Abstract
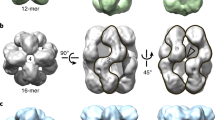
α-Crystallin is a major lens protein, comprising up to 40% of total lens proteins, where its structural function is to assist in maintaining the proper refractive index in the lens. In addition to its structural role, it has been shown to function in a chaperone-like manner. The chaperone-like function of α-crystallin will help prevent the formation of large light-scattering aggregates and possibly cataract. In the lens, α-crystallin is a polydisperse molecule consisting of a 3:1 ratio of αA to αB subunits. In this study, we expressed recombinant αA- and αB-crystallin in E. coli and compared the polydispersity, structure and aggregation state between each other and native bovine lens α-crystallin. Using gel permeation chromatography to assay for polydispersity, we found native α-crystallin to be significantly more polydisperse than either recombinant αA- or αB-crystallin, with αB-crystallin having the most homogeneous structure of the three. Reconstructed images of αB-crystallin obtained with cryo-electron microscopy support the concept that αB-crystallin is an extremely dynamic molecule and demonstrated that it has a hollow interior. Interestingly, we present evidence that native α-crystallin is significantly more thermally stable than either αA- or αB-crystallin alone. In fact, our experiments suggest that a 3:1 ratio of αA to αB subunit composition in an α-crystallin molecule is optimal in terms of thermal stability. This fascinating result explains the stoichiometric ratios of αA- and αB-crystallin subunits in the mammalian lens.
Similar content being viewed by others
Article PDF
References
de Jong WW, Lubsen NH, Kraft HJ . Molecular evolution of the eye lens. Prog Retinal Eye Res 1994;13:391–442.
Harding JJ . Lens. In: Harding JJ, ed. Biochemistry of the eye. London: Chapman & Hall, 1997:94–135.
Groenen PJTA, Merck KB, de Jong WW, Bloemedal H . Structure and modifications of the junior-chaperone alpha-crystallin: from lens transparency to molecular pathology. Eur J Biochem 1994;225:1–19.
Bhat SP, Nagineni CN . Alpha B subunit of lens specific protein alpha-crystallin is present in other ocular and non-ocular tissues. Biochem Biophys Res Commun 1989;158:319–25.
Dubin RA, Wawrousek EF, Piatigorsky J . Expression of the marine alpha B-crystallin gene is not restricted to the lens. Mol Cell Biol 1989;9:1083–91.
Klemenz R, Fröhli E, Steiger RH, Schäfer R, Aoyama A . Alpha B-crystallin is a small heat-shock protein. Proc Natl Acad Sci USA 1991;88:3652–6.
Horwitz J . Alpha-crystallin can function as a molecular chaperone. Proc Natl Acad Sci USA 1992;89:10449–53.
Horwitz J, Huang QL, Ding LL, Bova MP . Lens α-crystallin: chaperone-like qualities. In: Lorimar GH, Baldwin TO, eds. Methods in enzymology, vol 290: Molecular chaperones. San Diego: Academic Press, 1998:365–83.
Haley DA, Horwitz J, Stewart PL . The small heat-shock protein, alpha B-crystallin, has a variable quaternary structure. J Mol Biol 1998;277:27–35.
Harding JJ, Crabbe MJC . The lens: development, proteins, metabolism and cataract. In: Davson H, ed. The eye, 3rd edn, vol. 1B: Vegetative physiology and biochemistry. Orlando: Academic Press, 1984:207–492.
Roy D, Spector A . Absence of low-molecular weight alpha-crystallin in the nuclear region of old human lenses. Proc Natl Acad Sci USA 1976;73:3484–7.
McFall-Ngai MJ, Ding LL, Takemoto LJ, Horwitz J . Spatial and temporal mappings of the age-related changes in human lens crystallins. Exp Eye Res 1985;41:745–58.
Siezen RJ, Bindels JG, Hoenders HJ . The quaternary structure of bovine alpha-crystallin: size and change microheterogeneity; more than 1000 different hybrids. Eur J Biochem 1978;91:387–96.
Farnsworth P, Singh K . Alpha-crystallin. Int J Biol Macromol 1998;22(special issue):149–343.
Bindels JG, Siezen RJ, Hoenders HJ . A model for the architecture of alpha-crystallin. Ophthalmic Res 1979;11:441–52.
Tardieu A, Laporte D, Licinio P, Krop B, Delaye M . Calf lens alpha-crystallin quarternary structure: a three-layer tetrahedral model. J Mol Biol 1986;192:711–24.
Augusteyn RC, Koretz JF . A possible structure for alpha-crystallin. FEBS Lett 1987;222:1–5.
Wistow G . Possible tetramer-based quaternary structures for the alpha-crystallins and small heat-shock proteins. Exp Eye Res 1993;56:729–32.
Carver JA, Aquilina JA, Truscott RJW . A possible chaperone-like quaternary structure for alpha-crystallin. Exp Eye Res 1994;59:231–4.
Groth-Vasseli B, Kumosinski TF, Farnsworth PN . Computer-generated model of the quaternary structure of alpha-crystallin in the lens. Exp Eye Res 1995;61:249–53.
Smulders RHPH, van Boekel MAM, de Jong WW . Mutations and modifications support a ‘pitted-flexiball’ model for alpha-crystallin. Int J Biol Macromol 1998;22:187–96.
Jaffe NS, Horwitz J . Lens and cataract. In: Podos SM, Yanoff M, eds. Textbook of ophthalmology, vol 3. New York: Gower, 1992: chaps 1-4.
Wistow G, Piatigorsky J . Recruitment of enzymes as lens structural proteins. Science 1987;236:1554–6.
Wistow G, Piatigorsky J . Lens crystallins: the evolution and expression of proteins for a highly specialised tissue. Annu Rev Biochem 1988;57:479–504.
Piatigorsky J . Gene sharing in lens and cornea: facts and implications. In: Osborne NN, Chader GJ, eds. Prog Retinal Eye Res 1998;17:145–65.
Haynes JI, Duncan MK, Piatigorsky J . Spatial and temporal activity of the alpha B-crystallin/small heat-shock protein gene promoter in transgenic mice. Dev Dynamics 1996;207:75–88.
Rao PV, Huang QL, Horwitz J, Zigler SJ . Evidence that alpha-crystallin prevents non-specific protein aggregation in the intact eye lens. Biochim Biophys Acta 1995;1245:439–47.
Velasco FT, Lukas TJ, Murthy SNP, Duglas-Tabor Y, Garland D, Lorand L . Hierarchy of lens proteins requiring protection against heat-induced precipitation by the alpha-crystallin chaperone. Exp Eye Res 1997;65:497–505.
Sun TX, Liang JJN . Intermolecular exchange and stabilisation of recombinant human alpha A and alpha B-crystallin. J Biol Chem 1998;273:286–90.
Brady JP, Garland D, Duglas-Tabor Y, Robison WG, Groome A, Wawrousek EF . Targeted disruption of the mouse alpha A-crystallin gene induces cataract and cytoplasmic inclusion bodies containing the small head-shock protein alpha-B. Proc Natl Acad Sci USA 1997;94:884–9.
Van den Oetelaar PJM, Van Someren PFHM, Thomson JA, Siezen RJ, Hoenders HJ . A dynamic quaternary structure of bovine alpha-crystallin as indicated from intermolecular exchange of subunits. Biochemistry 1990;29:3488–93.
Bova MP, Ding LL, Horwitz J, Fung BKK . Subunit exchange of alpha A-crystallin. J Biol Chem 1997;272:29511–7.
Raman B, Rao CM . Chaperone-like activity and quaternary structure of alpha-crystallin. J Biol Chem 1994;269:27264–8.
Kim KK, Kim R, Kim S-H . Crystal structure of a small heat-shock protein. Nature 1998;394:595–9.
Haley D, Horwitz J, Stewart P . Image restrained modelling of alpha B-crystallin. Exp Eye Res 1999;68:133–6.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Eye Institute R37-EY3897 to J.H.
Rights and permissions
About this article
Cite this article
Horwitz, J., Bova, M., Ding, L . et al. Lens α-crystallin: Function and structure. Eye 13, 403–408 (1999). https://doi.org/10.1038/eye.1999.114
Issue Date:
DOI: https://doi.org/10.1038/eye.1999.114
Keywords
This article is cited by
-
Novel and known variants in GJA3 and LIM2 in congenital cataract families from North India
BMC Genomics (2024)
-
Mini-αA-Crystallin Stifled Melittin-Induced Haemolysis and Lymphocyte Lysis
International Journal of Peptide Research and Therapeutics (2023)
-
Qki activates Srebp2-mediated cholesterol biosynthesis for maintenance of eye lens transparency
Nature Communications (2021)
-
Elucidation of the mechanism of subunit exchange in αB crystallin oligomers
Scientific Reports (2021)
-
Imbalances in the eye lens proteome are linked to cataract formation
Nature Structural & Molecular Biology (2021)