Abstract
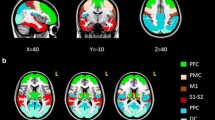
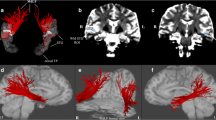
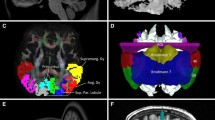
The subthalamic nucleus (STN) receives direct cortical inputs which constitute the so-called hyperdirect pathway. In monkeys, motor cortices innervate the whole extent of the STN whereas limbic cortices innervate only its anteromedial part extending more medially outside the nucleus. Tractography studies in humans have also identified motor cortical inputs to the STN, but little is known about the associative and limbic cortical projections. Therefore, the aim of this study was to investigate the anatomo-functional organization of the cortical projections to the STN and to the adjacent medial subthamic region (MSR). We used diffusion-weighted imaging-based tractography acquired from 30 subjects from the Human Connectome Project. We performed a whole-brain probabilistic tractography using MRTrix and extracted streamlines of interest between 39 cortical masks and both the STN and the MSR to provide track-density maps. Agglomerative clustering method was used to classify the voxels of the regions of interest. We found that the STN receives major inputs from the sensorimotor cortices and few inputs from the limbic cortices. On the other hand, the MSR receives mainly cortical limbic projections and few from the sensorimotor cortices. Weak connections were found between the associative cortices and both the STN and the MSR. We found a dominant motor cluster located in the posterolateral STN, a limbic cluster located medially in the MSR, and an intermediate motor-limbic cluster in between. Our findings show that the hyperdirect pathway is anatomo-functionally organized with a poor participation of associative cortices.






Similar content being viewed by others
Abbreviations
- BA:
-
Brodmann areas
- DBS:
-
Deep brain stimulation
- DWI:
-
Diffusion-weighted imaging
- FA:
-
Fractional anisotropy
- FOD:
-
Fiber orientation distributions
- MSR:
-
Medial subthalamic region
- MR:
-
Magnetic resonance
- MRI:
-
Magnetic resonance imaging
- PD:
-
Parkinson’s disease
- ROI:
-
Region of interest
- STN:
-
Subthalamic nucleus
- LH:
-
Left hemisphere
- RH:
-
Right hemisphere
References
Accolla EA, Dukart J, Helms G, Weiskopf N, Kherif F, Lutti A, Chowdhury R, Hetzer S, Haynes JR, Kühn AA, Draganski B (2014) Brain tissue properties differentiate between motor and limbic basal ganglia circuits. Hum Brain Mapp 35(10):5083–5092
Afsharpour S (1985) Topographical projections of the cerebral cortex to the subthalamic nucleus. J Comp Neurol 236:14–28
Akram H, Miller S, Lagrata S, Hariz M, Ashburner J, Behrens T, Matharu M, Zrinzo L (2017a) Optimal deep brain stimulation site and target connectivity for chronic cluster headache. Neurology 89(20):2083–2091. https://doi.org/10.1212/WNL.0000000000004646
Akram H, Sotiropoulos SN, Jbabdi S, Georgiev D, Mahlknecht P, Hyam J, Foltynie T, Limousin P, De Vita E, Jahanshahi M, Hariz M, Ashburner J, Behrens T, Zrinzo L (2017b) Subthalamic deep brain stimulation sweet spots and hyperdirect cortical connectivity in Parkinson’s disease. NeuroImage 158:332–345. https://doi.org/10.1016/j.neuroimage.2017b.07.012
Alkemade A, Forstmann BU (2014) Do we need to revise the tripartite subdivision hypothesis of the human subthalamic nucleus (STN)? NeuroImage 95:326–329
Andersson JL, Sotiropoulos SN (2015) Non-parametric representation and prediction of single- and multi-shell diffusion-weighted MRI data using Gaussian processes. NeuroImage 122:166–176
Aravamuthan BR, Muthusamy KA, Stein JF, Aziz TZ, Johansen-Berg H (2007) Topography of cortical and subcortical connections of the human pedunculopontine and subthalamic nuclei. NeuroImage 37:694–705. https://doi.org/10.1016/j.neuroimage.2007.05.050
Aron AR, Behrens TE, Smith S, Frank MJ, Poldrack RA (2007) Triangulating a cognitive control network using diffusion-weighted magnetic resonance imaging (MRI) and functional MRI. J Neurosci 27(14):3743–3752
Avecillas-Chasin JM, Rascón-Ramírez F (2016) Tractographical model of the cortico-basal ganglia and corticothalamic connections: improving our understanding of deep brain stimulation. Clin Anat 29(4):481–492
Bardinet E, Bhattacharjee M, Dormont D, Pidoux B, Malandain G, Schüpbach M, Ayache N, Cornu P, Agid Y, Yelnik J (2009) A three-dimensional histological atlas of the human basal ganglia. II. Atlas deformation strategy and evaluation in deep brain stimulation for Parkinson disease. J Neurosurg 110:208–219. https://doi.org/10.3171/2008.3.17469
Bejjani BP, Houeto JL, Hariz M, Yelnik J, Mesnage V, Bonnet AM, Pidoux B, Dormont D, Cornu P, Agid Y (2002) Aggressive behavior induced by intraoperative stimulation in the triangle of Sano. Neurology 59(9):1425–1427
Benabid AL, Chabardes S, Mitrofanis J, Pollak P (2000) Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol 8(1):67–81. https://doi.org/10.1016/S1474-4422(08)70291-6
Brunenberg EJ, Moeskops P, Backes WH, Pollo C, Cammoun L, Vilanova A, Janssen ML, Visser-Vandewalle VE, ter Haar Romeny BM, Thiran JP, Platel B (2012) Structural and resting state functional connectivity of the subthalamic nucleus: identification of motor STN parts and the hyperdirect pathway. PLoS One 7(6):e39061
Calamante F, Tournier JD, Jackson GD, Connelly A (2010) Track-density imaging (TDI): super-resolution white matter imaging using whole-brain track-density mapping. NeuroImage 53:1233–1243
Canteras NS, Shammah-Lagnado SJ, Silva BA, Ricardo JA (1990) Afferent connections of the subthalamic nucleus: a combined retrograde and anterograde horseradish peroxidase study in the rat. Brain Res 513:43–59
Chabardès S, Polosan M, Krack P, Bastin J, Krainik A, David O, Bougerol T, Benabid AL (2013) Deep brain stimulation for obsessive-compulsive disorder: subthalamic nucleus target. World Neurosurg 80(3–4):S31.e1-8. https://doi.org/10.1016/j.wneu.2012.03.010
Christiaens D, Reiser M, Dhollander T, Sunaert S, Suetens P, Global Maes F (2015) tractography of multi shell diffusion weighted imaging data using a multi-tissue model. NeuroImage 123:89–101
Coenen VA, Schlaepfer TE, Maedler B, Panksepp J (2011) Cross-species affective functions of the medial forebrain bundle-implications for the treatment of affective pain and depression in humans. Neurosci Biobehav Rev 35(9):1971–1981. https://doi.org/10.1016/j.neubiorev.2010.12.009
Coenen VA, Schumacher LV, Kaller C, Schlaepfer TE, Reinacher PC, Egger K, Urbach H, Reisert M (2018) The anatomy of the human medial forebrain bundle: ventral tegmental area connections to reward-associated subcortical and frontal lobe regions. Neuroimage Clin 18:770–783. https://doi.org/10.1016/j.nicl.2018.03.019
Coudé D, Parent A, Parent M (2018) Single-axon tracing of the corticosubthalamic hyperdirect pathway in primates. Brain Struct Funct 223(9):3959–3973. https://doi.org/10.1007/s00429-018-1726-x
Crossman AR, Sambrook MA, Jackson A (1984) Experimental hemichorea/hemiballismus in the monkey. Studies on the intracerebral site of action in a drug-induced dyskinesia. Brain 107(Pt 2):579–596
Dhollander T, Raffelt D, Connelly A (2016) Unsupervised 3-tissue response function estimation from single-shell or multi-shell diffusion MR data without a co-registered T1 image. ISMRM Workshop on Breaking the Barriers of Diffusion MRI, 5
Forel A (1877) Untersuchungen über die Haubenregion und ihre oberen Verknüpfungen im Gehirne des Menschen und einiger Säugethiere, mit Beiträgen zu den Methoden der Gehirnuntersuchung. Archiv für Psychiatrie und Nervenkrankheiten. Springer-Verlag 7(3):393–495
François C, Grabli D, McCairn K, Jan C, Karachi C, Hirsch EC, Féger J, Tremblay L (2004) Behavioural disorders induced by external globus pallidus dysfunction in primates II. Anatomical study. Brain 127(Pt 9):2055–2070
Franzini A, Ferroli P, Leone M, Broggi G (2003) Stimulation of the posterior hypothalamus for treatment of chronic intractable cluster headaches: first reported series. Neurosurgery 52(5):1095–1099
Franzini A, Broggi G, Cordella R, Dones I, Messina G (2013) Deep-brain stimulation for aggressive and disruptive behavior. World Neurosurg 80(3–4):S29
Gan G, Ma C, Wu J (2007) Data clustering: theory, algorithms, and applications. Vol 20 Siam
Glasser MF, Sotiropoulos SN, Wilson JA, Coalson TS, Fischl B, Andersson JL, Xu J, Jbabdi S, Webster M, Polimeni JR, Van Essen DC, Jenkinson M (2013) The minimal preprocessing pipelines for the Human Connectome Project. Neuroimage 80:105–124. https://doi.org/10.1016/j.neuroimage.2013.04.127
Goto M, Swanson LW (2004) Axonal projections from the parasubthalamic nucleus. J Comp Neurol 169(4):581–607
Haber SN, Lynd-Balta E, Mitchell SJ (1993) The organization of the descending ventral pallidal projections in the monkey. J Comp Neurol 329(1):111–128
Haber SN, Fudge JL, McFarland NR (2000) Striatonigrostriatal pathways in primates form an ascending spiral from the shell to the dorsolateral striatum. J Neurosci 20(6):2369–2382
Hammond C, Yelnik J (1983) Intracellular labelling of rat subthalamic neurones with horseradish peroxidase: computer analysis of dendrites and characterization of axon arborization. Neuroscience 8(4):781–790
Hartmann-von Monakow K, Akert K, Künzle H (1978) Projections of the precentral motor cortex and other cortical areas of the frontal lobe to the subthalamic nucleus in the monkey. Exp Brain Res 33:395–403
Haynes W, Haber S (2013) The organization of prefrontal subthalamic inputs in primates provides an anatomical substrate for both functional specificity and integration: implications for basal ganglia models and deep brain stimulation. J Neurosci 33:4804–4814
Jbabdi S, Sotiropoulos SN, Haber SN, Van Essen DC, Behrens TE (2015) Measuring macroscopic brain connections in vivo. Nat Neurosci 18(11):1546–1555. https://doi.org/10.1038/nn.4134Epub 2015 Oct 27
Jeurissen B, Tournier JD, Dhollander T, Connelly A, Sijbers J (2014) Multi-tissue constrained spherical deconvolution for improved analysis of multi-shell diffusion MRI data. Neuroimage 103:411–426
Jürgens U (1984) The efferent and afferent connections of the supplementary motor area. Brain Res 300:63–81
Karachi C, Yelnik J, Tandé D, Tremblay L, Hirsch E, François C (2005) The pallido- subthalamic projection: an anatomical substrate for nonmotor functions of the subthalamic nucleus in primates. Mov Disord 20(2):172–180
Karachi C, Grabli D, Baup N, Mounayar S, Tandé D, François C, Hirsch EC (2009) Dysfunction of the subthalamic nucleus induces behavioral and movement disorders in monkeys. Mov Disord 24(8):1183–1192. https://doi.org/10.1002/mds.22547
Kim JS, Kim HJ, Lee JY, Kim JM, Yun JY, Jeon BS (2012) Hypomania induced by subthalamic nucleus stimulation in a Parkinson’s disease patient: does it suggest a dysfunction of the limbic circuit? J Mov Disord 5(1):14–17. https://doi.org/10.14802/jmd.12004
Künzle H (1975) Bilateral projections from precentral motor cortex to the putamen and other parts of the basal ganglia. An autoradiographic study in Macaca fascicularis. Brain Res 88:195–209
Künzle H (1978) An autoradiographic analysis of the efferent connections from premotor and adjacent prefrontal regions (areas 6 and 9) in Macaca fascicularis. Brain Behav Evol 15:185–234
Künzle H, Akert K (1977) Efferent connections of cortical, area 8 (frontal eye field) in Macaca fascicularis. A reinvestigation using the autoradiographic technique. J Comp Neurol 173:147–164
Lambert C, Zrinzo L, Nagy Z, Lutti A, Hariz M, Foltynie T, Draganski B, Ashburner J, Frackowiak R (2012) Confirmation of functional zones within the human subthalamic nucleus: patterns of connectivity and sub-parcellation using diffusion weighted imaging. Neuroimage 60:83–94. https://doi.org/10.1016/j.neuroimage.2011.11.082
Lambert C, Zrinzo L, Nagy Z, Lutti A, Hariz M, Foltynie T, Draganski B, Ashburner J, Frackowiak R (2015) Do we need to revise the tripartite subdivision hypothesis of the human subthalamic nucleus (STN)? Response to Alkemade and Forstmann. Neuroimage 110:1–2. https://doi.org/10.1016/j.neuroimage.2015.01.038
Leone M, Franzini A, Bussone G (2001) Stereotactic stimulation of posterior hypothalamic gray matter in a patient with intractable cluster headache. N Engl J Med 345(19):1428–1429
Limousin P, Krack P, Pollak P, Benazzouz A, Ardouin C, Hoffman D, Benabid AL (1998) Electrical stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N Engl J Med 339:1105–1111
Maier-Hein KH, Neher PF, Houde JC, Côté MA, Garyfallidis E, Zhong J, Chamberland M, Descoteaux M (2017) The challenge of mapping the human connectome based on diffusion tractography. Nat Commun 8:1349. https://doi.org/10.1038/s41467-017-01285-x
Mallet L, Schüpbach M, N’Diaye K, Remy P, Bardinet E, Czernecki V, Welter ML, Pelissolo A, Ruberg M, Agid Y, Yelnik J (2007) Stimulation of subterritories of the subthalamic nucleus reveals its role in the integration of the emotional and motor aspects of behavior. Proc Natl Acad Sci USA 104(25):10661–10666
Mallet L, Polosan M, Jaafari N, Baup N, Welter ML, Fontaine D, du Montcel ST, Yelnik J, Chéreau I, Arbus C, Raoul S, Aouizerate B, Damier P, Chabardès S, Czernecki V, Ardouin C, Krebs MO, Bardinet E, Chaynes P, Burbaud P, Cornu P, Derost P, Bougerol T, Bataille B, Mattei V, Dormont D, Devaux B, Vérin M, Houeto JL, Pollak P, Benabid AL, Agid Y, Krack P, Millet B, Pelissolo A, STOC Study Group (2008) Subthalamic nucleus stimulation in severe obsessive-compulsive disorder. N Engl J Med 359(20):2121–2134
Maurice N, Deniau JM, Glowinski J, Thierry A (1998) Relationships between the prefrontal cortex and the basal ganglia in the rat: physiology of the corticosubthalamic circuits. J Neurosci 18(22):9539–9546
Mulder MJ, Boekel W, Ratcliff R, Forstmann BU (2014) Cortico-subthalamic connection predicts individual differences in value-driven choice bias. Brain Struct Funct 219(4):1239–1249
Nambu A, Takada M, Inase M, Tokuno H (1996) Dual somatotopical representations in the primate subthalamic nucleus: evidence for ordered but reversed body-map transformations from the primary motor cortex and the supplementary motor area. J Neurosci 16(8):2671–2683
Nambu A, Tokuno H, Inase M, Takada M (1997) Corticosubthalamic input zones from forelimb representations of the dorsal and ventral divisions of the premotor cortex in the macaque monkey: comparison with the input zones from the primary motor cortex and the supplementary motor area. Neurosci Lett 239:13–16
Neudorfer C, Maarouf M (2018) Neuroanatomical background and functional considerations for stereotactic interventions in the H fields of Forel. Brain Struct Funct 223(1):17–30. https://doi.org/10.1007/s00429-017-1570-4
Parent A, Hazrati LN (1995) Functional anatomy of the basal ganglia. II. The place of subthalamic nucleus and external pallidum in basal ganglia circuitry. Brain Res Rev 20(1):128–154
Petersen MV, Lund TE, Sunde N, Frandsen J, Rosendal F, Juul N, Østergaard K (2016) Probabilistic versus deterministic tractography for delineation of the cortico-subthalamic hyperdirect pathway in patients with Parkinson disease selected for deep brain stimulation. J Neurosurg 126(5):1657–1668
Rizzi M, Trezza A, Messina G, De Benedictis A, Franzini A, Marras CE (2017) Exploring the brain through posterior hypothalamus surgery for aggressive behavior. Neurosurg Focus 43(3):E14. https://doi.org/10.3171/2017.6.FOCUS17231
Rizzolatti G, Fogassi L, Gallese V (2002) Motor and cognitive functions of the ventral premotor cortex. Curr Opin Neurobiol 12(2):149–154
Sano K (1962) Sedative neurosurgery with special reference to posteromedial hypothalamotomy. Neurol Me Chi (Tokyo) 4:112–142
Sano K, Yoshioka M, Ogashiwa M, Ishijima B, Ohye C (1966) Postero-medial hypothalamotomy in the treatment of aggressive behaviors. Confinia neurol 27:164–167
Sano K, Mayanagi Y, Sekino H, Ogashiwa M, Ishijima B (1970) Results of stimulation and destruction of the posterior hypothalamus in man. J Neurosurg 33(6):689–707
Sébille SB, Belaid H, Philippe AC, André A, Lau B, François C, Karachi C, Bardinet E (2017) Anatomical evidence for functional diversity in the mesencephalic locomotor region of primates. Neuroimage 147:66–78. https://doi.org/10.1016/j.neuroimage.2016.12.011
Shink E, Bevan MD, Bolam JP, Smith Y (1996) The subthalamic nucleus and the external pallidum: two tightly interconnected structures that control the output of the basal ganglia in the monkey. Neuroscience 73:335–357
Shook BL, Schlag-Rey M, Schlag J (1991) Primate supplementary eye field. II. Comparative aspects of connections with the thalamus, corpus striatum, and related forebrain nuclei. J Comp Neurol 307:562–583
Sinke M, Otte W, Christiaens D, Schmitt O, Leemans A, Toorn A, Sarabdjitsingh A, Joels M, Dijkhuizen R (2018) Diffusion MRI-based cortical connectome reconstruction: dependency on tractography procedures and neuroanatomical characteristics. Brain Struct Func 223(5):2269–2285
Smith RE, Tournier JD, Calamante E, Connelly A (2013) SIFT: spherical-deconvolution informed filtering of tractograms. NeuroImage 67:298–312
Sotiropoulos SN, Jbabdi S, Xu J, Andersson JL, Moeller S, Auerbach EJ, Glasser MF, Hernandez M, Sapiro G, Jenkinson M, Feinberg DA, Yacoub E, Lenglet C, Van Essen DC, Ugurbil K, Behrens TEJ (2013) Advances in diffusion MRI acquisition and processing in the Human Connectome Project. Neuroimage 80:125–143. https://doi.org/10.1016/j.neuroimage.2013.05.057
Takada M, Tokuno H, Hamada I, Inase M, Ito Y, Imanishi M, Hasegawa N, Akazawa T, Hatanaka N, Nambu A (2001) Organization of inputs from cingulate motor areas to basal ganglia in macaque monkey. Eur J Neurosci 14(10):1633–1650
Tournier JD, Calamante F, Connelly A (2010) Improved probabilistic streamlines tractography by 2nd order integration over fibre orientation distributions. Proc Intl Soc Mag Reson Med (ISMRM) 18:1670
Tournier JD, Calamante F, Connelly A (2012) MRtrix: diffusion tractography in crossing fiber regions. Int J Imaging Syst Technol 22:53–66. https://doi.org/10.1002/ima.22005
Ugurlu D, Firat Z, Ture U, Unal G (2018) Neighborhood resolved fiber orientation distributions (NRFOD) in automatic labeling of white matter fiber pathways. Med Image Anal 46:130–145
Van Essen DC, Smith SM, Barch DM, Behrens TEJ, Yacoub E, Ugurbil K (2013) The WU-minn human connectome project: an overview. Neuroimage 80:62–79. https://doi.org/10.1016/j.neuroimage.2013.05.041
Veazey RB, Amaral DG, Cowan WM (1982) The morphology and connections of the posterior hypothalamus in the cynomolgus monkey (Macaca fascicularis). II. Efferent connections. J Comp Neurol 207(2):135–156
Wang L, Mruczek EB, Arcaro MJ, Kastner S (2018) Probabilistic maps of visual topography in human cortex. Cereb Cortex 25:3911–3931
Welter ML, Schüpbach M, Czernecki V, Karachi C, Fernandez-Vidal S, Golmard JL, Serra G, Navarro S, Welaratne A, Hartmann A, Mesnage V, Pineau F, Cornu P, Pidoux B, Worbe Y, Zikos P, Grabli D, Galanaud D, Bonnet AM, Belaid H, Dormont D, Vidailhet M, Mallet L, Houeto JL, Bardinet E, Yelnik J, Agid Y (2014) Optimal target localization for subthalamic stimulation in patients with Parkinson disease. Neurology 82(15):1352–1361
Wilkins B, Lee N, Gajawelli N, Law M, Leporé N (2015) Fiber estimation and tractography in diffusion MRI: development of simulated brain images and comparison of multi-fiber analysis methods at clinical b-values. Neuroimage 109:341–356
Acknowledgements
This work was supported by ‘Investissements d’avenir’ (Investing in the future programme ANR-10-IAIHU-O6). G. Temiz was supported by the “Fondation pour la Recherche” Médicale (FRM) (Project:DIC20161236441) and by Boston Scientific. The authors would like to thank Max Westby for language editing.
Funding
This work was supported by ‘Investissements d’avenir’ (Investing in the future programme ANR-10-IAIHU-O6). G. Temiz was supported by the “Fondation pour la Recherche” Médicale (FRM) (Project:DIC20161236441) and by Boston Scientific.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no other conflicts of interest
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Temiz, G., Sébille, S.B., Francois, C. et al. The anatomo-functional organization of the hyperdirect cortical pathway to the subthalamic area using in vivo structural connectivity imaging in humans. Brain Struct Funct 225, 551–565 (2020). https://doi.org/10.1007/s00429-019-02012-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-02012-6