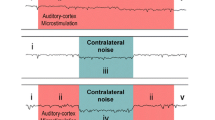
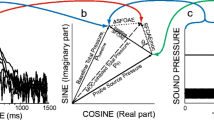
Abstract
Medial olivocochlear (MOC) neurons project to outer hair cells (OHC), forming the efferent arm of a reflex that affects sound processing and offers protection from acoustic overstimulation. The central pathways that trigger the MOC reflex in response to sound are poorly understood. Insight into these pathways can be obtained by examining the responses of single MOC neurons recorded from anesthetized guinea pigs. Response latencies of MOC neurons are as short as 5 ms. This latency is consistent with the idea that type I, but not type II, auditory-nerve fibers provide the major inputs to the reflex interneurons in the cochlear nucleus. This short latency also implies that the cochlear-nucleus interneurons have rapidly conducting axons. In the cochlear nucleus, lesions of the posteroventral subdivision (PVCN), but not the anteroventral (AVCN) or dorsal (DCN) subdivisions, produce permanent disruption of the MOC reflex, based on a metric of adaptation of the distortion-product otoacoustic emission (DPOAE). This finding supports earlier anatomical results demonstrating that some PVCN neurons project to MOC neurons. Within the PVCN, there are two general types of units when classified according to poststimulus time histograms: onset units and chopper units. The MOC response is sustained and cannot be produced solely by inputs having an onset pattern. The MOC reflex interneurons are thus likely to be chopper units of PVCN. Also supporting this conclusion, chopper units and MOC neurons both have sharp frequency tuning. Thus, the most likely pathway for the sound-evoked MOC reflex begins with the responses of hair cells, proceeds with type I auditory-nerve fibers, PVCN chopper units, and MOC neurons, and ends with the MOC terminations on OHC.





Similar content being viewed by others
References
Adams JC (1996) Axon terminals on olivocochlear neurons. Soc Neurosci Abstr 22:127
Borg E (1973) On the neuronal organization of the acoustic middle ear reflex. A physiological and anatomical study. Brain Res 49:101–123
Boyev K, Liberman M, Brown M (2002) Effects of anesthesia on efferent-mediated adaptation of the DPOAE. J Assoc Res Otolaryngol 3:362–373
Brawer JR, Morest DK, Kane EC (1974) The neuronal architecture of the cochlear nucleus of the cat. J Comp Neurol 155:251–300
Brown MC (1989) Morphology and response properties of single olivocochlear fibers in the guinea pig. Hearing Res 40:93–110
Brown MC (1993) Anatomical and physiological studies of type I and type II spiral ganglion neurons. In: Merchan MA, Juiz JM, Godfrey DA, Mugnaini E (eds) The mammalian cochlear nuclei: organization and function. Plenum, New York, pp 43–54
Brown MC (1994) Antidromic responses of single units from the spiral ganglion. J Neurophysiol 71:1835–1847
Brown MC (2001) Response adaptation of medial olivocochlear neurons is minimal. J Neurophysiol 86:2381–2392
Brown MC, Kujawa SG, Duca ML (1998) Single olivocochlear neurons in the guinea pig. I. Binaural facilitation of responses to high-level noise. J Neurophysiol 79:3077–3087
Cant NB, Gaston KC (1982) Pathways connecting the right and left cochlear nuclei. J Comp Neurol 212:313–326
de Venecia RK, Liberman MC, Guinan JJ Jr, Brown MC (2001) Effects of kainate lesions in different cochlear nucleus regions on the MOC reflex. Abstr Assoc Res Otolaryngol 46
Doucet JR, Ryugo DK (2002) Projections of multipolar neurons in the ventral cochlear nucleus to the lateral superior olive in rats. Abstr Assoc Res Otolaryngol
Evans EF (1972) The frequency response and other properties of single fibres in the guinea-pig cochlear nerve. J Physiol (Lond) 226:263–287
Faye-Lund H (1986) Projection from the inferior colliculus to the superior olivary complex in the albino rat. Anat Embryol 175:35–52
Fekete DM, Rouiller EM, Liberman MC, Ryugo DK (1984) The central projections of intracellularly labeled auditory nerve fibers in cats. J Comp Neurol 229:432–450
Fex J (1962) Auditory activity in centrifugal and centripetal cochlear fibers in cat. Acta Physiol Scand 55: S1891–68
Fex J (1965) Auditory activity in uncrossed centrifugal cochlear fibers in cat. Acta Physiol Scand 64:43–57
Friauf E, Ostwald J (1988) Divergent projections of physiologically characterized rat ventral cochlear nucleus neurons as shown by intra-axonal injection of horseradish peroxidase. Exp Brain Res 73:263–284
Godfrey DA, Kiang NYS, Norris BE (1975) Single unit activity in the posteroventral cochlear nucleus of the cat. J Comp Neurol 162:247–268
Guinan JJ Jr (1996) The physiology of olivocochlear efferents. In: Dallos P, Popper AN, Fay RR (eds) The cochlea. Springer, New York, pp 435–502
Guinan JJ Jr, Gifford ML (1988) Effects of electrical stimulation of efferent olivocochlear neurons on cat auditory-nerve fibers. I. Rate-level functions. Hearing Res 33:97–114
Gummer M, Yates GK, Johnstone BM (1988) Modulation transfer functions of efferent neurones in the guinea pig cochlea. Hearing Res 36:41–52
Hackney CM, Osen KK, Kolston J (1990) Anatomy of the cochlear nuclear complex of the guinea pig. Anat Embryol 182:123–149
Ito J, Honjo I (1988) Electrophysiological and HRP studies of the direct afferent inputs from the cochlear nuclei to the tensor tympani muscle motoneurons in the cat. Acta Otolaryngol 105:292–296
Itoh K, Nomura S, Konishi A, Yasui Y, Sugimoto T, Muzion N (1986) A morphological evidence of direct connections from the cochlear nuclei to tensor tympani motoneurons in the cat: a possible afferent limb of the acoustic middle ear reflex pathways. Brain Res 375:214–219
Jiang D, Palmer AR, Winter IM (1996) Frequency extent of two-tone facilitation in onset units in the ventral cochlear nucleus. J Neurophysiol 75:380–395
Kawase T, Delgutte B, Liberman MC (1993) Antimasking effects of the olivocochlear reflex. II. Enhancement of auditory-nerve response to masked tones. J Neurophysiol 70:2533–2549
Kiang NYS, Rho JM, Northrop CC, Liberman MC, Ryugo DK (1982) Hair-cell innervation by spiral ganglion cells in adult cats. Science 217:175–177
Kim DO (1986) Active and nonlinear cochlear biomechanics and the role of outer-hair-cell subsystem in the mammalian auditory system. Hearing Res 22:105–114
Kobler JB, Vacher SR, Guinan JJ Jr (1987) The recruitment order of stapedius motoneurons in the acoustic reflex varies with sound laterality. Brain Res 425:372–375
Kobler JB, Guinan JJ Jr, Vacher SR, Norris BE (1992) Acoustic reflex frequency selectivity in single stapedius motoneurons of the cat. J Neurophysiol 68:807–817
Kujawa S, Liberman MC (2001) Effects of olivocochlear feedback on distortion product otoacoustic emissions in guinea pig. J Assoc Res Otolaryngol 2:268–278
Liberman MC (1988) Response properties of cochlear efferent neurons: Monaural vs binaural stimulation and the effects of noise. J Neurophysiol 60:1779–1798
Liberman MC, Brown MC (1986) Physiology and anatomy of single olivocochlear neurons in the cat. Hearing Res 24:17–36
Liberman MC, Puria S, Guinan JJ Jr (1996) The ipsilaterally evoked olivocochlear reflex causes rapid adaptation of the 2f1–f2 distortion product otoacoustic emission. J Acoust Soc Am 99:3572–3584
McCue MP, Guinan JJ Jr (1988) Anatomical and functional segregation in the stapedius motoneuron pool of the cat. J Neurophysiol 60:1160–1180
Melcher JR, Knudson IM, Fullerton BC, Guinan JJ Jr, Norris BE, Kiang NYS (1996) Generators of the brainstem auditory evoked potential in cat I: An experimental approach to their identification. Hearing Res 93:1-27
Mugnaini E, Warr WB, Osen KK (1980) Distribution and light microscopic features of granule cells in the cochlear nucleus of cat, rat, and mouse. J Comp Neurol 191:581–606
Mulders WHAM, Robertson D (2000a) Evidence for direct cortical innervation of medial olivocochlear neurones in rats. Hearing Res 144:65–72
Osen KK (1969) Cytoarchitecture of the cochlear nuclei in the cat. J Comp Neurol 136:453–484
Rajan R (1988) Effect of electrical stimulation of the crossed olivocochlear bundle on temporary threshold shifts in auditory sensitivity. I. Dependence on electrical stimulation parameters. J Neurophysiol 60:549–568
Reiter ER, Liberman MC (1995) Efferent mediated protection from acoustic overexposure: relation to “slow” effects of olivocochlear stimulation. J Neurophysiol 73:506–514
Rhode WS, Greenberg S (1992) Physiology of the cochlear nuclei. In: Popper AN, Fay RR (eds) The mammalian auditory pathway: neurophysiology. Springer, New York, pp 94–152
Rhode WS, Oertel D, Smith PH (1983) Physiological response properties of cells labeled intracellularly with horseradish peroxidase in cat ventral cochlear nucleus. J Comp Neurol 213:448–463
Rhode WW, Kettner RE (1987) Physiological study of neurons in the dorsal and posteroventral cochlear nucleus of the unanesthetized cat. J Neurophysiol 57:414–442
Robertson D (1984) Horseradish peroxidase injection of physiologically characterized afferent and efferent neurons in the guinea pig spiral ganglion. Hearing Res 15:113–121
Robertson D, Gummer M (1985) Physiological and morphological characterization of efferent neurons in the guinea pig cochlea. Hearing Res 20:63–77
Robertson D, Winter IM (1988) Cochlear nucleus inputs to olivocochlear neurones revealed by combined anterograde and retrograde labelling in the guinea pig. Brain Res 462:47–55
Robertson D, Sellick PM, Patuzzi R (1999) The continuing search for outer hair cell afferents in the guinea pig spiral ganglion. Hearing Res 136:151–158
Rouiller EM, Ryugo DK (1984) Intracellular marking of physiologically characterized cells in the ventral cochlear nucleus of the cat. J Comp Neurol 225:167–186
Rouiller EM, Capt M, Dolivo M, Ribaupierre F de (1986) Tensor tympani reflex pathways studied with retrograde horseradish peroxidase and transneuronal viral tracing techniques. Neurosci Lett 72:247–252
Schofield BR, Cant NB (1996) Origins and targets of commissural connections between the cochlear nuclei in guinea pigs. J Comp Neurol 375:128–146
Spoendlin H (1971) Degeneration behaviour of the cochlear nerve. Arch Klin Exp Ohren Nasen Kehlkopfheilkd 200:275–291
Thompson AM, Thompson GC (1991) Posteroventral cochlear nucleus projections to olivocochlear neurons. J Comp Neurol 303:267–285
Vacher SR, Guinan JJ Jr, Kober JB (1989) Intracellularly labeled stapedius-motoneuron cell bodies in the cat are spatially organized according to their physiologic responses. J Comp Neurol 289:401–415
Warr WB (1969) Fiber degeneration following lesions in the posteroventral cochlear nucleus of the cat. Exp Neurol 23:140–155
Warr WB (1992) Organization of olivocochlear efferent systems in mammals. In: Webster DB, Popper AN, Fay RR (eds) The mammalian auditory pathway: neuroanatomy. Springer, New York, pp 410–448
Wenthold RJ (1987) Evidence for a glycinergic pathway connecting the two cochlear nuclei: an immunohistochemical and retrograde transport study. Brain Res 415:183–187
Wiederhold ML, Kiang NYS (1970) Effects of electric stimulation of the crossed olivocochlear bundle on single auditory-nerve fibers in the cat. J Acoust Soc Am 48:950–965
Winslow R, Sachs MB (1987) Effect of electrical stimulation of the crossed olivocochlear bundle on auditory nerve response to tones in noise. J Neurophysiol 57:1002–1021
Ye Y, Machado DG, Kim DO (2000) Projection of the marginal shell of the anteroventral cochlear nucleus to olivocochlear neurons in the cat. J Comp Neurol 420:127–138
Young ED, Robert J-M, Shofner WP (1988) Regularity and latency of units in ventral cochlear nucleus: implications for unit classification and generation of response properties. J Neurophysiol 60:1-29
Acknowledgements
We thank Dr. M. C. Liberman for comments on the manuscript and Dr. Wen Xu for technical assistance. Supported by NIDCD grants DC 01089 and DC 0119, and a grant from the Triological Society.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brown, M.C., de Venecia, R.K. & Guinan, J.J. Responses of medial olivocochlear neurons. Exp Brain Res 153, 491–498 (2003). https://doi.org/10.1007/s00221-003-1679-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1679-y