Abstract
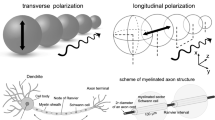
Almost 90 years ago, Lillie reported that rapid saltatory conduction arose in an iron wire model of nerve impulse propagation when he covered the wire with insulating sections of glass tubing equivalent to myelinated internodes. This led to his suggestion of a similar mechanism explaining rapid conduction in myelinated nerve. In both their evolution and their development, myelinating axons must make a similar transition between continuous and saltatory conduction. Achieving a smooth transition is a potential challenge that we examined in computer models simulating a segmented insulating sheath surrounding an axon having Hodgkin-Huxley squid parameters. With a wide gap under the sheath, conduction was continuous. As the gap was reduced, conduction initially slowed, owing to the increased extra-axonal resistance, then increased (the “rise”) up to several times that of the unmyelinated fiber, as saltatory conduction set in. The conduction velocity slowdown was little affected by the number of myelin layers or modest changes in the size of the “node,” but strongly affected by the size of the “internode” and axon diameter. The steepness of the rise of rapid conduction was greatly affected by the number of myelin layers and axon diameter, variably affected by internode length and little affected by node length. The transition to saltatory conduction occurred at surprisingly wide gaps and the improvement in conduction speed persisted to surprisingly small gaps. The study demonstrates that the specialized paranodal seals between myelin and axon, and indeed even the clustering of sodium channels at the nodes, are not necessary for saltatory conduction.






Similar content being viewed by others
References
Brill, M. H., Waxman, S. D., Moore, J. W., & Joyner, R. W. (1977). Conduction velocity and spike configuration in myelinated fibres: computed dependence on internode distance. Journal of Neurology, Neurosurgery, and Psychiatry, 40, 769–774.
Brösamle, C., & Halpern, M. E. (2002). Characterization of myelination in the developing zebrafish. Glia, 39, 47–57.
Bullock, T. H., & Horridge, G. A. (1965). Structure and function in the nervous system of invertebrates (Vol. 1). San Francisco: W. H. Freeman.
Calvin, W. H., & Hartline, D. K. (1977). Retrograde invasion of lobster stretch receptor somata in control of firing rate and extra spike patterning. Journal of Neurophysiology, 40, 106–118.
Carnevale, N. T., & Hines, M. L. (2005). The NEURON Book. Cambridge, U.K.: Cambridge University Press.
Carpenter, F. G., & Bergland, R. M. (1975). Excitation and conduction in immature nerve fibers of the developing chick. American Journal of Physiology, 190, 371–376.
Chomiak, T., & Hu, B. (2009). What is the optimal value of the g-ratio for myelinated fibers in the rat CNS? A theoretical approach. PLoS One, 4, e7754.
Foster, R. E., Connors, B. W., & Waxman, S. D. (1982). Rat optic nerve: electrophysiological, pharmacological and anatomical studies during development. Brain Research, 255, 371–386.
Freidländer, B. (1889). Über die markhaltigen Nervenfasern und Neurochorde der Crustaceen und Anneliden. Mittheilungen aus der Zoologischen Station zu Neapel, 9, 205–265.
Goldman, L., & Albus, J. S. (1968). Computation of impulse conduction in myelinated fibers: theoretical basis of the velocity-diameter relation. Biophysical Journal, 8, 596–607.
Goldstein, S. S., & Rall, W. (1974). Changes of action potential shape and velocity for changing core conductor geometry. Biophysical Journal, 14, 731–757.
Halter, J. A., & Clark, J. W. (1991). A distributed-parameter model of the myelinated nerve fiber. Journal of Theoretical Biology, 148, 345–382.
Hartline, D. K. (2008). What is myelin? Neuron Glia Biology, 4, 153–163.
Hartline, D. K., & Colman, D. R. (2007). Rapid conduction and the evolution of giant axons and myelinated fibers. Current Biology, 17, R29–R35.
Hille, B. (1992). Ionic channels of excitable membranes (2nd ed.). Sunderland: Sinauer Associates Inc.
Hines, M. L., & Shrager, P. (1991). A computational test of the requirements for conduction in demyelinated axons. Restorative Neurology and Neuroscience, 3, 81–93.
Hodgkin, A. L. (1938). The subthreshold potentials in a crustacean nerve fibre. Proceedings of the Royal Society of London Series B, 126, 87–121.
Hodgkin, A. L., & Huxley, A. F. (1952). A quantitative description of membrane current and its application to conduction and excitation in nerve. The Journal of Physiology, 117, 500–544.
Huxley, A. F., & Stämpfli, R. (1949). Evidence for saltatory conduction in peripheral myelinated nerve fibers. The Journal of Physiology, 108, 315–339.
Lillie, R. S. (1925). Factors affecting transmission and recovery in the passive iron nerve model. Journal of General Physiology, 7, 473–507.
Moore, J. W., Joyner, R. W., Brill, M. H., Waxman, S. D., & Najar-Joa, M. (1978). Simulations of conduction in uniform myelinated fibers: relative sensitivity to changes in nodal and internodal parameters. Biophysical Journal, 21, 147–160.
Pumphrey, R. J., & Young, J. Z. (1938). The rates of conduction of nerve fibres of various diameters in cephalopods. The Journal of Experimental Biology, 15, 453–466.
Raine, C. (1984). Morphology of myelin and myelination. In P. Morell (Ed.), Myelin (2nd ed., pp. 1–50). London: Plenum Press.
Rovainen, C. M. (1967). Physiological and anatomical studies on large neurons of central nervous system of the sea lamprey (Petromyzon marinus). I. Müller and Mauthner cells. Journal of Neurophysiology, 30, 1000–1023.
Seidl, A. H., Rubel, E. W., & Harris, D. M. (2010). Mechanisms for adjusting interaural time differences to achieve binaural coincidence detection. Journal of Neuroscience, 30, 70–80.
Smith, R. S., & Koles, Z. J. (1970). Myelinated nerve fibers: computed effect of myelin thickness on conduction velocity. American Journal of Physiology, 219, 1256–1258.
Tasaki, I. (1939). The electro-saltatory transmission of the nerve impulse and the effect of narcosis upon the nerve fiber. American Journal of Physiology, 127, 211–227.
Vabnick, I., & Shrager, P. (1999). Ion channel redistribution and function during development of the myelinated axon. Journal of Neurobiology, 37, 80–96.
Wilson, C. H., & Hartline, D. K. (2011). Novel organization and development of copepod myelin. I. Ontogeny. The Journal of Comparative Neurology, 519, 3259–3280.
Xu, K., Sun, H., & Sung, Y. (1994). Electron microscopic observation on ontogenesis of the myelinated nerve fiber in the shrimp (Panaeus orientalis). Chinese Journal of Neuroanatomy, 10, 239–241.
Zalc, B., Goujet, D., & Colman, D. R. (2008). The origin of the myelination program in vertebrates. Current Biology, 18, R511–R512.
Acknowledgments
We thank Drs. Michael Hines and Ted Carnevale for providing the NEURON simulation software, and Drs. Andrew Christie and Petra Lenz for valuable criticism of earlier drafts. We are very appreciative of support provided by the National Science Foundation (Interdisciplinary Training for Undergraduates in Biological and Mathematical Science award NSF DUE-0634624, and NSF IOS-0923692), and The Cades Foundation, Honolulu, HI.
Author information
Authors and Affiliations
Corresponding author
Additional information
Action Editor: Catherine E. Carr
Rights and permissions
About this article
Cite this article
Young, R.G., Castelfranco, A.M. & Hartline, D.K. The “Lillie Transition”: models of the onset of saltatory conduction in myelinating axons. J Comput Neurosci 34, 533–546 (2013). https://doi.org/10.1007/s10827-012-0435-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-012-0435-3