Summary
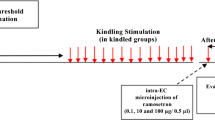
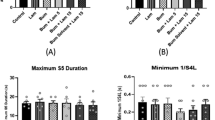
The purpose of this study was to evaluate the effect of adenosine A2A receptor antagonist ZM241385 on amygdala-kindled seizures and its roles in epileptogenesis. Electrodes were implanted into the right amygdala of male adult Wistar rats. Kindling was accomplished by using stimulus strength of 500 μA applied daily to the amygdala until 10 consecutive stage 5 seizues were induced. Then effect of ZM241385 was studied in fully kindled rats after intracerebroventricular administration of the drug. In addition, the effect on kindling progression was evaluated through ZM241385 injection before daily stimulation. In all experiments, behavioral changes in the rats in response to ZM241385 were monitored closely. The results showed that, in fully amygdala-kindled rats, ZM241385 (0.001–0.1 nmol/L) decreased afterdischage duration (ADD), motor seizure duration (MSD), stage 5 duration (S5D) and seizure duration (SD), but only the effect on ADD was dose-dependent. The doses of 0.001–0.1 nmol/L had no influence on stage 4 latency (S4L) and seizure stage (SS). The dosages of 0.0001 and 1 nmol/L of ZM241385 did not exert any effect on all seizure parameters. In contrast to the results in fully amygdala-kindled rats, ZM241385 (0.001–0.1 nmol/L) had minimal or no effects on the progression of amygdala-kindled seizures. We are led to the conclusion that although ZM241385 had no influence on the progression of amygdala-kindled seizures, it had potent anti-convulsant profile and little adverse effects at the dosage of 0.001–0.1 nmol/L, suggesting that the agent is effective against the amygdala-kindled seizures.
Similar content being viewed by others
References
Fredholm BB, Chen JF, Cunha RA, et al. Adenosine and brain function. Int Rev Neurobiol, 2005,63:191–270
Fredholm BB, Ijzerman AP, Jacobson KA, et al. International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol Rev, 2001,53(4):527–552
Gomes CV, Kaster MP, Tome AR, et al. Adenosine receptors and brain diseases: neuroprotection and neurodegeneration. Biochim Biophys Acta, 2011,1808(5): 1380–1399
Boison D. Adenosine and epilepsy: from therapeutic rationale to new therapeutic strategies. Neuroscientist, 2005,11(1):25–36
Dolphin AC, Prestwich SA. Pertussis toxin reverses adenosine inhibition of neuronal glutamate release. Nature, 1985,316(6024):148–150
Ribeiro JA, Cunha RA, Correia-De-Sa P, et al. Purinergic regulation of acetylcholine release. Prog Brain Res, 1996,109:231–241
Greene RW, Haas HL. The electrophysiology of adenosine in the mammalian central nervous system. Prog Neurobiol, 1991,6(4):329–341
Gouder N, Fritschy JM, Boison D. Seizure suppression by adenosine A1 receptor activation in a mouse model of pharmacoresistant epilepsy. Epilepsia, 2003,44(7): 877–885
Fedele DE, Li T, Lan JQ, et al. Adenosine A1 receptors are crucial in keeping an epileptic focus localized. Exp Neurol, 2006,200(1):184–190
Shryock JC, Belardinelli L. Adenosine and adenosine receptors in the cardiovascular system: biochemistry, physiology, and pharmacology. Am J Cardiol, 1997,79(12A):2–10
Svenningsson P, Le Moine C, Aubert I, et al. Cellular distribution of adenosine A2A receptor mRNA in the primate striatum. J Comp Neurol, 1998,399(2):229–240
Rebola N, Sebastiao AM, de Mendonca A, et al. Enhanced adenosine A2A receptor facilitation of synaptic transmission in the hippocampus of aged rats. J Neurophysiol, 2003,90(2):1295–1303
Rebola N, Porciuncula LO, Lopes LV, et al. Long-term effect of convulsive behavior on the density of adenosine A1 and A2A receptors in the rat cerebral cortex. Epilepsia, 2005,46 (Suppl 5):159–165
Loscher W, Schmidt D. Which animal models should be used in the search for new antiepileptic drugs? A proposal based on experimental and clinical considerations. Epilepsy Res, 1988,2(3):145–181
Zeraati M, Mirnajafi-Zadeh J, Fathollahi Y, et al. Adenosine A1 and A2A receptors of hippocampal CA1 region have opposite effects on piriform cortex kindled seizures in rats. Seizure, 2006,15(1):41–48
Hosseinmardi N, Mirnajafi-Zadeh J, Fathollahi Y, et al. The role of adenosine A1 and A2A receptors of entorhinal cortex on piriform cortex kindled seizures in rats. Pharmacol Res, 2007,56(2):110–117
Loscher W. Animal models of epilepsy for the development of antiepileptogenic and disease-modifying drugs. A comparison of the pharmacology of kindling and post-status epilepticus models of temporal lobe epilepsy. Epilepsy Res, 2002,50(1–2):105–123
Paxinos G, Watson CR, Emson PC. AChE-stained horizontal sections of the rat brain in stereotaxic coordinates. J Neurosci Methods, 1980,3(2):129–149
Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol, 1972,32(3):281–294
Boeck CR, Martinello C, de Castro AA, et al. Blockade of adenosine A2A receptor counteracts neuropeptide-S-induced hyperlocomotion in mice. Naunyn Schmiedebergs Arch Pharmacol, 2010,381(2):153–160
Lopes LV, Halldner L, Rebola N, et al. Binding of the prototypical adenosine A(2A) receptor agonist CGS 21680 to the cerebral cortex of adenosine A(1) and A(2A) receptor knockout mice. 21680 to the cerebral cortex of adenosine A(1) and A(2A) receptor knockout mice. Br J Pharmacol, 2004,141(6): 1006–1014
Cunha RA, Milusheva E, Vizi ES, et al. Excitatory and inhibitory effects of A1 and A2A adenosine receptor activation on the electrically evoked [3H]acetylcholine release from different areas of the rat hippocampus. J Neurochem, 1994,63(1):207–214
Tomiyama M, Kimura T, Maeda T, et al. Upregulation of striatal adenosine A2A receptor mRNA in 6-hydroxy-dopamine-lesioned rats intermittently treated with L-DOPA. Synapse, 2004,52(3):218–222
D’Alimonte I, D’Auro M, Citraro R, et al. Altered distribution and function of A2A adenosine receptors in the brain of WAG/Rij rats with genetic absence epilepsy, before and after appearance of the disease. Eur J Neurosci, 2009,30(6):1023–1035
De Sarro G, De Sarro A, Di Paola ED, et al. Effects of adenosine receptor agonists and antagonists on audiogenic seizure-sensible DBA/2 mice. Eur J Pharmacol, 1999,371(2–3):137–145
Zhang G, Franklin PH, Murray TF. Activation of adenosine A1 receptors underlies anticonvulsant effect of CGS21680. Eur J Pharmacol, 1994,255(1–3):239–243
Yacoubi ME, Ledent C, Parmentier M, et al. Adenosine A2A receptor deficient mice are partially resistant to limbic seizures. Naunyn Schmiedebergs Arch Pharmacol, 2009,380(3):223–232
Yacoubi ME, Ledent C, Parmentier M, et al. Absence of the adenosine A(2A) receptor or its chronic blockade decrease ethanol withdrawal-induced seizures in mice. Neuropharmacology, 2001,40(3):424–432
Tchekalarova J, Sotiriou E, Georgiev V, et al. Up-regulation of adenosine A1 receptor binding in pentylenetetrazol kindling in mice: effects of angiotensin IV. Brain Res, 2005,1032(1–2):94–103
Jones PA, Smith RA, Stone TW. Protection against hippocampal kainate excitotoxicity by intracerebral administration of an adenosine A2A receptor antagonist. Brain Res, 1998,800(2):328–335
Palmer TM, Poucher SM, Jacobson KA, et al. 125I-4-(2-[7-amino-2-[2-furyl][1, 2, 4]triazolo[2, 3-a][1, 3, 5] triazin-5-yl-amino]ethyl)phenol, a high affinity antagonist radioligand selective for the A2a adenosine receptor. Mol Pharmacol, 1995,48(6):970–974
Blum D, Galas MC, Pintor A, et al. A dual role of adenosine A2A receptors in 3-nitropropionic acid-induced striatal lesions: implications for the neuroprotective potential of A2A antagonists. J Neurosci, 2003,23(12):5361–5369
Popoli P, Pintor A, Domenici MR, et al. Blockade of striatal adenosine A2A receptor reduces, through a presynaptic mechanism, quinolinic acid-induced excitotoxicity: possible relevance to neuroprotective interventions in neurodegenerative diseases of the striatum. J Neurosci, 2002,22(5):1967–1975
Monopoli A, Casati C, Lozza G, et al. Cardiovascular pharmacology of the A2A adenosine receptor antagonist, SCH 58261, in the rat. J Pharmacol Exp Ther, 1998,285(1):9–15
Marcoli M, Raiteri L, Bonfanti A, et al. Sensitivity to selective adenosine A1 and A2A receptor antagonists of the release of glutamate induced by ischemia in rat cerebrocortical slices. Neuropharmacology, 2003,45(2): 201–210
Popoli P, Frank C, Tebano MT, et al. Modulation of glutamate release and excitotoxicity by adenosine A2A receptors. Neurology. 2003,61(11 Suppl 6):S69–S71
Xiaoqin Z, Zhengli L, Zhu CG, et al. Changes in behavior and amino acid neurotransmitters in the brain of rats with seizure induced by IL-1beta or IL-6. J Huazhong Univ Sci Technolog [Med Sci], 2005,25(3):236–239
Deng X, Jia H, Yang Z, et al. Correlation study on expression of GST-pi protein in brain tissue and peripheral blood of epilepsy rats induced by pilocarpine. J Huazhong Univ Sci Technolog [Med Sci], 2011,31(5):701–704
Roseti C, Palma E, Martinello K, et al. Blockage of A2A and A3 adenosine receptors decreases the desensitization of human GABA (A) receptors microtransplanted to Xenopus oocytes. Proc Natl Acad Sci USA, 2009,106(37): 15927–15931
Weiss SR, Post RM. Kindling: separate vs. shared mechanisms in affective disorders and epilepsy. Neuropsychobiology, 1998,38(3):167–180
Hamada K, Song HK, Ishida S, et al. Contrasting effects of zonisamide and acetazolamide on amygdaloid kindling in rats. Epilepsia, 2001,42(11):1379–1386
Weiss SR, Post RM. Carbamazepine and carbamazepine-10, 11-epoxide inhibit amygdala-kindled seizures in the rat but do not block their development. Clin Neuropharmacol, 1987,10(3):272–279
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a grant from the National Natural Science Foundation of China (No. 30770752).
Rights and permissions
About this article
Cite this article
Li, X., Kang, H., Liu, X. et al. Effect of adenosine A2A receptor antagonist ZM241385 on amygdala-kindled seizures and progression of amygdala kindling. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 32, 257–264 (2012). https://doi.org/10.1007/s11596-012-0046-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-012-0046-2