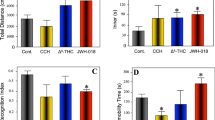
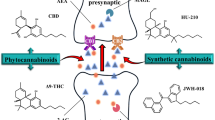
Abstract
Studies of the endocannabinoid system in the CNS have been mostly focused on endocannabinoid receptors and inactivating mechanisms. Until recently, very little was known about the role of biosynthetic enzymes in endocannabinoid signaling. New data from the recent development of pharmacological and genetic tools for the study of these enzymes point to their fundamental role in determining where and when endocannabinoids function, and raise the possibility of new intriguing and previously unsuspected concepts in the general strategy of endocannabinoid signaling. However, even with these new tools, the cross-talk between anandamide and 2-arachidonoylglycerol biosynthesis makes it difficult to dissect one from the other, and data will need to be interpreted with this in mind.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Devane, W.A. et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258, 1946–1949 (1992).
Di Marzo, V. Targeting the endocannabinoid system: to enhance or reduce? Nat. Rev. Drug Discov. 7, 438–455 (2008).
Leung, D., Saghatelian, A., Simon, G.M. & Cravatt, B.F. Inactivation of N-acyl phosphatidylethanolamine phospholipase D reveals multiple mechanisms for the biosynthesis of endocannabinoids. Biochemistry 45, 4720–4726 (2006).
Simon, G.M. & Cravatt, B.F. Characterization of mice lacking candidate N-acyl ethanolamine biosynthetic enzymes provides evidence for multiple pathways that contribute to endocannabinoid production in vivo. Mol. Biosyst. 6, 1411–1418 (2010).
van der Stelt, M. et al. Anandamide acts as an intracellular messenger amplifying Ca2+ influx via TRPV1 channels. EMBO J. 24, 3026–3037 (2005).
Starowicz, K., Nigam, S. & Di Marzo, V. Biochemistry and pharmacology of endovanilloids. Pharmacol. Ther. 114, 13–33 (2007).
Nyilas, R. et al. Enzymatic machinery for endocannabinoid biosynthesis associated with calcium stores in glutamatergic axon terminals. J. Neurosci. 28, 1058–1063 (2008).
Egertová, M., Simon, G.M., Cravatt, B.F. & Elphick, M.R. Localization of N-acyl phosphatidylethanolamine phospholipase D (NAPE-PLD) expression in mouse brain: a new perspective on N-acylethanolamines as neural signaling molecules. J. Comp. Neurol. 506, 604–615 (2008).
Cristino, L. et al. Immunohistochemical localization of anabolic and catabolic enzymes for anandamide and other putative endovanilloids in the hippocampus and cerebellar cortex of the mouse brain. Neuroscience 151, 955–968 (2008).
Suárez, J. et al. Immunohistochemical description of the endogenous cannabinoid system in the rat cerebellum and functionally related nuclei. J. Comp. Neurol. 509, 400–421 (2008).
Cristino, L. et al. Immunohistochemical localization of cannabinoid type 1 and vanilloid transient receptor potential vanilloid type 1 receptors in the mouse brain. Neuroscience 139, 1405–1415 (2006).
Bisogno, T. et al. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol. 163, 463–468 (2003).
Katona, I. et al. Molecular composition of the endocannabinoid system at glutamatergic synapses. J. Neurosci. 26, 5628–5637 (2006).
Yoshida, T. et al. Localization of diacylglycerol lipase-α around postsynaptic spine suggests close proximity between production site of an endocannabinoid, 2-arachidonoyl-glycerol, and presynaptic cannabinoid CB1 receptor. J. Neurosci. 26, 4740–4751 (2006).
Nomura, S., Fukaya, M., Tsujioka, T., Wu, D. & Watanabe, M. Phospholipase Cβ3 is distributed in both somatodendritic and axonal compartments and localized around perisynapse and smooth endoplasmic reticulum in mouse Purkinje cell subsets. Eur. J. Neurosci. 25, 659–672 (2007).
Maejima, T. et al. Synaptically driven endocannabinoid release requires Ca2+-assisted metabotropic glutamate receptor subtype 1 to phospholipase Cβ4 signaling cascade in the cerebellum. J. Neurosci. 25, 6826–6835 (2005).
Fukaya, M. et al. Predominant expression of phospholipase Cbeta1 in telencephalic principal neurons and cerebellar interneurons, and its close association with related signaling molecules in somatodendritic neuronal elements. Eur. J. Neurosci. 28, 1744–1759 (2008).
Lafourcade, M. et al. Molecular components and functions of the endocannabinoid system in mouse prefrontal cortex. PLoS ONE 2, e709 (2007).
Roloff, A.M., Anderson, G.R., Martemyanov, K.A. & Thayer, S.A. Homer 1a gates the induction mechanism for endocannabinoid-mediated synaptic plasticity. J. Neurosci. 30, 3072–3081 (2010).
Marrs, W.R. et al. The serine hydrolase ABHD6 controls the accumulation and efficacy of 2-AG at cannabinoid receptors. Nat. Neurosci. 13, 951–957 (2010).
Marinelli, S. et al. The endocannabinoid 2-arachidonoylglycerol is responsible for the slow self-inhibition in neocortical interneurons. J. Neurosci. 28, 13532–13541 (2008).
Marinelli, S., Pacioni, S., Cannich, A., Marsicano, G. & Bacci, A. Self-modulation of neocortical pyramidal neurons by endocannabinoids. Nat. Neurosci. 12, 1488–1490 (2009).
Tanimura, A. et al. The endocannabinoid 2-arachidonoylglycerol produced by diacylglycerol lipase α mediates retrograde suppression of synaptic transmission. Neuron 65, 320–327 (2010).
Gao, Y. et al. Loss of retrograde endocannabinoid signaling and reduced adult neurogenesis in diacylglycerol lipase knock-out mice. J. Neurosci. 30, 2017–2024 (2010).
Nomura, D.K. et al. Monoacylglycerol lipase regulates 2-arachidonoylglycerol action and arachidonic acid levels. Bioorg. Med. Chem. Lett. 18, 5875–5878 (2008).
D'Asti, E. et al. Maternal dietary fat determines metabolic profile and the magnitude of endocannabinoid inhibition of the stress response in neonatal rat offspring. Endocrinology 151, 1685–1694 (2010).
Wettschureck, N. et al. Forebrain-specific inactivation of Gq/G11 family G proteins results in age-dependent epilepsy and impaired endocannabinoid formation. Mol. Cell. Biol. 26, 5888–5894 (2006).
Harkany, T. et al. The emerging functions of endocannabinoid signaling during CNS development. Trends Pharmacol. Sci. 28, 83–92 (2007).
Goncalves, M.B. et al. A diacylglycerol lipase-CB2 cannabinoid pathway regulates adult subventricular zone neurogenesis in an age-dependent manner. Mol. Cell. Neurosci. 38, 526–536 (2008).
Bisogno, T. et al. Development of the first potent and specific inhibitors of endocannabinoid biosynthesis. Biochim. Biophys. Acta 1761, 205–212 (2006).
Bisogno, T. et al. Synthesis and pharmacological activity of a potent inhibitor of the biosynthesis of the endocannabinoid 2-arachidonoylglycerol. ChemMedChem 4, 946–950 (2009).
Ortar, G. et al. Tetrahydrolipstatin analogues as modulators of endocannabinoid 2-arachidonoylglycerol metabolism. J. Med. Chem. 51, 6970–6979 (2008).
Straiker, A. et al. Monoacylglycerol lipase limits the duration of endocannabinoid-mediated depolarization-induced suppression of excitation in autaptic hippocampal neurons. Mol. Pharmacol. 76, 1220–1227 (2009).
Melis, M. et al. Prefrontal cortex stimulation induces 2-arachidonoyl-glycerol-mediated suppression of excitation in dopamine neurons. J. Neurosci. 24, 10707–10715 (2004).
Szabo, B. et al. Depolarization-induced retrograde synaptic inhibition in the mouse cerebellar cortex is mediated by 2-arachidonoylglycerol. J. Physiol. (Lond.) 577, 263–280 (2006).
Hashimotodani, Y., Ohno-Shosaku, T., Maejima, T., Fukami, K. & Kano, M. Pharmacological evidence for the involvement of diacylglycerol lipase in depolarization-induced endocannabinoid release. Neuropharmacology 54, 58–67 (2008).
Hashimotodani, Y., Ohno-Shosaku, T. & Kano, M. Presynaptic monoacylglycerol lipase activity determines basal endocannabinoid tone and terminates retrograde endocannabinoid signaling in the hippocampus. J. Neurosci. 27, 1211–1219 (2007).
Chevaleyre, V. & Castillo, P.E. Heterosynaptic LTD of hippocampal GABAergic synapses: a novel role of endocannabinoids in regulating excitability. Neuron 38, 461–472 (2003).
Edwards, D.A., Kim, J. & Alger, B.E. Multiple mechanisms of endocannabinoid response initiation in hippocampus. J. Neurophysiol. 95, 67–75 (2006).
Min, R. et al. Diacylglycerol lipase is not involved in depolarization-induced suppression of inhibition at unitary inhibitory connections in mouse hippocampus. J. Neurosci. 30, 2710–2715 (2010).
Edwards, D.A., Zhang, L. & Alger, B.E. Metaplastic control of the endocannabinoid system at inhibitory synapses in hippocampus. Proc. Natl. Acad. Sci. USA 105, 8142–8147 (2008).
Safo, P.K. & Regehr, W.G. Endocannabinoids control the induction of cerebellar LTD. Neuron 48, 647–659 (2005).
Oddi, S. et al. Evidence for the intracellular accumulation of anandamide in adiposomes. Cell. Mol. Life Sci. 65, 840–850 (2008).
Maccarrone, M. et al. Anandamide inhibits metabolism and physiological actions of 2-arachidonoylglycerol in the striatum. Nat. Neurosci. 11, 152–159 (2008).
Schlosburg, J.E. et al. Chronic monoacylglycerol lipase blockade causes functional antagonism of the endocannabinoid system. Nat. Neurosci. 13, 1113–1119 (2010).
Kim, J. & Alger, B.E. Reduction in endocannabinoid tone is a homeostatic mechanism for specific inhibitory synapses. Nat. Neurosci. 13, 592–600 (2010).
Gomez, O. et al. The constitutive production of the endocannabinoid 2-arachidonoylglycerol participates in oligodendrocyte differentiation. Glia 58, 1913–1927 (2010).
Maione, S. et al. Elevation of endocannabinoid levels in the ventrolateral periaqueductal grey through inhibition of fatty acid amide hydrolase affects descending nociceptive pathways via both cannabinoid receptor type 1 and transient receptor potential vanilloid type-1 receptors. J. Pharmacol. Exp. Ther. 316, 969–982 (2006).
Melis, M. et al. Protective activation of the endocannabinoid system during ischemia in dopamine neurons. Neurobiol. Dis. 24, 15–27 (2006).
Hoover, H.S., Blankman, J.L., Niessen, S. & Cravatt, B.F. Selectivity of inhibitors of endocannabinoid biosynthesis evaluated by activity-based protein profiling. Bioorg. Med. Chem. Lett. 18, 5838–5841 (2008).
Acknowledgements
The author thanks T. Bisogno and S. Petrosino for their help. Support from the US National Institutes of Health (grant DA-009789) is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 (PDF 136 kb)
Rights and permissions
About this article
Cite this article
Di Marzo, V. Endocannabinoid signaling in the brain: biosynthetic mechanisms in the limelight. Nat Neurosci 14, 9–15 (2011). https://doi.org/10.1038/nn.2720
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2720
This article is cited by
-
CB1 antagonism increases excitatory synaptogenesis in a cortical spheroid model of fetal brain development
Scientific Reports (2021)
-
mGluR5-Mediated eCB Signaling in the Nucleus Accumbens Controls Vulnerability to Depressive-Like Behaviors and Pain After Chronic Social Defeat Stress
Molecular Neurobiology (2021)
-
Current state of evidence of cannabis utilization for treatment of autism spectrum disorders
BMC Psychiatry (2019)
-
The Role of the Endocannabinoid System and Genetic Variation in Adolescent Brain Development
Neuropsychopharmacology (2018)
-
Monoacylglycerol lipase inhibitors produce pro- or antidepressant responses via hippocampal CA1 GABAergic synapses
Molecular Psychiatry (2017)