Abstract
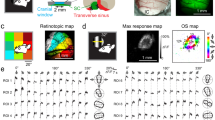
Orientation and spatial frequency tuning are highly salient properties of neurons in primary visual cortex (V1). The combined organization of these particular tuning properties in the cortical space will strongly shape the V1 population response to different visual inputs, yet it is poorly understood. In this study, we used two-photon imaging in macaque monkey V1 to demonstrate the three-dimensional cell-by-cell layout of both spatial frequency and orientation tuning. We first found that spatial frequency tuning was organized into highly structured maps that remained consistent across the depth of layer II/III, similarly to orientation tuning. Next, we found that orientation and spatial frequency maps were intimately related at the fine spatial scale observed with two-photon imaging. Not only did the map gradients tend notably toward orthogonality, but they also co-varied negatively from cell to cell at the spatial scale of cortical columns.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
03 December 2012
In the version of this article initially published, the scale bar length for Figure 1e was misstated as 500 μm. The correct length is 50 μm. The error has been corrected in the HTML and PDF versions of the article.
09 January 2013
In the version of this article initially published, the computation performed to yield the values on the x axis of Figure 6c was incorrectly defined in the text and on the axis label as the absolute difference between Aθ and Aϕ (mod 90°). The correct computation is 90° − || Aθ − Aϕ | − 90°|, which yields values near 0° for parallel gradients and values near 90° for perpendicular gradients. The error has been corrected in the HTML and PDF versions of the article.
11 January 2013
In the version of this article initially published, in the equation for Aθ on p. 5, the subscript to the variable f was given as an e. The correct character is θ. The error has been corrected in the HTML and PDF versions of the article.
References
Hubel, D.H. & Wiesel, T.N. Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J. Physiol. (Lond.) 160, 106–154 (1962).
Hubel, D.H. & Wiesel, T.N. Receptive fields and functional architecture of monkey striate cortex. J. Physiol. (Lond.) 195, 215–243 (1968).
Blasdel, G.G. & Salama, G. Voltage-sensitive dyes reveal a modular organization in monkey striate cortex. Nature 321, 579–585 (1986).
Ts'o, D.Y., Frostig, R., Lieke, E. & Grinvald, A. Functional organization of primate visual cortex revealed by high resolution optical imaging. Science 249, 417–420 (1990).
Horton, J.C. & Adams, D.L. The cortical column: a structure without a function. Phil. Trans. R. Soc. Lond. B 360, 837–862 (2005).
Hubel, D.H. & Wiesel, T.N. Functional architecture of macaque monkey visual cortex (Ferrier Lecture). Proc. R. Soc. Lond. B Biol. Sci. 198, 1–59 (1977).
Obermayer, K. & Blasdel, G.G. Geometry of orientation and ocular dominance columns in monkey striate cortex. J. Neurosci. 13, 4114–4129 (1993).
Bartfeld, E. & Grinvald, A. Relationships between orientation-preference pinwheels, cytochrome oxidase blobs, and ocular-dominance columns in primate striate cortex. Proc. Natl. Acad. Sci. USA 89, 11905–11909 (1992).
Crair, M.C., Ruthazer, E.S., Gillespie, D.C. & Stryker, M.P. Ocular dominance peaks at pinwheel center singularities of the orientation map in cat visual cortex. J. Neurophysiol. 77, 3381–3385 (1997).
Blasdel, G. & Campbell, D. Functional retinotopy of monkey visual cortex. J. Neurosci. 21, 8286–8301 (2001).
Hübener, M., Shoham, D., Grinvald, A. & Bonhoeffer, T. Spatial relationships among three columnar systems in cat area 17. J. Neurosci. 17, 9270–9284 (1997).
Issa, N.P., Trepel, C. & Stryker, M.P. Spatial frequency maps in cat visual cortex. J. Neurosci. 20, 8504–8514 (2000).
Yu, H., Farley, B.J., Jin, D.Z. & Sur, M. The coordinated mapping of visual space and response features in visual cortex. Neuron 47, 267–280 (2005).
Sirovich, L. & Uglesich, R. The organization of orientation and spatial frequency in primary visual cortex. Proc. Natl. Acad. Sci. USA 101, 16941–16946 (2004).
Kim, D.S., Matsuda, Y., Ohki, K., Ajima, A. & Tanaka, S. Geometrical and topological relationships between multiple functional maps in cat primary visual cortex. Neuroreport 10, 2515–2522 (1999).
Edwards, D.P., Purpura, K.P. & Kaplan, E. Contrast sensitivity and spatial frequency response of primate cortical neurons in and around the cytochrome oxidase blobs. Vision Res. 35, 1501–1523 (1995).
Born, R.T. & Tootell, R.B. Spatial frequency tuning of single units in macaque supragranular striate cortex. Proc. Natl. Acad. Sci. USA 88, 7066–7070 (1991).
Silverman, M.S., Grosof, D.H., De Valois, R.L. & Elfar, S.D. Spatial-frequency organization in primate striate cortex. Proc. Natl. Acad. Sci. USA 86, 711–715 (1989).
Tootell, R.B., Silverman, M.S., Hamilton, S.L., Switkes, E. & De Valois, R.L. Functional anatomy of macaque striate cortex. V. Spatial frequency. J. Neurosci. 8, 1610–1624 (1988).
Horton, J.C. Cytochrome oxidase patches: a new cytoarchitectonic feature of monkey visual cortex. Phil. Trans. R. Soc. Lond. B 304, 199–253 (1984).
Stosiek, C., Garaschuk, O., Holthoff, K. & Konnerth, A. In vivo two-photon calcium imaging of neuronal networks. Proc. Natl. Acad. Sci. USA 100, 7319–7324 (2003).
Ohki, K. et al. Highly ordered arrangement of single neurons in orientation pinwheels. Nature 442, 925–928 (2006).
Ringach, D.L., Sapiro, G. & Shapley, R. A subspace reverse-correlation technique for the study of visual neurons. Vision Res. 37, 2455–2464 (1997).
Nauhaus, I., Nielsen, K.J. & Callaway, E.M. Nonlinearity of two-photon Ca2+ imaging yields distorted measurements of tuning for V1 neuronal populations. J. Neurophysiol. 107, 923–936 (2012).
Maffei, L. & Fiorentini, A. Spatial frequency rows in the striate visual cortex. Vision Res. 17, 257–264 (1977).
Berardi, N., Bisti, S., Cattaneo, A., Fiorentini, A. & Maffei, L. Correlation between the preferred orientation and spatial frequency of neurones in visual areas 17 and 18 of the cat. J. Physiol. (Lond.) 323, 603–618 (1982).
Tootell, R.B., Silverman, M.S. & De Valois, R.L. Spatial frequency columns in primary visual cortex. Science 214, 813–815 (1981).
Tolhurst, D.J. & Thompson, I. Organization of neurones preferring similar spatial frequencies in cat striate cortex. Exp. Brain Res. 48, 217–227 (1982).
DeAngelis, G.C., Ghose, G.M., Ohzawa, I. & Freeman, R.D. Functional micro-organization of primary visual cortex: receptive field analysis of nearby neurons. J. Neurosci. 19, 4046–4064 (1999).
Molotchnikoff, S., Gillet, P.-C., Shumikhina, S. & Bouchard, M. Spatial frequency characteristics of nearby neurons in cats' visual cortex. Neurosci. Lett. 418, 242–247 (2007).
Shoham, D., Huebener, M., Schulze, S., Grinvald, A. & Bonhoeffer, T. Spatio-temporal frequency domains and their relation to cytochrome oxidase staining in cat visual cortex. Nature 385, 529–33 (1997).
Everson, R.M. et al. Representation of spatial frequency and orientation in the visual cortex. Proc. Natl. Acad. Sci. USA 95, 8334–8338 (1998).
Xu, X., Anderson, T.J. & Casagrande, V.A. How do functional maps in primary visual cortex vary with eccentricity? J. Comp. Neurol. 501, 741–755 (2007).
Gilbert, C.D. Microcircuitry of the visual cortex. Annu. Rev. Neurosci. 6, 217–247 (1983).
Martin, K.A.C. Neuronal circuits in cat striate cortex. in Cerebral Cortex (eds. Jones, E.G. & Peters, A.) 2:241–2:284 (Plenum, New York, 1984).
Callaway, E.M. Local circuits in primary visual cortex of the macaque monkey. Annu. Rev. Neurosci. 21, 47–74 (1998).
Nassi, J.J. & Callaway, E.M. Parallel processing strategies of the primate visual system. Nat. Rev. Neurosci. 10, 360–372 (2009).
Boyd, J.D. & Matsubara, J.A. Laminar and columnar patterns of geniculocortical projections in the cat: relationship to cytochrome oxidase. J. Comp. Neurol. 365, 659–682 (1996).
Swindale, N.V., Shoham, D., Grinvald, A., Bonhoeffer, T. & Hübener, M. Visual cortex maps are optimized for uniform coverage. Nat. Neurosci. 3, 822–826 (2000).
Issa, N.P., Trachtenberg, J.T., Chapman, B., Zahs, K.R. & Stryker, M.P. The critical period for ocular dominance plasticity in the ferret's visual cortex. J. Neurosci. 19, 6965–6978 (1999).
White, L.E., Bosking, W. & Fitzpatrick, D. Consistent mapping of orientation preference across irregular functional domains in ferret visual cortex. Vis. Neurosci. 18, 65–76 (2001).
Durbin, R. & Mitchison, G. A dimension reduction framework for understanding cortical maps. Nature 343, 644–647 (1990).
Koulakov, A.A. & Chklovskii, D. Orientation preference patterns in mammalian visual cortex: a wire length minimization approach. Neuron 29, 519–527 (2001).
Swindale, N.V. The development of topography in the visual cortex: a review of models. Network 7, 161–247 (1996).
Ferster, D. & Miller, K. Neural mechanisms of orientation selectivity in the visual cortex. Annu. Rev. Neurosci. 23, 441–471 (2000).
McLaughlin, D., Shapley, R. & Shelley, M. Large-scale modeling of the primary visual cortex: influence of cortical architecture upon neuronal response. J. Physiol. Paris 97, 237–252 (2003).
Paik, S.-B. & Ringach, D.L. Retinal origin of orientation maps in visual cortex. Nat. Neurosci. 14, 919–925 (2011).
Kaschube, M. et al. Universality in the evolution of orientation columns in the visual cortex. Science 330, 1113–1116 (2010).
Li, Y., Van Hooser, S.D., Mazurek, M., White, L.E. & Fitzpatrick, D. Experience with moving visual stimuli drives the early development of cortical direction selectivity. Nature 456, 952–956 (2008).
Kara, P. & Boyd, J.D. A micro-architecture for binocular disparity and ocular dominance in visual cortex. Nature 458, 627–631 (2009).
Nimmerjahn, A., Kirchhoff, F., Kerr, J.N.D. & Helmchen, F. Sulforhodamine 101 as a specific marker of astroglia in the neocortex in vivo. Nat. Methods 1, 31–37 (2004).
Pologruto, T.A., Sabatini, B.L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003).
Brainard, D.H. The psychophysics toolbox. Spat. Vis. 10, 433–436 (1997).
Pelli, D.G. The VideoToolbox software for visual psychophysics: transforming numbers to movies. Spat. Vis. 10, 437–442 (1997).
Acknowledgements
We are grateful to S. Chatterjee, K. Ohki and C. Reid for preliminary designs of the imaging chamber and for graciously helping us to get started with monkey two-photon imaging. We also thank D. Ringach for comments on an earlier version of the manuscript. Finally, we thank M. De La Parra for technical assistance with the experiments. This work was supported by US National Eye Institute grants EY-010742 to E.M.C., EY-019821 to I.N and MH093567 to A.A.D.
Author information
Authors and Affiliations
Contributions
I.N., K.J.N. and E.M.C. designed the research. I.N., K.J.N., A.A.D. and E.M.C. performed experiments. I.N. analyzed the data. I.N., K.J.N. and E.M.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 860 kb)
Supplementary Video 1
The acquired images during a flashed grating stimulus, played at 2x the actual speed, are shown on the left. The movie begins about 8 sec after the start of the trial, as depicted by the "progress bar" along the x-axis in each of the two panels on the right. The stimulus begins 2 s after the start of the timecourse, and lasts for 60 s. The right panels also contain the measured (black) and predicted (blue/red) response of the two circled neurons in the movie. The red and blue circles move with the measured location of each neuron as determined by the movement correction algorithm. The dominant movement in this trial is coupled to the animal's breathing. The heartbeat can be seen as well, but is more subtle. This difference between the two motion sources was typical, although this is not always the case, as demonstrated by the trial from Supp. Fig. 6b. (AVI 34564 kb)
Rights and permissions
About this article
Cite this article
Nauhaus, I., Nielsen, K., Disney, A. et al. Orthogonal micro-organization of orientation and spatial frequency in primate primary visual cortex. Nat Neurosci 15, 1683–1690 (2012). https://doi.org/10.1038/nn.3255
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3255
This article is cited by
-
Juvenile depletion of microglia reduces orientation but not high spatial frequency selectivity in mouse V1
Scientific Reports (2022)
-
A theory of cortical map formation in the visual brain
Nature Communications (2022)
-
Processing of visual statistics of naturalistic videos in macaque visual areas V1 and V4
Brain Structure and Function (2022)
-
Microendoscopic calcium imaging of the primary visual cortex of behaving macaques
Scientific Reports (2021)
-
Occipital cortex is modulated by transsaccadic changes in spatial frequency: an fMRI study
Scientific Reports (2021)