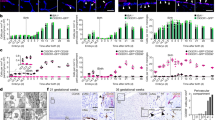
Abstract
Perivascular, subdural meningeal and choroid plexus macrophages are non-parenchymal macrophages that mediate immune responses at brain boundaries. Although the origin of parenchymal microglia has recently been elucidated, much less is known about the precursors, the underlying transcriptional program and the dynamics of the other macrophages in the central nervous system (CNS). It was assumed that they have a high turnover from blood-borne monocytes. However, using parabiosis and fate-mapping approaches in mice, we found that CNS macrophages arose from hematopoietic precursors during embryonic development and established stable populations, with the notable exception of choroid plexus macrophages, which had dual origins and a shorter life span. The generation of CNS macrophages relied on the transcription factor PU.1, whereas the MYB, BATF3 and NR4A1 transcription factors were not required.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ransohoff, R.M. & Cardona, A.E. The myeloid cells of the central nervous system parenchyma. Nature 468, 253–262 (2010).
Prinz, M., Priller, J., Sisodia, S.S. & Ransohoff, R.M. Heterogeneity of CNS myeloid cells and their roles in neurodegeneration. Nat. Neurosci. 14, 1227–1235 (2011).
Prinz, M. & Priller, J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat. Rev. Neurosci. 15, 300–312 (2014).
Alliot, F., Godin, I. & Pessac, B. Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res. Dev. Brain Res. 117, 145–152 (1999).
Ginhoux, F. et al. Fate-mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010).
Schulz, C. et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science 336, 86–90 (2012).
Kierdorf, K. et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. 16, 273–280 (2013).
Aguzzi, A., Barres, B.A. & Bennett, M.L. Microglia: scapegoat, saboteur, or something else? Science 339, 156–161 (2013).
Hickey, W.F. & Kimura, H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science 239, 290–292 (1988).
Hickey, W.F., Vass, K. & Lassmann, H. Bone marrow–derived elements in the central nervous system: an immunohistochemical and ultrastructural survey of rat chimeras. J. Neuropathol. Exp. Neurol. 51, 246–256 (1992).
Yang, Y. et al. Perivascular, but not parenchymal, cerebral engraftment of donor cells after non-myeloablative bone marrow transplantation. Exp. Mol. Pathol. 95, 7–17 (2013).
Barr, C.M., Manning, J., Lewis, C.A., Rossi, F.M. & Krieger, C. Submyeloablative conditioning with busulfan permits bone marrow–derived cell accumulation in a murine model of Alzheimer's disease. Neurosci. Lett. 588, 196–201 (2015).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 10, 1544–1553 (2007).
Mildner, A. et al. Distinct and non-redundant roles of microglia and myeloid subsets in mouse models of Alzheimer's disease. J. Neurosci. 31, 11159–11171 (2011).
Kierdorf, K., Katzmarski, N., Haas, C.A. & Prinz, M. Bone marrow cell recruitment to the brain in the absence of irradiation or parabiosis bias. PLoS One 8, e58544 (2013).
Galea, I. et al. Mannose receptor expression specifically reveals perivascular macrophages in normal, injured and diseased mouse brain. Glia 49, 375–384 (2005).
Kim, J.V., Kang, S.S., Dustin, M.L. & McGavern, D.B. Myelomonocytic cell recruitment causes fatal CNS vascular injury during acute viral meningitis. Nature 457, 191–195 (2009).
Zeisel, A. et al. Brain structure. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science 347, 1138–1142 (2015).
Jamieson, A.R. et al. Exploring nonlinear feature space dimension reduction and data representation in breast Cadx with Laplacian eigenmaps and t-SNE. Med. Phys. 37, 339–351 (2010).
Bechmann, I. et al. Turnover of rat brain perivascular cells. Exp. Neurol. 168, 242–249 (2001).
Priller, J. et al. Targeting gene-modified hematopoietic cells to the central nervous system: use of green fluorescent protein uncovers microglial engraftment. Nat. Med. 7, 1356–1361 (2001).
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 518, 547–551 (2015).
Molawi, K. et al. Progressive replacement of embryo-derived cardiac macrophages with age. J. Exp. Med. 211, 2151–2158 (2014).
Ensan, S. et al. Self-renewing resident arterial macrophages arise from embryonic CX3CR1 precursors and circulating monocytes immediately after birth. Nat. Immunol. 17, 159–168 (2015).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91 (2013).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nat. Neurosci. 16, 1618–1626 (2013).
Rosenbauer, F. & Tenen, D.G. Transcription factors in myeloid development: balancing differentiation with transformation. Nat. Rev. Immunol. 7, 105–117 (2007).
Geissmann, F. et al. Development of monocytes, macrophages, and dendritic cells. Science 327, 656–661 (2010).
Ajami, B., Bennett, J.L., Krieger, C., Tetzlaff, W. & Rossi, F.M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Wieghofer, P., Knobeloch, K.P. & Prinz, M. Genetic targeting of microglia. Glia 63, 1–22 (2015).
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nat. Neurosci. 8, 752–758 (2005).
Ajami, B., Bennett, J.L., Krieger, C., McNagny, K.M. & Rossi, F.M. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat. Neurosci. 14, 1142–1149 (2011).
Butovsky, O. et al. Identification of a unique TGF-β–dependent molecular and functional signature in microglia. Nat. Neurosci. 17, 131–143 (2014).
Hickman, S.E. et al. The microglial sensome revealed by direct RNA sequencing. Nat. Neurosci. 16, 1896–1905 (2013).
Ginhoux, F. & Jung, S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 14, 392–404 (2014).
Chorro, L. et al. Langerhans cell (LC) proliferation mediates neonatal development, homeostasis, and inflammation-associated expansion of the epidermal LC network. J. Exp. Med. 206, 3089–3100 (2009).
Guilliams, M. et al. Alveolar macrophages develop from fetal monocytes that differentiate into long-lived cells in the first week of life via GM-CSF. J. Exp. Med. 210, 1977–1992 (2013).
Epelman, S. et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 40, 91–104 (2014).
Sheng, J., Ruedl, C. & Karjalainen, K. Most tissue-resident macrophages except microglia are derived from fetal hematopoietic stem cells. Immunity 43, 382–393 (2015).
Adolfsson, J. et al. Upregulation of Flt3 expression within the bone marrow Lin(−)Sca1(+)c-kit(+) stem cell compartment is accompanied by loss of self-renewal capacity. Immunity 15, 659–669 (2001).
Epelman, S., Lavine, K.J. & Randolph, G.J. Origin and functions of tissue macrophages. Immunity 41, 21–35 (2014).
Kunis, G. et al. IFN-γ-dependent activation of the brain's choroid plexus for CNS immune surveillance and repair. Brain 136, 3427–3440 (2013).
Engelhardt, B., Wolburg-Buchholz, K. & Wolburg, H. Involvement of the choroid plexus in central nervous system inflammation. Microsc. Res. Tech. 52, 112–129 (2001).
Axtell, R.C. & Steinman, L. Gaining entry to an uninflamed brain. Nat. Immunol. 10, 453–455 (2009).
Kivisäkk, P. et al. Localizing central nervous system immune surveillance: meningeal antigen-presenting cells activate T cells during experimental autoimmune encephalomyelitis. Ann. Neurol. 65, 457–469 (2009).
Anandasabapathy, N. et al. Flt3L controls the development of radiosensitive dendritic cells in the meninges and choroid plexus of the steady-state mouse brain. J. Exp. Med. 208, 1695–1705 (2011).
Brendecke, S.M. & Prinz, M. Do not judge a cell by its cover—diversity of CNS resident, adjoining and infiltrating myeloid cells in inflammation. Semin. Immunopathol. 37, 591–605 (2015).
Misgeld, T., Nikic, I. & Kerschensteiner, M. In vivo imaging of single axons in the mouse spinal cord. Nat. Protoc. 2, 263–268 (2007).
Romanelli, E. et al. Cellular, subcellular and functional in vivo labeling of the spinal cord using vital dyes. Nat. Protoc. 8, 481–490 (2013).
Goldmann, T. et al. USP18 lack in microglia causes destructive interferonopathy of the mouse brain. EMBO J. 34, 1612–1629 (2015).
Acknowledgements
We thank M. Oberle, M. Ditter and T. el Gaz for excellent technical assistance and T. Leng Tay for critical reading of the manuscript. Supported by the DFG (SFB 992, SFB 1160, PR 577/8-1, Reinhart Koselleck Grant for M.P.; SFB 1160, ZE872/3-1 for R.Z.; and FOR1336 for J.P., I.B., M.P. and S.J.), the Fritz-Thyssen Foundation (M.P.), the European Union's Seventh Framework Program FP7 under Grant agreement 607962 (nEUROinflammation for M.P.), the Gemeinnützige Hertie Foundation (GHST for M.P.), the Sobek Foundation (M.P.) and the BMBF-funded Competence Network on Multiple Sclerosis (KKNMS for M.P. and M. Kerschensteiner).
Author information
Authors and Affiliations
Contributions
T.G., P.W., M.J.C.J., F.P., N.H., K.F., O.S., K.K., L.A., M. Krueger, G.L. and H.H. conducted the experiments and analyzed the data. R.Z., S.E., F.G., J.P., F.M.V.R., I.B., S.L., M. Kerschensteiner and S.J. analyzed the data, contributed to the in vivo studies and provided mice or reagents. T.G. and M.P. supervised the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Single-cell RNA sequencing of pvMΦ, microglia and monocytes.
(a) Work flow for obtaining and analyzing single-cell RNA-seq from mouse cortical cells and monocytes from dissection to single-cell RNA-seq and unbiased biclustering.
(b) Representative image of single cells captured in the chip. All cells were checked individually by light microscopy and chambers with no or damaged cells (red squares) were omitted from subsequent analysis.
(c-f) Bar graphs for commonly expressed genes (c), markers selectively expressed by pvMΦ (d), selectively expressed by cortical microglia (e) or just by monocytes (f) evaluated by single cell RNA-seq.
Picture from cell capture for RNAseq is representative of one independent experiment (b). Single cell RNAseq data (c-e) is representative of two independent experiments with 167 ptMΦ, 246 monocytes, 33 microglia and 65 pvMΦ. Data is represented as mean ± s.e.m.
Supplementary Figure 2 Flow cytometry of cortical pvMΦ and microglia.
(a) Representative gating strategy for the flow cytometry-based isolation of bone marrow monocytes for subsequent single-cell RNA-sequencing as shown in Fig. 1d-g.
(b) Representative gating strategy for the isolation of cortical CD11b+ CD45lo microglia and CD11b+ CD45hi cells from the cortex (meninges and choroid plexus were removed) as shown in Fig. 1j-k.
(c) CD11b+ CD45hi pvMΦ can be further separated from CD11b+CD45lo microglia by gating them as CD45hi CD11b+ Ly6C− Ly6G− CD206+ F4/80+ cells. Representative histograms for CD206, CD36 and F4/80 staining on pvMΦ and microglia (black line) are shown over isotype controls (filled gray).
d) Representative gating strategy for the flow cytometry-based characterization of Ly6C cells as shown in Fig. 6a-b. Peripheral blood leukocytes were gated according to physical parameters and further subdivided in myeloid cells by the expression of CD45 and CD11b. CD45+ CD11b+ SSClo CD115+ monocytes are then further discriminated into Ly6Chi inflammatory monocytes and Ly6Clo patrolling monocytes.
Gating strategies are representative of six mice from two independent experiments (a, d) or from three biological replicates from two independent experiments (b, c).
Supplementary Figure 3 Irradiation induces engraftment of bone-marrow-derived CNS macrophages and microglia.
a) Scheme for the induction of recombination (injection of tamoxifen [TAM]) and subsequent analysis in Cx3cr1CreERRosa26-YFP animals.
b) Scheme and timeline for labelling and analyses of pvMΦ, mMΦ and cpMΦ in adulthood using TAM injection in adult Cx3cr1CreERRosa26-YFP animals.
c) Direct fluorescence microscopic visualization revealed numerous GFP+ donor-derived Iba-1+ pvMΦ, mMΦ, cpMΦ and few microglia 20 weeks after transfer of bone marrow from Acta1-GFP mice into lethally irradiated wild-type mice. Arrows indicate double positive, asterisks single Iba-1 (red) positive cells. Scale bar = 25 μm.
d) Quantification of donor-derived GFP+ Iba-1+ cells.
Immunofluorescence pictures (c) are representative of four mice from one independent experiment. Data were obtained from five mice per group from one independent experiment (d). Each symbol represents one mouse and three tissue sections per mouse were quantified. (means ± s.e.m.)
Supplementary Figure 4 CNS macrophages do not require Batf3.
a) Localization and presence of pvMΦ, mMΦ and cpMΦ in adult wild-type (WT) and Batf3−/− mice evaluated using Iba-1 immunohistochemistry. Representative figure are presented (upper images) and quantification thereof.
b) Confocal pictures showing Tomato+ cells in the choroid plexus of Cx3cr1CreERRosa26-Tomato mice (tomato, red) at 8 weeks after TAM expressing the macrophage marker F4/80 and to a much lesser extent CD206. Scale bar: 25 μm.
Immunofluorescence pictures are representative of of five mice from two independent experiments (b). Immunohistochemistry pictures (a) are representative of three mice per genotype from one independent experiment. Macrophage density (a) is representative of three mice per genotype from one independent experiment (Meninges: P= 0.7; perivascular space: P=0.7; choroid plexus P= 1). Mann Whitney test was applied. Each symbol represents one mouse with quantification of a minimum of three tissue sections. (error bars, s.e.m.). N.S. = not significant.
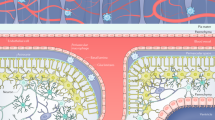
Supplementary Figure 5 Model for experimental findings on the origin, fate and dynamics of microglia and macrophages at CNS boundaries.
Traditional and proposed view on the origin, fate and turnover of non-parenchymal CNS macrophages and microglia.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 1295 kb)
Location and morphology of pvMΦ, mMΦ and microglial cells.
3D-rendering (Imaris, Bitplane) of the confocal image stack represented in Fig. 5b that illustrates the distinct pvMΦ, mMΦ and microglial cells in the intact spinal cord of a Cx3cr1CreER:Rosa26-Tomato mice (tomato, red) at 8 weeks after TAM and injected with dextran-AF647 (blood vessel, blue). (MP4 8147 kb)
Dynamics of the myeloid cells.
Confocal projection of the dorsal spinal cord as shown in Fig. 5c and 5d followed by in vivo 2-photon time-lapse imaging of the different myeloid cells in Cx3cr1CreER:Rosa26-Tomato mice at 8 weeks after TAM. (MP4 979 kb)
Rights and permissions
About this article
Cite this article
Goldmann, T., Wieghofer, P., Jordão, M. et al. Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat Immunol 17, 797–805 (2016). https://doi.org/10.1038/ni.3423
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3423
This article is cited by
-
A P2RY12 deficiency results in sex-specific cellular perturbations and sexually dimorphic behavioral anomalies
Journal of Neuroinflammation (2024)
-
Border-associated macrophages in the central nervous system
Journal of Neuroinflammation (2024)
-
IKKβ deletion from CNS macrophages increases neuronal excitability and accelerates the onset of EAE, while from peripheral macrophages reduces disease severity
Journal of Neuroinflammation (2024)
-
Blood–brain borders: a proposal to address limitations of historical blood–brain barrier terminology
Fluids and Barriers of the CNS (2024)
-
Role of meningeal immunity in brain function and protection against pathogens
Journal of Inflammation (2024)