder that is characterized by instability in moods, interpersonal relationships, self-image and behaviour; thought to affect approximately 2% of adults.
- Heteroreceptor
-
A receptor that modulates the synthesis and/or release of neurotransmitters other than its own ligand.
- Glutamic acid decarboxylase
-
An enzyme that produces GABA (γ-aminobutyric acid) from l-glutamic acid in an irreversible reaction.
- Gene chip
-
A method for analysing the expression of numerous genes simultaneously.
- Hormone response element
-
Sequences of DNA in promoter regions that are bound by hormone receptors. Binding of the receptor complex promotes transcription.
- Resident–intruder test
-
An aggression test in which an intruder is introduced into a resident's home cage. Residents are typically more aggressive because they are familiar with the environment and are defending their home territory.
Rights and permissions
About this article
Cite this article
Nelson, R., Trainor, B. Neural mechanisms of aggression. Nat Rev Neurosci 8, 536–546 (2007). https://doi.org/10.1038/nrn2174
Issue Date:
DOI: https://doi.org/10.1038/nrn2174
This article is cited by
-
Long-term consumption of rooibos herbal tea affects hypothalamic neurotransmission and social behavior of adult Sprague–Dawley male rats
European Food Research and Technology (2024)
-
Relationship between chronotypes and aggression in adolescents: a cross-sectional study
BMC Psychiatry (2023)
-
Dopamine transporter blockade during adolescence increases adult dopamine function, impulsivity, and aggression
Molecular Psychiatry (2023)
-
The basolateral amygdala to lateral septum circuit is critical for regulating social novelty in mice
Neuropsychopharmacology (2023)
-
A hypothalamic pathway that suppresses aggression toward superior opponents
Nature Neuroscience (2023)