Abstract
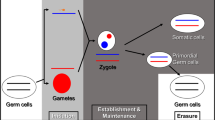
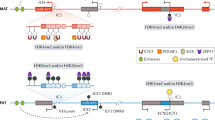
Mammals inherit two complete sets of chromosomes, one from the father and one from the mother, and most autosomal genes are expressed from both maternal and paternal alleles. Imprinted genes show expression from only one member of the gene pair (allele) and their expression are determined by the parent during production of the gametes. Imprinted genes represent only a small subset of mammalian genes that are present but not imprinted in other vertebrates. Genomic imprints are erased in both germlines and reset accordingly; thus, reversible depending on the parent of origin and leads to differential expression in the course of development. Genomic imprinting has been studied in humans since the early 1980’s and accounts for several human disorders. The first report in humans occurred in Prader-Willi syndrome due to a paternal deletion of chromosome 15 or uniparental disomy 15 (both chromosome 15s from only one parent) and similar genetic disturbances were reported later in Angelman syndrome.

Similar content being viewed by others
References
Butler MG, Palmer CG. Parental origin of chromosome 15 deletion in Prader-Willi syndrome. Lancet. 1983;1(8336):1285–6.
Nicholls RD, Knoll JH, Butler MG, Karam S, Lalande M. Genetic imprinting suggested by maternal heterodisomy in nondeletion Prader-Willi syndrome. Nature. 1989;342(6247):281–5.
Bartolomei MS, Tilghman SM. Genomic imprinting in mammals. Annu Rev Genet. 1997;31:493–525.
Walter J, Paulsen M. Imprinting and disease. Semin Cell Dev Biol. 2003;14:101–10.
Delaval K, Wagschal A, Feil R. Epigenetic deregulation of imprinting in congenital diseases of aberrant growth. Bioessays. 2006;28(5):453–9.
Platonov ES, Isaev DA. Genomic imprinting in the epigenetics of mammals. Genetika. 2006;42(9):1235–49.
Murphy SK, Jirtle RL. Imprinting evolution and the price of silence. Bioessays. 2003;25(6):577–88.
Haig D, Graham C. Genomic imprinting and the strange case of the insulin-like growth factor II receptor. Cell. 1991;64(6):1045–6.
Zhang Y, Tycko B. Monoallelic expression of the human H19 gene. Nat Genet. 1992;1(1):40–4.
Niemitz EL, Feinberg AP. Epigenetics and assisted reproductive technology: a call for investigation. Am J Hum Genet. 2004;74(4):599–609.
Luedi PP, Dietrich FS, Weidman JR, Bosko JM, Jirtle RL, Hartemink AJ. Computational and experimental identification of novel human imprinted genes. Genome Res. 2007;17(12):1723–30.
Butler MG. Imprinting disorders: non-Mendelian mechanisms affecting growth. J Pediatr Endocrinol Metab. 2002;15(Suppl 5):1279–88.
Falk MJ, Curtis CA, Bass NE, Zinn AB, Schwartz S. Maternal uniparental disomy chromosome 14: case report and literature review. Pediatr Neurol. 2005;32(2):116–20.
Temple K, Shrubb V, Lever M, Bullman H, Mackey DJG. Isolated imprinting mutation of the DLK1/GTL2 locus associated with a clinical presentation of maternal uniparental disomy of chromosome 14. J Med Genet. 2007;44:637–40.
Luedi PP, Hartemink AJ, Jirtle RL. Genome-wide prediction of imprinted murine genes. Genome Res. 2005;15(6):875–84.
Zakharova IS, Shevchenko AI, Zakian SM. Monoallelic gene expression in mammals. Chromosoma. 2009;118(3):279–90.
Eggermann T, Eggermann K, Schonherr N. Growth retardation versus overgrowth: Silver-Russell syndrome is genetically opposite to Beckwith-Wiedemann syndrome. Trends Genet. 2008;24(4):195–204.
Bliek J, Verde G, Callaway J, Maas SM, De Crescenzo A, Sparago A, et al. Hypomethylation at multiple maternally methylated imprinted regions including PLAGL1 and GNAS loci in Beckwith-Wiedemann syndrome. Eur J Hum Genet. 2009;17(5):611–9.
Barton SC, Surani MA, Norris ML. Role of paternal and maternal genomes in mouse development. Nature. 1984;311(5984):374–6.
McGrath J, Solter D. Inability of mouse blastomere nuclei transferred to enucleated zygotes to support development in vitro. Science. 1984;226(4680):1317–9.
Cattanach BM, Kirk M. Differential activity of maternally and paternally derived chromosome regions in mice. Nature. 1985;315(6019):496–798.
Cattanach BM, Beechey CV, Peters J. Interactions between imprinting effects: summary and review. Cytogenet Genome Res. 2006;113(1–4):17–23.
Willadsen SM, Janzen RE, McAlistre RJ. The viability of late morulae and blastocysts produced by nuclear transplantation in cattle. Theriogenology. 1991;35:161–70.
Walker SK, Hartwich KM, Seamark RF. The production of unusually large offspring following embryo manipulation: concepts and challenges. Theriogenology. 1996;45:111–20.
Kruip TAM, den Daas JHG. In vitro produced and cloned embryos: effects on pregnancy, parturition and offspring. Theriogenology. 1997;47:141–52.
Young LE, Fernandes K, McEvoy TG. Epigenetic change in IGF2R is associated with fetal overgrowth after sheep embryo culture. Nat Genet. 2001;27:153–4.
Doherty AS, Mann MR, Tremblay KD, Bartolomei MS, Schultz RM. Differential effects of culture on imprinted H19 expression in the preimplantation mouse embryo. Biol Reprod. 2000;62(6):1526–35.
DeBaun MR, Neimitz EL, Feinberg AP. Association of in vitro fertilization with Beckwith-Wiedemann syndrome and epigenetic alterations of LIT1 and H19. Am J Hum Genet. 2003;72:156–60.
Maher TR, Brueton LA, Bowdin SC. Beckwith-Wiedemann syndrome and assisted reproduction technology (ART). J Med Genet. 2003;40:62–4.
Schieve LA, Meikle SF, Ferre C, Peterson HB, Jeng G, Wilcox LS. Low and very low birth weight in infants conceived with use of assisted reproductive technology. N Engl J Med. 2002;346(10):731–7.
Manipalviratn S, DeCherney A, Segars J. Imprinting disorders and assisted reproductive technology. Fertil Steril. 2009;91(2):305–15.
Cox GF, Burger JL, Mau UA. Intracytoplasmic sperm injection may increase the risk of imprinting defects. Am J Hum Genet. 2002;71:162–4.
Moll AC, Imhof SM, Cruysberg JR. Schouten-van Meeteren AY, Boers M, van Leeuwen FE. Incidence of retinoblastoma in children born after in-vitro fertilization. Lancet. 2003;361(9354):309–10.
Butler MG. Prader-Willi syndrome: current understanding of cause and diagnosis. Am J Med Genet. 1990;35(3):319–32.
Butler MG, Thompson T. Prader-Willi syndrome: clinical and genetic finding. The Endocrinologist. 2000;10:3S–16.
Cassidy SB, Driscoll DJ. Prader-Willi syndrome. Eur J Hum Genet. 2009;17(1):3–13.
Bittel DC, Butler MG. Prader-Willi syndrome: clinical genetics, cytogenetics and molecular biology. Expert Rev Mol Med. 2005;7(14):1–20.
Prader A, Labhart A, Willi H. Ein syndrom von adipositas, kleinwuchs, kryptorchismus und oligophrenie nach myatonieartigem zustand im neugeborenenalter. Schweiz Med Wochenschr. 1956;86:1260–1.
Ledbetter DH, Riccardi VM, Airhart SD, Strobel RJ, Keenan BS, Crawford JD. Deletions of chromosome 15 as a cause of the Prader-Willi syndrome. N Engl J Med. 1981;304(6):325–9.
Butler MG, Lee PDK, Whitman BY. In: Butler MG, Lee PDK, Whitman BY, editors. Management of Prader-Willi syndrome. 3rd ed. New York: Springer-Verlag; 2006. p. 1–550.
Williams CA. Angelman syndrome. In: Butler MG, Meaney FJ, editors. Genetics of developmental disabilities. 1st ed. Boca Raton: Taylor & Francis; 2005. p. 319–36.
Butler MG, Fischer W, Kibiryeva N, Bittel DC. Array comparative genomic hybridization (aCGH) analysis in Prader-Willi syndrome. Am J Med Genet. 2008;146(7):854–60.
Butler MG, Bittel DC, Kibiryeva N, Talebizadeh Z, Thompson T. Behavioral differences among subjects with Prader-Willi syndrome and type I or type II deletion and maternal disomy. Pediatrics. 2004;113(3 Pt 1):565–73.
Nicholls RD, Knepper JL. Genome organization, function, and imprinting in Prader-Willi and Angelman syndromes. Annu Rev Genomics Hum Genet. 2001;2:153–75.
Butler MG. Prader-Willi syndrome: an example of genomic imprinting. In: Butler MG, Meaney FJ, editors. Genetics of developmental disabilities. 1st ed. Boca Raton: Taylor & Francis; 2005. p. 279–318.
Sahoo T, del Gaudio D, German JR, Shinawi M, Peters SU, Person RE, et al. Prader-Willi phenotype caused by paternal deficiency for the HBII-85 C/D box small nucleolar RNA cluster. Nat Genet. 2008;40(6):719–21.
Cassidy SB, Lai LW, Erickson RP, Magnuson L, Thomas E, Gendron R, et al. Trisomy 15 with loss of the paternal 15 as a cause of Prader-Willi syndrome due to maternal disomy. Am J Hum Genet. 1992;51(4):701–8.
Silver HK, Kiyasu W, George J, Deamer WC. Syndrome of congenital hemihypertrophy, shortness of stature, and elevated urinary gonadotropins. Pediatrics. 1953;12(4):368–76.
Russell A. A syndrome of intra-uterine dwarfism recognizable at birth with cranio-facial dysostosis, disproportionately short arms, and other anomalies (5 examples). Proc R Soc Med. 1954;47(12):1040–4.
Jones KL, ed. Smith’s recognizable patterns of human malformation. 6th ed. Philadelphia: W.B. Saunders Company; 2006. p. 1–954.
Abu-Amero S, Monk D, Frost J, Preece M, Stanier P, Moore GE. The genetic aetiology of Silver-Russell syndrome. J Med Genet. 2008;45(4):193–9.
Yoshihashi H, Maeyama K, Kosaki R, Ogata T, Tsukahara M, Goto Y, et al. Imprinting of human GRB10 and its mutations in two patients with Russell-Silver syndrome. Am J Hum Genet. 2000;67(2):476–82.
Bullman H, Lever M, Robinson DO, Mackay DJ, Holder SE, Wakeling EL. Mosaic maternal uniparental disomy of chromosome 11 in a patient with Silver-Russell syndrome. J Med Genet. 2008;45(6):396–9.
Wiedemann HR. Complex malformatif familial avec hernie ombilicale et macroglossie – un “syndrome nouveau”? J Genet Hum. 1964;13:223.
Beckwith JB. Macroglossia, Omphalocele, adrenal cytomegaly, gigantism, and hyperplasic visceromegaly. Birth Defects. 1969;5(2):188.
Pettenati MJ, Haines JL, Higgins RR, Wappner RS, Palmer CG, Weaver DD. Wiedemann-Beckwith syndrome: Presentation of clinical and cytogenetic data on 22 new cases and review of the literature. Hum Genet. 1986;74(2):143–54.
Viville M, Surani MA. Toward unraveling the Igf2/H19 imprinted domain. Bioessays. 1995;17(10):835–8.
Albright F, Burnett CH, Smith PH, Parson W. Pseudo-hypoparathyroidism-an example of ‘Seabright-Bantam syndrome’: report of three cases. Endocrinology. 1942;30:922–32.
Bastepe M. The GNAS locus and pseudohypoparathyroidism. Adv Exp Med Biol. 2008;626:27–40.
Bastepe M, Juppner H. GNAS locus and pseudohypoparathyroidism. Horm Res. 2005;63(2):65–74.
Fitch N. Albright’s hereditary osteodystrophy: a review. Am J Med Genet. 1982;11(1):11–29.
Levine MA. Clinical spectrum and pathogenesis of pseudohypoparathyroidism. Rev Endocr Metab Disord. 2000;1(4):265–74.
Wang JC, Passage MB, Yen PH, Shapiro LJ, Mohandas TK. Uniparental heterodisomy for chromosome 14 in a phenotypically abnormal familial balanced 13/14 Robertsonian translocation carrier. Am J Hum Genet. 1991;48(6):1069–74.
Temple IK, Cockwell A, Hassold T, Pettay D, Jacobs P. Maternal uniparental disomy for chromosome 14. J Med Genet. 1991;28(8):511–4.
Berends MJ, Hordijk R, Scheffer H, Oosterwijk JC, Halley DJ, Sorgedrager N. Two cases of maternal uniparental disomy 14 with a phenotype overlapping with the Prader-Willi phenotype. Am J Med Genet. 1999;84(1):76–9.
Cotter PD, Kaffe S, McCurdy LD, Jhaveri M, Willner JP, Hirschhorn K. Paternal uniparental disomy for chromosome 14: a case report and review. Am J Med Genet. 1997;70(1):74–9.
Acknowledgements
I thank Carla Meister for expert preparation of the manuscript. Partial funding support was provided from the NIH rare disease grant (1U54RR019478) and a grant from PWSA (USA).
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Disturbances in imprinted genes cause several human diseases involving neurological disorders, obesity, diabetes and malignancies with expression patterns of imprinted genes potentially influenced by the environment including assisted reproductive technology.
Rights and permissions
About this article
Cite this article
Butler, M.G. Genomic imprinting disorders in humans: a mini-review. J Assist Reprod Genet 26, 477–486 (2009). https://doi.org/10.1007/s10815-009-9353-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-009-9353-3