Abstract
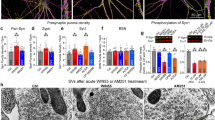
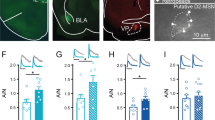
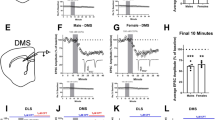
Synaptic modifications in the nucleus accumbens (NAc) are important for adaptive and pathological reward-dependent learning. Medium spiny neurons (MSNs), the major cell type in the NAc, participate in two parallel circuits that subserve distinct behavioral functions, yet little is known about differences in their electrophysiological and synaptic properties. Using bacterial artificial chromosome transgenic mice, we found that synaptic activation of group I metabotropic glutamate receptors in NAc MSNs in the indirect, but not direct, pathway led to the production of endocannabinoids, which activated presynaptic CB1 receptors to trigger endocannabinoid-mediated long-term depression (eCB-LTD) as well as postsynaptic transient receptor potential vanilloid 1 (TRPV1) channels to trigger a form of LTD resulting from endocytosis of AMPA receptors. These results reveal a previously unknown action of TRPV1 channels and indicate that the postsynaptic generation of endocannabinoids can modulate synaptic strength in a cell type–specific fashion by activating distinct pre- and postsynaptic targets.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Sesack, S.R. & Grace, A.A. Cortico-basal ganglia reward network: microcircuitry. Neuropsychopharmacology 35, 27–47 (2010).
Kalivas, P.W. The glutamate homeostasis hypothesis of addiction. Nat. Rev. Neurosci. 10, 561–572 (2009).
Kauer, J.A. & Malenka, R.C. Synaptic plasticity and addiction. Nat. Rev. Neurosci. 8, 844–858 (2007).
Conrad, K.L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121 (2008).
Kasanetz, F. et al. Transition to addiction is associated with a persistent impairment in synaptic plasticity. Science 328, 1709–1712 (2010).
Kreitzer, A.C. & Malenka, R.C. Striatal plasticity and basal ganglia circuit function. Neuron 60, 543–554 (2008).
Hikida, T., Kimura, K., Wada, N., Funabiki, K. & Nakanishi, S. Distinct roles of synaptic transmission in direct and indirect striatal pathways to reward and aversive behavior. Neuron 66, 896–907 (2010).
Kravitz, A.V. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626 (2010).
Gong, S. et al. A gene expression atlas of the central nervous system based on bacterial artificial chromosomes. Nature 425, 917–925 (2003).
Caterina, M.J. & Julius, D. The vanilloid receptor: a molecular gateway to the pain pathway. Annu. Rev. Neurosci. 24, 487–517 (2001).
Kauer, J.A. & Gibson, H.E. Hot flash: TRPV channels in the brain. Trends Neurosci. 32, 215–224 (2009).
Ramsey, I.S., Delling, M. & Clapham, D.E. An introduction to TRP channels. Annu. Rev. Physiol. 68, 619–647 (2006).
Zygmunt, P.M. et al. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature 400, 452–457 (1999).
Gibson, H.E., Edwards, J.G., Page, R.S., Van Hook, M.J. & Kauer, J.A. TRPV1 channels mediate long-term depression at synapses on hippocampal interneurons. Neuron 57, 746–759 (2008).
Maione, S. et al. TRPV1 channels control synaptic plasticity in the developing superior colliculus. J. Physiol. (Lond.) 587, 2521–2535 (2009).
Kreitzer, A.C. & Malenka, R.C. Endocannabinoid-mediated rescue of striatal LTD and motor deficits in Parkinson's disease models. Nature 445, 643–647 (2007).
Robbe, D., Kopf, M., Remaury, A., Bockaert, J. & Manzoni, O.J. Endogenous cannabinoids mediate long-term synaptic depression in the nucleus accumbens. Proc. Natl. Acad. Sci. USA 99, 8384–8388 (2002).
Heifets, B.D. & Castillo, P.E. Endocannabinoid signaling and long-term synaptic plasticity. Annu. Rev. Physiol. 71, 283–306 (2009).
Schoch, S. et al. Nature 415, 321–326 (2002).
Hoffman, A.F., Oz, M., Caulder, T. & Lupica, C.R. Functional tolerance and blockade of long-term depression at synapses in the nucleus accumbens after chronic cannabinoid exposure. J. Neurosci. 23, 4815–4820 (2003).
Micale, V. et al. Anxiolytic effects in mice of a dual blocker of fatty acid amide hydrolase and transient receptor potential vanilloid type-1 channels. Neuropsychopharmacology 34, 593–606 (2009).
Roberts, J.C., Davis, J.B. & Benham, C.D. [3H]Resiniferatoxin autoradiography in the CNS of wild-type and TRPV1 null mice defines TRPV1 (VR-1) protein distribution. Brain Res. 995, 176–183 (2004).
Starowicz, K., Cristino, L. & Di Marzo, V. TRPV1 receptors in the central nervous system: potential for previously unforeseen therapeutic applications. Curr. Pharm. Des. 14, 42–54 (2008).
Lüscher, C. et al. Role of AMPA receptor cycling in synaptic transmission and plasticity. Neuron 24, 649–658 (1999).
Fourgeaud, L. et al. A single in vivo exposure to cocaine abolishes endocannabinoid-mediated long-term depression in the nucleus accumbens. J. Neurosci. 24, 6939–6945 (2004).
Mato, S. et al. A single in-vivo exposure to Delta 9THC blocks endocannabinoid-mediated synaptic plasticity. Nat. Neurosci. 7, 585–586 (2004).
Malenka, R.C. & Bear, M.F. LTP and LTD: an embarrassment of riches. Neuron 44, 5–21 (2004).
Vezina, P. & Leyton, M. Conditioned cues and the expression of stimulant sensitization in animals and humans. Neuropharmacology 56 (Suppl 1): 160–168 (2009).
Wettschureck, N. et al. Forebrain-specific inactivation of Gq/G11 family G proteins results in age-dependent epilepsy and impaired endocannabinoid formation. Mol. Cell. Biol. 26, 5888–5894 (2006).
van der Stelt, M. et al. Anandamide acts as an intracellular messenger amplifying Ca2+ influx via TRPV1 channels. EMBO J. 24, 3026–3037 (2005).
Maccarrone, M. et al. Anandamide inhibits metabolism and physiological actions of 2-arachidonoylglycerol in the striatum. Nat. Neurosci. 11, 152–159 (2008).
Chavez, A.E., Chiu, C.Q. & Castillo, P.E. TRPV1 activation by endogenous anandamide triggers postsynaptic LTD in dentate gyrus. Nat. Neurosci. advanced online publication, doi:10.1038/nn.2684 (14 November 2010).
Marsch, R. et al. Reduced anxiety, conditioned fear, and hippocampal long-term potentiation in transient receptor potential vanilloid type 1 receptor–deficient mice. J. Neurosci. 27, 832–839 (2007).
de Lago, E., de Miguel, R., Lastres-Becker, I., Ramos, J.A. & Fernandez-Ruiz, J. Involvement of vanilloid-like receptors in the effects of anandamide on motor behavior and nigrostriatal dopaminergic activity: in vivo and in vitro evidence. Brain Res. 1007, 152–159 (2004).
Di Marzo, V. et al. Hypolocomotor effects in rats of capsaicin and two long chain capsaicin homologues. Eur. J. Pharmacol. 420, 123–131 (2001).
Lee, J., Di Marzo, V. & Brotchie, J.M. A role for vanilloid receptor 1 (TRPV1) and endocannabinnoid signaling in the regulation of spontaneous and L-DOPA induced locomotion in normal and reserpine-treated rats. Neuropharmacology 51, 557–565 (2006).
Tzavara, E.T. et al. Endocannabinoids activate transient receptor potential vanilloid 1 receptors to reduce hyperdopaminergia-related hyperactivity: therapeutic implications. Biol. Psychiatry 59, 508–515 (2006).
Szumlinski, K.K., Kalivas, P.W. & Worley, P.F. Homer proteins: implications for neuropsychiatric disorders. Curr. Opin. Neurobiol. 16, 251–257 (2006).
Thomas, M.J., Beurrier, C., Bonci, A. & Malenka, R.C. Long-term depression in the nucleus accumbens: a neural correlate of behavioral sensitization to cocaine. Nat. Neurosci. 4, 1217–1223 (2001).
Kourrich, S., Rothwell, P.E., Klug, J.R. & Thomas, M.J. Cocaine experience controls bidirectional synaptic plasticity in the nucleus accumbens. J. Neurosci. 27, 7921–7928 (2007).
Brebner, K. et al. Nucleus accumbens long-term depression and the expression of behavioral sensitization. Science 310, 1340–1343 (2005).
McFarland, K. & Kalivas, P.W. The circuitry mediating cocaine-induced reinstatement of drug-seeking behavior. J. Neurosci. 21, 8655–8663 (2001).
Tang, X.C., McFarland, K., Cagle, S. & Kalivas, P.W. Cocaine-induced reinstatement requires endogenous stimulation of mu-opioid receptors in the ventral pallidum. J. Neurosci. 25, 4512–4520 (2005).
Torregrossa, M.M., Tang, X.C. & Kalivas, P.W. The glutamatergic projection from the prefrontal cortex to the nucleus accumbens core is required for cocaine-induced decreases in ventral pallidal GABA. Neurosci. Lett. 438, 142–145 (2008).
Durieux, P.F. et al. D2R striatopallidal neurons inhibit both locomotor and drug reward processes. Nat. Neurosci. 12, 393–395 (2009).
Grueter, B.A. et al. Extracellular-signal regulated kinase 1-dependent metabotropic glutamate receptor 5–induced long-term depression in the bed nucleus of the stria terminalis is disrupted by cocaine administration. J. Neurosci. 26, 3210–3219 (2006).
Acknowledgements
We thank members of the Malenka lab for their comments. This work was supported by grants from the National Institute on Drug Abuse (DA009264, DA008227 to R.C.M.; 5F32DA023741-2 to B.A.G.).
Author information
Authors and Affiliations
Contributions
B.A.G., G.B. and R.C.M. designed the experiments, interpreted the results and wrote the paper. B.A.G. and G.B. performed all of the experiments and analyzed the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 (PDF 693 kb)
Rights and permissions
About this article
Cite this article
Grueter, B., Brasnjo, G. & Malenka, R. Postsynaptic TRPV1 triggers cell type–specific long-term depression in the nucleus accumbens. Nat Neurosci 13, 1519–1525 (2010). https://doi.org/10.1038/nn.2685
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2685
This article is cited by
-
Cocaine-induced loss of LTD and social impairments are restored by fatty acid amide hydrolase inhibition
Scientific Reports (2023)
-
Cocaine restricts nucleus accumbens feedforward drive through a monoamine-independent mechanism
Neuropsychopharmacology (2022)
-
Restoring glutamate homeostasis in the nucleus accumbens via endocannabinoid-mimetic drug prevents relapse to cocaine seeking behavior in rats
Neuropsychopharmacology (2021)
-
Kappa opioid receptor modulation of excitatory drive onto nucleus accumbens fast-spiking interneurons
Neuropsychopharmacology (2021)
-
Cannabinoid type 1 receptors in A2a neurons contribute to cocaine-environment association
Psychopharmacology (2021)