Abstract
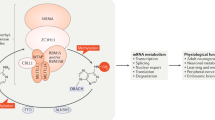
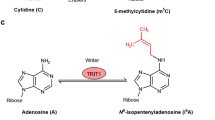
In this Perspective, we expand the notion of temporal regulation of RNA in the brain and propose that the qualitative nature of RNA and its metabolism, together with RNA abundance, are essential for the molecular mechanisms underlying experience-dependent plasticity. We discuss emerging concepts in the newly burgeoning field of epitranscriptomics, which are predicted to be heavily involved in cognitive function. These include activity-induced RNA modifications, RNA editing, dynamic changes in the secondary structure of RNA, and RNA localization. Each is described with an emphasis on its role in regulating the function of both protein-coding genes, as well as various noncoding regulatory RNAs, and how each might influence learning and memory.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Alberini, C.M. & Kandel, E.R. The regulation of transcription in memory consolidation. Cold Spring Harb. Perspect. Biol. 7, a021741 (2014).
McGaugh, J.L. Memory–a century of consolidation. Science 287, 248–251 (2000).
Sweatt, J.D. Neural plasticity & behavior–sixty years of conceptual advances. J. Neurochem. 2016, 14 (2016).
Rabani, M. et al. Metabolic labeling of RNA uncovers principles of RNA production and degradation dynamics in mammalian cells. Nat. Biotechnol. 29, 436–442 (2011).
Vogel, C. & Marcotte, E.M. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat. Rev. Genet. 13, 227–232 (2012).
Schwanhäusser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Cheng, Z. et al. Differential dynamics of the mammalian mRNA and protein expression response to misfolding stress. Mol. Syst. Biol. 12, 855 (2016).
Jovanovic, M. et al. Immunogenetics. Dynamic profiling of the protein life cycle in response to pathogens. Science 347, 1259038 (2015).
Li, J.J. et al. System wide analyses have underestimated protein abundances and the importance of transcription in mammals. PeerJ https://peerj.com/articles/270/ (2014).
Baker-Andresen, D., Ratnu, V.S. & Bredy, T.W. Dynamic DNA methylation: a prime candidate for genomic metaplasticity and behavioral adaptation. Trends Neurosci. 36, 3–13 (2013).
Lopez-Atalaya, J.P. & Barco, A. Can changes in histone acetylation contribute to memory formation? Trends Genet. 12, 529–539 (2014).
Fu, Y., Dominissini, D., Rechavi, G. & He, C. Gene expression regulation mediated through reversible m6A RNA methylation. Nat. Rev. Genet. 15, 293–306 (2014).
Dominissini, D. et al. The dynamic N(1)-methyladenosine methylome in eukaryotic messenger RNA. Nature 530, 441–446 (2016).
Carlile, T.M. et al. Pseudouridine profiling reveals regulated mRNA pseudouridylation in yeast and human cells. Nature 515, 143–146 (2014).
Liu, N. et al. N(6)-methyladenosine-dependent RNA structural switches regulate RNA-protein interactions. Nature 518, 560–564 (2015).
Behm, M. & Öhman, M. RNA editing: a contributor to neuronal dynamics in the mammalian brain. Trends Genet. 32, 165–175 (2016).
Liu, N. & Pan, T. N6-methyladenosine–encoded epitranscriptomics. Nat. Struct. Mol. Biol. 23, 98–102 (2016).
Machnicka, M.A. et al. MODOMICS: a database of RNA modification pathways—2013 update. Nucleic Acids Res. 41, D262–D267 (2013).
Saletore, Y. et al. The birth of the epitranscriptome: deciphering the function of RNA modifications. Genome Biol. 13, 175 (2012).
Liu, K. et al. Structural and functional characterization of the proteins responsible for N6-methyladenosine modification and recognition. Curr. Protein Pept. Sci. (2015).
Spitale, R.C. et al. Structural imprints in vivo decode RNA regulatory mechanisms. Nature 519, 486–490 (2015).
Zhou, J. et al. Dynamic m(6)A mRNA methylation directs translational control of heat shock response. Nature 526, 591–594 (2015).
Frye, M., Jaffrey, S.R., & Pan, T., Rechavi G. & Suzuki, T. RNA modifications: what have we learned and where are we headed? Nat. Rev. Genet. 6, 365–372 (2016).
Roundtree, I.A. & He, C. RNA epigenetics--chemical messages for posttranscriptional gene regulation. Curr. Opin. Chem. Biol. 30, 46–51 (2016).
Dominissini, D. et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature 485, 201–206 (2012).
Meyer, K.D. et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell 149, 1635–1646 (2012).
Meyer, K.D. & Jaffrey, S.R. The dynamic epitranscriptome: N6-methyladenosine and gene expression control. Nat. Rev. Mol. Cell Biol. 15, 313–326 (2014).
Widagdo, J. et al. Experience-dependent accumulation of N6-methyladenosine in the prefrontal cortex is associated with memory processes in mice. J. Neurosci. 36, 6771–6777 (2016).
Hess, M.E. et al. The fat mass and obesity associated gene (Fto) regulates activity of the dopaminergic midbrain circuitry. Nat. Neurosci. 16, 1042–1048 (2013).
Alarcón, C.R., Lee, H., Goodarzi, H., Halberg, N. & Tavazoie, S.F. N6-methyladenosine marks primary microRNAs for processing. Nature 519, 482–485 (2015).
Wang, X. et al. N(6)-methyladenosine modulates messenger RNA translation efficiency. Cell 161, 1388–1399 (2015).
Siciliano, V. et al. MiRNAs confer phenotypic robustness to gene networks by suppressing biological noise. Nat. Commun. 4, 2364 (2013).
Ule, J., Jensen, K., Mele, A. & Darnell, R.B. CLIP: a method for identifying protein-RNA interaction sites in living cells. Methods 37, 376–386 (2005).
Ofengand, J. Ribosomal RNA pseudouridines and pseudouridine synthases. FEBS Lett. 514, 17–25 (2002).
Roovers, M. et al. Formation of the conserved pseudouridine at position 55 in archaeal tRNA. Nucleic Acids Res. 34, 4293–4301 (2006).
Li, X. et al. Chemical pulldown reveals dynamic pseudouridylation of the mammalian transcriptome. Nat. Chem. Biol. 8, 592–597 (2015).
Wu, G., Huang, C. & Yu, Y.T. Pseudouridine in mRNA: Incorporation, Detection, and Recoding. Methods Enzymol. 560, 187–217 (2015).
Preumont, A., Snoussi, K., Stroobant, V., Collet, J.F. & Van Schaftingen, E. Molecular identification of pseudouridine-metabolizing enzymes. J. Biol. Chem. 283, 25238–25246 (2008).
Charette, M. & Gray, M.W. Pseudouridine in RNA: what, where, how, and why. IUBMB Life 49, 341–351 (2000).
Neumann, J.M., Bernassau, J.M., Guéron, M. & Tran-Dinh, S. Comparative conformations of uridine and pseudouridine and their derivatives. Eur. J. Biochem. 108, 457–463 (1980).
Arnez, J.G. & Steitz, T.A. Crystal structure of unmodified tRNA(Gln) complexed with glutaminyl-tRNA synthetase and ATP suggests a possible role for pseudo-uridines in stabilization of RNA structure. Biochemistry 33, 7560–7567 (1994).
Davis, D.R. Stabilization of RNA stacking by pseudouridine. Nucleic Acids Res. 23, 5020–5026 (1995).
Yu, Y.T. & Meier, U.T. RNA-guided isomerization of uridine to pseudouridine–pseudouridylation. RNA Biol. 11, 1483–1494 (2014).
Ge, J. & Yu, Y.T. RNA pseudouridylation: new insights into an old modification. Trends Biochem. Sci. 38, 210–218 (2013).
Garcia, G.A. & Kittendorf, J.D. Transglycosylation: a mechanism for RNA modification (and editing?). Bioorg. Chem. 33, 229–251 (2005).
Li, J.B. & Church, G.M. Deciphering the functions and regulation of brain-enriched A-to-I RNA editing. Nat. Neurosci. 16, 1518–1522 (2013).
Kim, D.D. et al. Widespread RNA editing of embedded alu elements in the human transcriptome. Genome Res. 14, 1719–1725 (2004).
Ramaswami, G. et al. Accurate identification of human Alu and non-Alu RNA editing sites. Nat. Methods 9, 579–581 (2012).
Bazak, L. et al. A-to-I RNA editing occurs at over a hundred million genomic sites, located in a majority of human genes. Genome Res. 24, 365–376 (2014).
Barry, G. & Mattick, J.S. The role of regulatory RNA in cognitive evolution. Trends Cogn. Sci. 10, 497–503 (2012).
Paupard, M.-C., O'Connell, M.A., Gerber, A.P. & Zukin, R.S. Patterns of developmental expression of the RNA editing enzyme rADAR2. Neuroscience 95, 869–879 (2000).
Sansam, C.L., Wells, K.S. & Emeson, R.B. Modulation of RNA editing by functional nucleolar sequestration of ADAR2. Proc. Natl. Acad. Sci. USA 100, 14018–14023 (2003).
Cattenoz, P.B., Taft, R.J., Westhof, E. & Mattick, J.S. Transcriptome-wide identification of A > I RNA editing sites by inosine specific cleavage. RNA 19, 257–270 (2013).
Franchini, D.M. et al. Processive DNA demethylation via DNA deaminase-induced lesion resolution. PLoS One 9, e97754 (2014).
Wan, Y., Kertesz, M., Spitale, R.C., Segal, E. & Chang, H.Y. Understanding the transcriptome through RNA structure. Nat. Rev. Genet. 12, 641–655 (2011).
Mortimer, S.A., Kidwell, M.A. & Doudna, J.A. Insights into RNA structure and function from genome-wide studies. Nat. Rev. Genet. 15, 469–479 (2014).
Bernard, D. et al. A long nuclear-retained non-coding RNA regulates synaptogenesis by modulating gene expression. EMBO J. 29, 3082–3093 (2010).
Zhang, B. et al. The lncRNA Malat1 is dispensable for mouse development but its transcription plays a cis-regulatory role in the adult. Cell Rep. 2, 111–123 (2012).
Barry, G. et al. The long non-coding RNA Gomafu is acutely regulated in response to neuronal activation and involved in schizophrenia-associated alternative splicing. Mol. Psychiatry 19, 486–494 (2014).
Spadaro, P.A. et al. Long noncoding RNA-directed epigenetic regulation of gene expression is associated with anxiety-like behavior in mice. Biol. Psychiatry 78, 848–859 (2015).
Helm, M. Post-transcriptional nucleotide modification and alternative folding of RNA. Nucleic Acids Res. 34, 721–733 (2006).
Fukuchi, M. & Tsuda, M. Involvement of the 3′-untranslated region of the brain-derived neurotrophic factor gene in activity-dependent mRNA stabilization. J. Neurochem. 115, 1222–1233 (2010).
Subramanian, M. et al. G-quadruplex RNA structure as a signal for neurite mRNA targeting. EMBO Rep. 12, 697–704 (2011).
Kar, A. et al. RNA helicase p68 (DDX5) regulates tau exon 10 splicing by modulating a stem-loop structure at the 5′ splice site. Mol. Cell. Biol. 31, 1812–1821 (2011).
Koeris, M., Funke, L., Shrestha, J., Rich, A. & Maas, S. Modulation of ADAR1 editing activity by Z-RNA in vitro. Nucleic Acids Res. 33, 5362–5370 (2005).
Ding, Y. et al. In vivo genome-wide profiling of RNA secondary structure reveals novel regulatory features. Nature 505, 696–700 (2014).
Pop, C. et al. Causal signals between codon bias, mRNA structure, and the efficiency of translation and elongation. Mol. Syst. Biol. 10, 770 (2014).
Rouskin, S., Zubradt, M., Washietl, S., Kellis, M. & Weissman, J.S. Genome-wide probing of RNA structure reveals active unfolding of mRNA structures in vivo. Nature 505, 701–705 (2014).
Kubota, M., Tran, C. & Spitale, R.C. Progress and challenges for chemical probing of RNA structure inside living cells. Nat. Chem. Biol. 11, 933–941 (2015).
Lipshitz, H.D. & Smibert, C.A. Mechanisms of RNA localization and translational regulation. Curr. Opin. Genet. Dev. 10, 476–488 (2000).
Willingham, A.T. et al. A strategy for probing the function of noncoding RNAs finds a repressor of NFAT. Science 309, 1570–1573 (2005).
Gong, C. & Maquat, L.E. lncRNAs transactivate STAU1-mediated mRNA decay by duplexing with 3′ UTRs via Alu elements. Nature 470, 284–288 (2011).
Ingolia, N.T. Genome-wide translational profiling by ribosome footprinting. Methods Enzymol. 470, 119–142 (2010).
Jan, C.H., Williams, C.C. & Weissman, J.S. Principles of ER cotranslational translocation revealed by proximity-specific ribosome profiling. Science 346, 1257521 (2014).
Williams, C.C., Jan, C.H. & Weissman, J.S. Targeting and plasticity of mitochondrial proteins revealed by proximity-specific ribosome profiling. Science 346, 748–751 (2014).
Dominguez, A.A., Lim, W.A. & Qi, L.S. Beyond editing: repurposing CRISPR-Cas9 for precision genome regulation and interrogation. Nat. Rev. Mol. Cell Biol. 17, 5–15 (2016).
Nelles, D.A. et al. Programmable RNA tracking in live cells with CRISPR/Cas9. Cell 165, 488–496 (2016).
Abudayyeh, O.O. et al. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science http://dx.doi.org/10.1126/science.aaf5573 (2016).
Mili, S. & Steitz, J.A. Evidence for reassociation of RNA-binding proteins after cell lysis: implications for the interpretation of immunoprecipitation analyses. RNA 10, 1692–1694 (2004).
Riley, K.J., Yario, T.A. & Steitz, J.A. Association of Argonaute proteins and microRNAs can occur after cell lysis. RNA 18, 1581–1585 (2012).
Hung, V. et al. Proteomic mapping of the human mitochondrial intermembrane space in live cells via ratiometric APEX tagging. Mol. Cell 55, 332–341 (2014).
Lee, S.Y. et al. APEX fingerprinting reveals the subcellular localization of proteins of interest. Cell Rep. 15, 1837–1847 (2016).
Lake, B.B. et al. Neuronal subtypes and diversity revealed by single-nucleus RNA sequencing of the human brain. Science 352, 1586–1590 (2016).
Morisaki, T. et al. Real-time quantification of single RNA translation dynamics in living cells. Science 352, 1425–1429 (2016).
Acknowledgements
The authors thank R. Tweedale for editing the manuscript and gratefully acknowledge grant support from the NIH (5R01MH105398-T.W.B.), NIH (1R01MH109588-01-T.W.B. and R.C.S.), NIGMS (1DP2GM119164-01-R.C.S.) and the NHMRC (APP1033127-T.W.B.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nainar, S., Marshall, P., Tyler, C. et al. Evolving insights into RNA modifications and their functional diversity in the brain. Nat Neurosci 19, 1292–1298 (2016). https://doi.org/10.1038/nn.4378
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4378
This article is cited by
-
The effects of N6-methyladenosine RNA methylation on the nervous system
Molecular and Cellular Biochemistry (2023)
-
Underexplored reciprocity between genome-wide methylation status and long non-coding RNA expression reflected in breast cancer research: potential impacts for the disease management in the framework of 3P medicine
EPMA Journal (2023)
-
Cellular and genetic drivers of RNA editing variation in the human brain
Nature Communications (2022)
-
The role m6A RNA methylation is CNS development and glioma pathogenesis
Molecular Brain (2021)
-
m6A modification in RNA: biogenesis, functions and roles in gliomas
Journal of Experimental & Clinical Cancer Research (2020)