Abstract
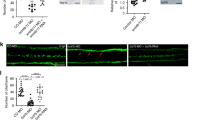
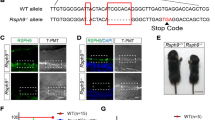
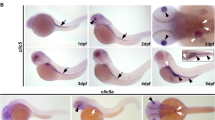
Ependymal cells form the epithelial lining of cerebral ventricles. Their apical surface is covered by cilia that beat in a coordinated fashion to facilitate circulation of the cerebrospinal fluid (CSF). The genetic factors that govern the development and function of ependymal cilia remain poorly understood. We found that the planar cell polarity cadherins Celsr2 and Celsr3 control these processes. In Celsr2-deficient mice, the development and planar organization of ependymal cilia are compromised, leading to defective CSF dynamics and hydrocephalus. In Celsr2 and Celsr3 double mutant ependyma, ciliogenesis is markedly impaired, resulting in lethal hydrocephalus. The membrane distribution of Vangl2 and Fzd3, two key planar cell polarity proteins, was disturbed in Celsr2 mutants, and even more so in Celsr2 and Celsr3 double mutants. Our findings suggest that planar cell polarity signaling is involved in ependymal cilia development and in the pathophysiology of hydrocephalus, with possible implications in other ciliopathies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
23 May 2010
In the version of this article initially published online, the subheading ‘Celsr2 mutant mice develop progressive hydrocephalus’ incorrectly appeared twice. In addition, asterisks noting statistical significance in Figure 6d were missing. These errors have been corrected for the print, PDF and HTML versions of the article.
References
Davenport, J.R. & Yoder, B.K. An incredible decade for the primary cilium: a look at a once-forgotten organelle. Am. J. Physiol. Renal Physiol. 289, F1159–F1169 (2005).
Marshall, W.F. & Kintner, C. Cilia orientation and the fluid mechanics of development. Curr. Opin. Cell Biol. 20, 48–52 (2008).
Salathe, M. Regulation of mammalian ciliary beating. Annu. Rev. Physiol. 69, 401–422 (2007).
Voronina, V.A. et al. Inactivation of Chibby affects function of motile airway cilia. J. Cell Biol. 185, 225–233 (2009).
Ibañez-Tallon, I. et al. Dysfunction of axonemal dynein heavy chain Mdnah5 inhibits ependymal flow and reveals a novel mechanism for hydrocephalus formation. Hum. Mol. Genet. 13, 2133–2141 (2004).
Banizs, B. et al. Dysfunctional cilia lead to altered ependyma and choroid plexus function, and result in the formation of hydrocephalus. Development 132, 5329–5339 (2005).
Lechtreck, K.F., Delmotte, P., Robinson, M.L., Sanderson, M.J. & Witman, G.B. Mutations in Hydin impair ciliary motility in mice. J. Cell Biol. 180, 633–643 (2008).
Davy, B.E. & Robinson, M.L. Congenital hydrocephalus in hy3 mice is caused by a frameshift mutation in Hydin, a large novel gene. Hum. Mol. Genet. 12, 1163–1170 (2003).
Simons, M. & Mlodzik, M. Planar cell polarity signaling: from fly development to human disease. Annu. Rev. Genet. 42, 517–540 (2008).
Strutt, D.I., Weber, U. & Mlodzik, M. The role of RhoA in tissue polarity and Frizzled signaling. Nature 387, 292–295 (1997).
Wang, Y. & Nathans, J. Tissue/planar cell polarity in vertebrates: new insights and new questions. Development 134, 647–658 (2007).
Kibar, Z. et al. Ltap, a mammalian homolog of Drosophila Strabismus/Van Gogh, is altered in the mouse neural tube mutant Loop-tail. Nat. Genet. 28, 251–255 (2001).
Curtin, J.A. et al. Mutation of Celsr1 disrupts planar polarity of inner ear hair cells and causes severe neural tube defects in the mouse. Curr. Biol. 13, 1129–1133 (2003).
Montcouquiol, M. et al. Asymmetric localization of Vangl2 and Fz3 indicate novel mechanisms for planar cell polarity in mammals. J. Neurosci. 26, 5265–5275 (2006).
Ravni, A., Qu, Y., Goffinet, A.M. & Tissir, F. Planar cell polarity cadherin Celsr1 regulates skin hair patterning in the mouse. J. Invest. Dermatol. 129, 2507–2509 (2009).
Guo, N., Hawkins, C. & Nathans, J. Frizzled6 controls hair patterning in mice. Proc. Natl. Acad. Sci. USA 101, 9277–9281 (2004).
Devenport, D. & Fuchs, E. Planar polarization in embryonic epidermis orchestrates global asymmetric morphogenesis of hair follicles. Nat. Cell Biol. 10, 1257–1268 (2008).
Shima, Y. et al. Opposing roles in neurite growth control by two seven-pass transmembrane cadherins. Nat. Neurosci. 10, 963–969 (2007).
Tissir, F., Bar, I., Jossin, Y. & Goffinet, A.M. Protocadherin Celsr3 is crucial in axonal tract development. Nat. Neurosci. 8, 451–457 (2005).
Wang, Y., Thekdi, N., Smallwood, P.M., Macke, J.P. & Nathans, J. Frizzled-3 is required for the development of major fiber tracts in the rostral CNS. J. Neurosci. 22, 8563–8573 (2002).
Park, T.J., Mitchell, B.J., Abitua, P.B., Kintner, C. & Wallingford, J.B. Dishevelled controls apical docking and planar polarization of basal bodies in ciliated epithelial cells. Nat. Genet. 40, 871–879 (2008).
Park, T.J., Haigo, S.L. & Wallingford, J.B. Ciliogenesis defects in embryos lacking inturned or fuzzy function are associated with failure of planar cell polarity and Hedgehog signaling. Nat. Genet. 38, 303–311 (2006).
Zhou, L. et al. Early forebrain wiring: genetic dissection using conditional Celsr3 mutant mice. Science 320, 946–949 (2008).
Shima, Y. et al. Differential expression of the seven-pass transmembrane cadherin genes Celsr1–3 and distribution of the Celsr2 protein during mouse development. Dev. Dyn. 223, 321–332 (2002).
Tissir, F., De-Backer, O., Goffinet, A.M. & Lambert de Rouvroit, C. Developmental expression profiles of Celsr (Flamingo) genes in the mouse. Mech. Dev. 112, 157–160 (2002).
Formstone, C.J. & Little, P.F. The flamingo-related mouse Celsr family (Celsr1–3) genes exhibit distinct patterns of expression during embryonic development. Mech. Dev. 109, 91–94 (2001).
Town, T. et al. The stumpy gene is required for mammalian ciliogenesis. Proc. Natl. Acad. Sci. USA 105, 2853–2858 (2008).
Mirzadeh, Z., Merkle, F.T., Soriano-Navarro, M. & Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Neural stem cells confer unique pinwheel architecture to the ventricular surface in neurogenic regions of the adult brain. Cell Stem Cell 3, 265–278 (2008).
Danilov, A.I. et al. Ultrastructural and antigenic properties of neural stem cells and their progeny in adult rat subventricular zone. Glia 57, 136–152 (2009).
Sawamoto, K. et al. New neurons follow the flow of cerebrospinal fluid in the adult brain. Science 311, 629–632 (2006).
Spassky, N. et al. Adult ependymal cells are postmitotic and are derived from radial glial cells during embryogenesis. J. Neurosci. 25, 10–18 (2005).
Boisvieux-Ulrich, E. & Sandoz, D. Determination of ciliary polarity precedes differentiation in the epithelial cells of quail oviduct. Biol. Cell 72, 3–14 (1991).
Dawe, H.R., Farr, H. & Gull, K. Centriole/basal body morphogenesis and migration during ciliogenesis in animal cells. J. Cell Sci. 120, 7–15 (2007).
Mitchell, B., Jacobs, R., Li, J., Chien, S. & Kintner, C. A positive feedback mechanism governs the polarity and motion of motile cilia. Nature 447, 97–101 (2007).
Boisvieux-Ulrich, E., Laine, M.C. & Sandoz, D. Cytochalasin D inhibits basal body migration and ciliary elongation in quail oviduct epithelium. Cell Tissue Res. 259, 443–454 (1990).
Boisvieux-Ulrich, E., Laine, M.C. & Sandoz, D. In vitro effects of benzodiazepines on ciliogenesis in the quail oviduct. Cell Motil. Cytoskeleton 8, 333–344 (1987).
Marshall, W.F. The cell biological basis of ciliary disease. J. Cell Biol. 180, 17–21 (2008).
Goebbels, S. et al. Genetic targeting of principal neurons in neocortex and hippocampus of NEX-Cre mice. Genesis 44, 611–621 (2006).
Jones, C. et al. Ciliary proteins link basal body polarization to planar cell polarity regulation. Nat. Genet. 40, 69–77 (2008).
Wang, Y., Guo, N. & Nathans, J. The role of Frizzled3 and Frizzled6 in neural tube closure and in the planar polarity of inner-ear sensory hair cells. J. Neurosci. 26, 2147–2156 (2006).
Ross, A.J. et al. Disruption of Bardet-Biedl syndrome ciliary proteins perturbs planar cell polarity in vertebrates. Nat. Genet. 37, 1135–1140 (2005).
Mirzadeh, Z., Han, Y.G., Soriano-Navarro, M., Garcia-Verdugo, J.M. & Alvarez-Buylla, A. Cilia organize ependymal planar polarity. J. Neurosci. 30, 2600–2610 (2010).
Wong, L.L. & Adler, P.N. Tissue polarity genes of Drosophila regulate the subcellular location for prehair initiation in pupal wing cells. J. Cell Biol. 123, 209–221 (1993).
Chen, J., Knowles, H.J., Hebert, J.L. & Hackett, B.P. Mutation of the mouse hepatocyte nuclear factor/forkhead homologue 4 gene results in an absence of cilia and random left-right asymmetry. J. Clin. Invest. 102, 1077–1082 (1998).
Brody, S.L., Yan, X.H., Wuerffel, M.K., Song, S.K. & Shapiro, S.D. Ciliogenesis and left-right axis defects in forkhead factor HFH-4-null mice. Am. J. Respir. Cell Mol. Biol. 23, 45–51 (2000).
Pan, J., You, Y., Huang, T. & Brody, S.L. RhoA-mediated apical actin enrichment is required for ciliogenesis and promoted by Foxj1. J. Cell Sci. 120, 1868–1876 (2007).
Gomperts, B.N., Gong-Cooper, X. & Hackett, B.P. Foxj1 regulates basal body anchoring to the cytoskeleton of ciliated pulmonary epithelial cells. J. Cell Sci. 117, 1329–1337 (2004).
Ropper, A.H. & Brown, R.H. Adams and Victor's Principles of Neurology (McGraw Hill, 2005).
Sharma, N., Berbari, N.F. & Yoder, B.K. Ciliary dysfunction in developmental abnormalities and diseases. Curr. Top. Dev. Biol. 85, 371–427 (2008).
Tissir, F. & Goffinet, A.M. Expression of planar cell polarity genes during development of the mouse CNS. Eur. J. Neurosci. 23, 597–607 (2006).
Acknowledgements
We thank K. Jones for Emx1-Cre mice, G. Meyer for the antibody to SCO-spondin, and K.-A. Nave for NEX-Cre mice. We also thank V. Bonte, I. Lambermont and E. Paitre for technical assistance and M.F. Boulanger, Y. Houbion, M.-J. Vertez and R. Vigneron for help with electron microscopy. This work was supported by grants from the Actions de Recherches Concertées (ARC-186), FRFC 2.4504.01, FRSM 3.4501.07, Interuniversity Poles of Attraction (SSTC, PAI p6/20), the Fondation médicale Reine Elisabeth, the Wallonne and Bruxelloise régions from Belgium and from the Japan Science and Technology Corporation. F.T. is a research associate at the Belgian Fonds National de la Recherche Scientifique.
Author information
Authors and Affiliations
Contributions
F.T. characterized the Celsr2 mutant allele, studied Celsr2 and Celsr3 expression and carried out histological and immunofluorescence studies; Y.Q. performed SEM and TEM with the help of Y.P.; L.Z. and J.L. studied cilia function in brain slices; K.K. monitored cilia beats and analyzed data with D.S. and T.F.; M.M. provided the Vangl2 antibody and commented on the manuscript; D.S. and P.C. helped with the confocal microscopy and commented on the manuscript; T.U. analyzed the ciliary beat data, provided the Celsr2 antibody and commented on the manuscript; F.T. and A.M.G. designed research, analyzed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1 and 2 and Supplementary Data (PDF 2869 kb)
Supplementary Movie 1
Wild-type cilia beats, “seaweed” type. (MOV 3752 kb)
Supplementary Movie 2
Wild-type cilia beats, “pigeon neck” type. (MOV 3752 kb)
Supplementary Movie 3
Celsr2 mutant cilia beats, “seaweed” type. (MOV 3752 kb)
Supplementary Movie 4
Celsr2 mutant cilia beats, “pigeon neck” type. (MOV 3752 kb)
Supplementary Movie 5
Movement of latex beads in lateral ventricle of wild-type and Celsr2 mutant tissue. (MOV 5949 kb)
Rights and permissions
About this article
Cite this article
Tissir, F., Qu, Y., Montcouquiol, M. et al. Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus. Nat Neurosci 13, 700–707 (2010). https://doi.org/10.1038/nn.2555
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2555
This article is cited by
-
Dual-color live imaging unveils stepwise organization of multiple basal body arrays by cytoskeletons
EMBO Reports (2024)
-
Outcomes of the 2019 hydrocephalus association workshop, "Driving common pathways: extending insights from posthemorrhagic hydrocephalus"
Fluids and Barriers of the CNS (2023)
-
Celsr2 Knockout Alleviates Inhibitory Synaptic Stripping and Benefits Motoneuron Survival and Axon Regeneration After Branchial Plexus Avulsion
Molecular Neurobiology (2023)
-
LRP2 contributes to planar cell polarity-dependent coordination of motile cilia function
Cell and Tissue Research (2023)
-
Exonic mutations in cell–cell adhesion may contribute to CADASIL-related CSVD pathology
Human Genetics (2023)