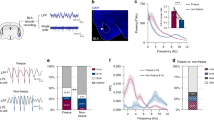
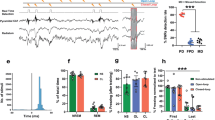
Abstract
The hippocampus plays a central role as a hub for episodic memory and as an integrator of multimodal sensory information in time and space. Thereby, it critically determines contextual setting and specificity of episodic memories. It is also a key site for the control of innate anxiety states and involved in psychiatric diseases with heightened anxiety and generalized fear memory such as post-traumatic stress disorder (PTSD). Expression of both innate “unlearned” anxiety and “learned” fear requires contextual processing and engagement of a brain-wide network including the hippocampus together with the amygdala and medial prefrontal cortex. Strikingly, the hippocampus is also the site of emergence of oscillatory rhythms that coordinate information processing and filtering in this network. Here, we review data on how the hippocampal network oscillations and their coordination with amygdalar and prefrontal oscillations are engaged in innate threat evaluation. We further explore how such innate oscillatory communication might have an impact on contextualization and specificity of “learned” fear. We illustrate the partial overlap of fear and anxiety networks that are built by the hippocampus in conjunction with amygdala and prefrontal cortex. We further propose that (mal)-adaptive interplay via (dis)-balanced oscillatory communication between the anxiety network and the fear network may determine the strength of fear memories and their resistance to extinction.




Similar content being viewed by others
References
Abel T, Lattal KM (2001) Molecular mechanisms of memory acquisition, consolidation and retrieval. Curr Opin Neurobiol 11:180–187. https://doi.org/10.1016/S0959-4388(00)00194-X
Adhikari A (2014) Distributed circuits underlying anxiety. Front Behav Neurosci 8:112. https://doi.org/10.3389/fnbeh.2014.00112
Adhikari A, Topiwala MA, Gordon JA (2010) Synchronized activity between the ventral hippocampus and the medial prefrontal cortex during anxiety. Neuron 65:257–269. https://doi.org/10.1016/j.neuron.2009.12.002
Adhikari A, Topiwala MA, Gordon JA (2011) Single units in the medial prefrontal cortex with anxiety-related firing patterns are preferentially influenced by ventral hippocampal activity. Neuron 71:898–910. https://doi.org/10.1016/j.neuron.2011.07.027
Adhikari A, Lerner TN, Finkelstein J et al (2015) Basomedial amygdala mediates top-down control of anxiety and fear. Nature 527:179–185. https://doi.org/10.1038/nature15698
Akirav I, Richter-Levin G (1999) Biphasic modulation of hippocampal plasticity by behavioral stress and basolateral amygdala stimulation in the rat. J Neurosci 19:10530–10535
Alberini CM (2005) Mechanisms of memory stabilization: are consolidation and reconsolidation similar or distinct processes? Trends Neurosci 28:51–56. https://doi.org/10.1016/j.tins.2004.11.001
Alberini CM, Ledoux JE (2013) Memory reconsolidation. Curr Biol 23:R746–R750. https://doi.org/10.1016/j.cub.2013.06.046
Albrecht A, Bergado-Acosta JR, Pape H-C, Stork O (2010) Role of the neural cell adhesion molecule (NCAM) in amygdalo-hippocampal interactions and salience determination of contextual fear memory. Int J Neuropsychopharmacol 13:661–674. https://doi.org/10.1017/S1461145709991106
Albrecht A, Çalişkan G, Oitzl MS et al (2013) Long-lasting increase of corticosterone after fear memory reactivation: anxiolytic effects and network activity modulation in the ventral hippocampus. Neuropsychopharmacology 38. https://doi.org/10.1038/npp.2012.192
An B, Hong I, Choi S (2012) Long-term neural correlates of reversible fear learning in the lateral amygdala. J Neurosci 32:16845–16856. https://doi.org/10.1523/JNEUROSCI.3017-12.2012
Anderson DJ, Adolphs R (2014) A framework for studying emotions across species. Cell 157:187–200. https://doi.org/10.1016/j.cell.2014.03.003
Baldi E, Bucherelli C (2015) Brain sites involved in fear memory reconsolidation and extinction of rodents. Neurosci Biobehav Rev 53:160–190. https://doi.org/10.1016/j.neubiorev.2015.04.003
Bangasser DA, Eck SR, Ordoñes Sanchez E (2018) Sex differences in stress reactivity in arousal and attention systems. Neuropsychopharmacology. https://doi.org/10.1038/s41386-018-0137-2
Bastos AM, Vezoli J, Fries P (2015) Communication through coherence with inter-areal delays. Curr Opin Neurobiol 31:173–180. https://doi.org/10.1016/j.conb.2014.11.001
Battaglia FP, Sutherland GR, McNaughton BL (2004) Hippocampal sharp wave bursts coincide with neocortical “up-state” transitions. Learn Mem 11:697–704. https://doi.org/10.1101/lm.73504
Bazelot M, Bocchio M, Kasugai Y et al (2015) Hippocampal theta input to the amygdala shapes feedforward inhibition to gate heterosynaptic plasticity. Neuron 87:1290–1303. https://doi.org/10.1016/j.neuron.2015.08.024
Bergado-Acosta JR, Sangha S, Narayanan RT et al (2008) Critical role of the 65-kDa isoform of glutamic acid decarboxylase in consolidation and generalization of Pavlovian fear memory. Learn Mem 15:163–171. https://doi.org/10.1101/lm.705408
Bocchio M, Nabavi S, Capogna M (2017) Synaptic plasticity, engrams, and network oscillations in amygdala circuits for storage and retrieval of emotional memories. Neuron 94:731–743. https://doi.org/10.1016/j.neuron.2017.03.022
Börgers C, Kopell N (2003) Synchronization in networks of excitatory and inhibitory neurons with sparse, random connectivity. Neural Comput 15:509–538. https://doi.org/10.1162/089976603321192059
Börgers C, Walker B (2013) Toggling between gamma-frequency activity and suppression of cell assemblies. Front Comput Neurosci 7:33. https://doi.org/10.3389/fncom.2013.00033
Born J, Wilhelm I (2012) System consolidation of memory during sleep. Psychol Res 76:192–203. https://doi.org/10.1007/s00426-011-0335-6
Bourtchuladze R, Frenguelli B, Blendy J et al (1994) Deficient long-term memory in mice with a targeted mutation of the cAMP-responsive element-binding protein. Cell 79:59–68. https://doi.org/10.1016/0092-8674(94)90400-6
Bouton ME (1988) Context and ambiguity in the extinction of emotional learning: implications for exposure therapy. Behav Res Ther 26:137–149. https://doi.org/10.1016/0005-7967(88)90113-1
Boyce R, Glasgow SD, Williams S, Adamantidis A (2016) Causal evidence for the role of REM sleep theta rhythm in contextual memory consolidation. Science (80- ) 352:812. https://doi.org/10.1126/science.aad5252
Bragin A, Jandó G, Nádasdy Z et al (1995) Gamma (40-100 Hz) oscillation in the hippocampus of the behaving rat. Hippocampus 15:47–60
Brownlow JA, Harb GC, Ross RJ (2015) Treatment of sleep disturbances in post-traumatic stress disorder: a review of the literature. Curr Psychiatry Rep 17. https://doi.org/10.1007/s11920-015-0587-8
Buzsaki G (1989) Two-stage model of memory trace formation: a role for “noisy” brain states. Neuroscience 31:551–570
Buzsáki G (2002) Theta oscillations in the hippocampus. Neuron 33:325–340. https://doi.org/10.1016/S0896-6273(02)00586-X
Buzsáki G (2015) Hippocampal sharp wave-ripple: a cognitive biomarker for episodic memory and planning. Hippocampus 25:1073–1188. https://doi.org/10.1002/hipo.22488
Buzsáki G, Draguhn A (2004) Neuronal oscillations in cortical networks. Science (80- ) 304:1926–1929. https://doi.org/10.1126/science.1099745
Buzsáki G, Moser EI (2013) Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci 16:130–138. https://doi.org/10.1038/nn.3304
Buzsáki, Wang X-J (2012) Mechanisms of gamma oscillations. Annu Rev Neurosci:203–225. https://doi.org/10.1146/annurev-neuro-062111-150444
Buzsáki G, Lai-Wo SL, Vanderwolf CH (1983) Cellular bases of hippocampal EEG in the behaving rat. Brain Res Rev 6:139–171. https://doi.org/10.1016/0165-0173(83)90037-1
Buzsáki G, Horváth Z, Urioste R et al (1992) High-frequency network oscillation in the hippocampus. Science 256:1025–1027. https://doi.org/10.1126/science.1589772
Buzsáki G, Anastassiou CA, Koch C (2012) The origin of extracellular fields and currents--EEG, ECoG, LFP and spikes. Nat Rev Neurosci 13:407–420. https://doi.org/10.1038/nrn3241
Cairney SA, Durrant SJ, Hulleman J, Lewis PA (2014) Targeted memory reactivation during slow wave sleep facilitates emotional memory consolidation. Sleep 37:701–707. https://doi.org/10.5665/sleep.3572
Çalişkan G, Stork O (2018) Hippocampal network oscillations as mediators of behavioural metaplasticity: insights from emotional learning. Neurobiol Learn Mem
Çalışkan G, Schulz SB, Gruber D et al (2015) Corticosterone and corticotropin-releasing factor acutely facilitate gamma oscillations in the hippocampus in vitro. Eur J Neurosci 41:31–44. https://doi.org/10.1111/ejn.12750
Çalişkan G, Müller I, Semtner M et al (2016) Identification of Parvalbumin interneurons as cellular substrate of fear memory persistence. Cereb Cortex 26:2325–2340. https://doi.org/10.1093/cercor/bhw001
Canolty R, Edwards E, Dalal S et al (2006) High gamma power is phase-locked to theta oscillations in human neocortex. Science (80- ) 313:1626–1628. https://doi.org/10.1126/science.1128115
Cardin JA, Carlén M, Meletis K et al (2009) Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459:663–667. https://doi.org/10.1038/nature08002
Cellini N, Capuozzo A (2018) Shaping memory consolidation via targeted memory reactivation during sleep. Ann N Y Acad Sci:1–20. https://doi.org/10.1111/nyas.13855
Chen Z-Y, Jing D, Bath KG et al (2006) Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science (80- ) 314:140–143. https://doi.org/10.1126/science.1129663
Ciocchi S, Passecker J, Malagon-Vina H et al (2015) Selective information routing by ventral hippocampal CA1 projection neurons. Science (80- ) 348:560–563. https://doi.org/10.1126/science.aaa3245
Colgin LL, Denninger T, Fyhn M et al (2009) Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 462:353–357. https://doi.org/10.1038/nature08573
Contreras D, Steriade M (1995) Cellular basis of EEG slow rhythms: a study of dynamic corticothalamic relationships. J Neurosci 15:604–622
Courtin J, Karalis N, Gonzalez-Campo C et al (2014) Persistence of amygdala gamma oscillations during extinction learning predicts spontaneous fear recovery. Neurobiol Learn Mem 113:82–89. https://doi.org/10.1016/j.nlm.2013.09.015
Craske MG, Kircanski K, Zelikowsky M et al (2008) Optimizing inhibitory learning during exposure therapy. Behav Res Ther 46:5–27. https://doi.org/10.1016/j.brat.2007.10.003
Crawley JN (1985) Exploratory behavior models of anxiety in mice. Neurosci Biobehav Rev 9:37–44. https://doi.org/10.1016/0149-7634(85)90030-2
Csicsvari J, Hirase H, Mamiya a BG (2000) Ensemble patterns of hippocampal CA3-CA1 neurons during sharp wave-associated population events. Neuron 28:585–594. https://doi.org/10.1016/S0896-6273(00)00135-5
Csicsvari J, Jamieson B, Wise KD, Buzsáki G (2003) Mechanisms of gamma oscillations in the hippocampus of the behaving rat. Neuron 37:311–322. https://doi.org/10.1016/S0896-6273(02)01169-8
Dannenberg H, Pabst M, Braganza O et al (2015) Synergy of direct and indirect cholinergic septo-hippocampal pathways coordinates firing in hippocampal networks. J Neurosci 35:8394–8410. https://doi.org/10.1523/JNEUROSCI.4460-14.2015
Davis M, Whalen PJ (2001) The amygdala: vigilance and emotion. Mol Psychiatry 6:13–34. https://doi.org/10.1038/sj.mp.4000812
Davis M, Walker DL, Miles L, Grillon C (2010) Phasic vs sustained fear in rats and humans: role of the extended amygdala in fear vs anxiety. Neuropsychopharmacology 35:105–135. https://doi.org/10.1038/npp.2009.109
Davis P, Zaki Y, Maguire J, Reijmers LG (2017) Cellular and oscillatory substrates of fear extinction learning. Nat Neurosci 20:1624–1633. https://doi.org/10.1038/nn.4651
de Almeida L, Idiart M, Lisman J (2009) A second function of gamma-frequency oscillations: a E%-max winner-take-all mechanism selects which cells fire. J Neurosci 29:211–220. https://doi.org/10.1007/s11103-011-9767-z.Plastid
De Boer M, Hofman M (2017) The spectral fingerprint of sleep problems in post-traumatic stress disorder. bioRxiv Prepr 1–22. https://doi.org/10.1101/209452
Dejean C, Courtin J, Rozeske RR et al (2015) Neuronal circuits for fear expression and recovery: recent advances and potential therapeutic strategies. Biol Psychiatry 78:298–306. https://doi.org/10.1016/j.biopsych.2015.03.017
Dejean C, Courtin J, Karalis N et al (2016) Prefrontal neuronal assemblies temporally control fear behaviour. Nature 535:420–424. https://doi.org/10.1038/nature18630
Desmedt A, Marighetto A, Piazza P-V (2015) Abnormal fear memory as a model for posttraumatic stress disorder. Biol Psychiatry 78:290–297. https://doi.org/10.1016/j.biopsych.2015.06.017
Diekelmann S, Born J (2007) One memory, two ways to consolidate? Nat Neurosci 10:1085–1086. https://doi.org/10.1038/nn0907-1085
Diekelmann S, Born J (2010) The memory function of sleep. Nat Rev Neurosci 11:114–126. https://doi.org/10.1038/nrn2762
Dilgen JE, Tompa T, Saggu S et al (2013) Optogenetically evoked gamma oscillations are disturbed by cocaine administration. Front Cell Neurosci 7:213. https://doi.org/10.3389/fncel.2013.00213
Dudai Y (2002) Molecular bases of long-term memories: a question of persistence. Curr Opin Neurobiol 12:211–216. https://doi.org/10.1016/S0959-4388(02)00305-7
Dunkley BT, Doesburg SM, Jetly R et al (2015) Characterising intra- and inter-intrinsic network synchrony in combat-related post-traumatic stress disorder. Psychiatry Res Neuroimaging 234:172–181. https://doi.org/10.1016/j.pscychresns.2015.09.002
Einevoll GT, Kayser C, Logothetis NK, Panzeri S (2013) Modelling and analysis of local field potentials for studying the function of cortical circuits. Nat Rev Neurosci 14:770–785. https://doi.org/10.1038/nrn3599
Etkin A, Egner T, Kalisch R (2011) Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci 15:85–93. https://doi.org/10.1016/j.tics.2010.11.004
Euston DR, Tatsuno M, Mcnaughton BL (2007) Fast-forward playback of recent in prefrontal memory sequences cortex during sleep. Science (80- ) 318:1147–1150. https://doi.org/10.1126/science.1148979
Fanselow MS, Dong HW (2010) Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65:7–19. https://doi.org/10.1016/j.neuron.2009.11.031
Felix-Ortiz AC, Beyeler A, Seo C et al (2013) BLA to vHPC inputs modulate anxiety-related behaviors. Neuron 79:658–664. https://doi.org/10.1016/j.neuron.2013.06.016
Fell J, Axmacher N (2011) The role of phase synchronization in memory processes. Nat Rev Neurosci 12:105–118. https://doi.org/10.1038/nrn2979
Fell J, Klaver P, Lehnertz K et al (2001) Human memory formation is accompanied by rhinal-hippocampal coupling and decoupling. Nat Neurosci 4:1259–1264. https://doi.org/10.1038/nn759
Fenton GE, Pollard AK, Halliday DM et al (2014) Persistent prelimbic cortex activity contributes to enhanced learned fear expression in females. Learn Mem 21:55–60. https://doi.org/10.1101/lm.033514.113
Fenton GE, Halliday DM, Mason R et al (2016) Sex differences in learned fear expression and extinction involve altered gamma oscillations in medial prefrontal cortex. Neurobiol Learn Mem 135:66–72. https://doi.org/10.1016/j.nlm.2016.06.019
Fisahn A, Pike FG, Buhl EH, Paulsen O (1998) Cholinergic induction of network oscillations at 40|[thinsp]|Hz in the hippocampus in vitro. Nature 394:186–189. https://doi.org/10.1038/28179
Frankland PW, Bontempi B (2005) The organization of recent and remote memories. Nature 6:119–130. https://doi.org/10.1101/lm.97905
Fries P (2005) A mechanism for cognitive dynamics: neuronal communication through neuronal coherence. Trends Cogn Sci 9:474–480. https://doi.org/10.1016/j.tics.2005.08.011
Fries P, Reynolds JH, Rorie a E, Desimone R (2001) Modulation of oscillatory neuronal synchronization by selective visual attention. Science (80- ) 291:1560–1563. https://doi.org/10.1126/science.291.5508.1560
Fries P, Nikolić D, Singer W (2007) The gamma cycle. Trends Neurosci 30:309–316. https://doi.org/10.1016/j.tins.2007.05.005
Gangadharan G, Shin J, Kim S-W et al (2016) Medial septal GABAergic projection neurons promote object exploration behavior and type 2 theta rhythm. Proc Natl Acad Sci U S A 113:6550–6555. https://doi.org/10.1073/pnas.1605019113
Garner AR, Rowland DC, Hwang SY et al (2012) Generation of a synthetic memory trace. Science (80- ) 335:1513–1516. https://doi.org/10.1126/science.1214985
Girardeau G, Benchenane K, Wiener SI et al (2009) Selective suppression of hippocampal ripples impairs spatial memory. Nat Neurosci 12:1222–1223. https://doi.org/10.1038/nn.2384
Girardeau G, Inema I, Buzsáki G (2017) Reactivations of emotional memory in the hippocampus–amygdala system during sleep. Nat Neurosci 20:1634–1642. https://doi.org/10.1038/nn.4637
Goldstein AN, Walker MP (2014) The role of sleep in emotional brain function. Annu Rev Clin Psychol 10:679–708. https://doi.org/10.1146/annurev-clinpsy-032813-153716
Gordon JA, Lacefield CO, Kentros CG, Hen R (2005) State-dependent alterations in hippocampal oscillations in serotonin 1A receptor-deficient mice. J Neurosci 25:6509–6519. https://doi.org/10.1523/JNEUROSCI.1211-05.2005
Gottesmann C (2011) The involvement of noradrenaline in rapid eye movement sleep mentation. Front Neurol DEC:1–10. https://doi.org/10.3389/fneur.2011.00081
Gray CM, König P, Engel AK, Singer W (1989) Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature 338:334–337
Grupe DW, Nitschke JB (2013) Uncertainty and anticipation in anxiety: an integrated neurobiological and psychological perspective. Nat Rev Neurosci 14:488–501. https://doi.org/10.1038/nrn3524
Hájos N, Paulsen O (2009) Network mechanisms of gamma oscillations in the CA3 region of the hippocampus. Neural Netw 22:1113–1119. https://doi.org/10.1016/j.neunet.2009.07.024
Hajszan T, Alreja M, Leranth C (2004) Intrinsic vesicular glutamate transporter 2-immunoreactive input to septohippocampal parvalbumin-containing neurons: novel glutamatergic local circuit cells. Hippocampus 14:499–509. https://doi.org/10.1002/hipo.10195
Han JH, Kushner SA, Yiu AP et al (2009) Selective erasure of a fear memory. Science (80- ) 323:1492–1496. https://doi.org/10.1126/science.1164139
Hasenstaub A, Shu Y, Haider B et al (2005) Inhibitory postsynaptic potentials carry synchronized frequency information in active cortical networks. Neuron 47:423–435. https://doi.org/10.1016/j.neuron.2005.06.016
Hauner KK, Howard JD, Zelano C, Gottfried JA (2013) Stimulus-specific enhancement of fear extinction during slow-wave sleep. Nat Neurosci 16:1553–1555. https://doi.org/10.1038/nn.3527
He J, Sun H-Q, Li S-X et al (2015) Effect of conditioned stimulus exposure during slow wave sleep on fear memory extinction in humans. Sleep 38:423–431. https://doi.org/10.5665/sleep.4502
Headley DB, Paré D (2013) In sync: gamma oscillations and emotional memory. Front Behav Neurosci 7:170. https://doi.org/10.3389/fnbeh.2013.00170
Headley DB, Paré D (2017) Common oscillatory mechanisms across multiple memory systems. npj Sci Learn 2(1). https://doi.org/10.1038/s41539-016-0001-2
Hermans D, Craske MG, Mineka S, Lovibond PF (2006) Extinction in human fear conditioning. Biol Psychiatry 60:361–368. https://doi.org/10.1016/j.biopsych.2005.10.006
Herry C, Ciocchi S, Senn V et al (2008) Switching on and off fear by distinct neuronal circuits. Nature 454:600–606. https://doi.org/10.1038/nature07166
Hölscher C, Anwyl R, Rowan MJ (1997) Stimulation on the positive phase of hippocampal theta rhythm induces long-term potentiation that can be depotentiated by stimulation on the negative phase in area CA1 in vivo. J Neurosci 17:6470–6477
Hübner C, Bosch D, Gall A et al (2014) Ex vivo dissection of optogenetically activated mPFC and hippocampal inputs to neurons in the basolateral amygdala: implications for fear and emotional memory. Front Behav Neurosci 8. https://doi.org/10.3389/fnbeh.2014.00064
Huerta PT, Lisman JE (1995) Bidirectional synaptic plasticity induced by a single burst during cholinergic theta oscillation in CA1 in vitro. Neuron 15:1053–1063. https://doi.org/10.1016/0896-6273(95)90094-2
Huh CYL, Goutagny R, Williams S (2010) Glutamatergic neurons of the mouse medial septum and diagonal band of Broca synaptically drive hippocampal pyramidal cells: relevance for hippocampal theta rhythm. J Neurosci 30:15951–15961. https://doi.org/10.1523/JNEUROSCI.3663-10.2010
Hyman JM, Wyble BP, Goyal V et al (2003) Stimulation in hippocampal region CA1 in behaving rats yields long-term potentiation when delivered to the peak of theta and long-term depression when delivered to the trough. J Neurosci 23:11725–11731
Iaccarino HF, Singer AC, Martorell AJ et al (2016) Gamma frequency entrainment attenuates amyloid load and modifies microglia. Nature 540:230–235. https://doi.org/10.1038/nature20587
Isomura Y, Sirota A, Özen S et al (2006) Integration and segregation of activity in entorhinal-hippocampal subregions by neocortical slow oscillations. Neuron 52:871–882. https://doi.org/10.1016/j.neuron.2006.10.023
Izquierdo A (2006) Brief uncontrollable stress causes dendritic retraction in infralimbic cortex and resistance to fear extinction in mice. J Neurosci 26:5733–5738. https://doi.org/10.1523/JNEUROSCI.0474-06.2006
Jacinto LR, Reis JS, Dias NS et al (2013) Stress affects theta activity in limbic networks and impairs novelty-induced exploration and familiarization. Front Behav Neurosci 7:1–11. https://doi.org/10.3389/fnbeh.2013.00127
Jacinto LR, Cerqueira JJ, Sousa N (2016) Patterns of theta activity in limbic anxiety circuit preceding exploratory behavior in approach-avoidance conflict. Front Behav Neurosci 10. https://doi.org/10.3389/fnbeh.2016.00171
Jarosiewicz B, McNaughton BL, Skaggs WE (2002) Hippocampal population activity during the small-amplitude irregular activity state in the rat. J Neurosci 22:1373–1384
Jin J, Maren S (2015) Prefrontal-hippocampal interactions in memory and emotion. Front Syst Neurosci 9:1–8. https://doi.org/10.3389/fnsys.2015.00170
Joëls M, Baram TZ (2009) The neuro-symphony of stress. Nat Rev Neurosci 10:459–466. https://doi.org/10.1038/nrn2632
Jouvet M (1969) Biogenic amines and the states of sleep. Science 163:32–41
Jovanovic T, Norrholm SD, Blanding NQ et al (2010) Impaired fear inhibition is a biomarker of PTSD but not depression. Depress Anxiety 27:244–251. https://doi.org/10.1002/da.20663
Kametani H, Kawamura H (1990) Alterations in acetylcholine release in the rat hippocampus during sleep-wakefulness detected by intracerebral dialysis. 47:421–426
Karalis N, Dejean C, Chaudun F et al (2016) 4-Hz oscillations synchronize prefrontal–amygdala circuits during fear behavior. Nat Neurosci 19:605–612. https://doi.org/10.1038/nn.4251
Karunakaran S, Chowdhury A, Donato F et al (2016) PV plasticity sustained through D1/5 dopamine signaling required for long-term memory consolidation. Nat Neurosci 19:454–464. https://doi.org/10.1038/nn.4231
Khemka S, Barnes G, Dolan RJ, Bach DR (2017) Dissecting the function of hippocampal oscillations in a human anxiety model. J Neurosci 37:6869–6876. https://doi.org/10.1523/JNEUROSCI.1834-16.2017
Kim J, Kwon J-T, Kim H-S et al (2013) Memory recall and modifications by activating neurons with elevated CREB. Nat Neurosci 17:65–72. https://doi.org/10.1038/nn.3592
King C, Henze D, Leinekugel X, Buzsáki G (1999) Hebbian modification of a hippocampal population pattern in the rat. J Physiol 521:159–167. https://doi.org/10.1111/j.1469-7793.1999.00159.x
Kitamura T, Ogawa SK, Roy DS et al (2017) Engrams and circuits crucial for systems consolidation of a memory. Science (80- ) 356:73–78. https://doi.org/10.1126/science.aam6808
Kjelstrup KG, Tuvnes FA, Steffenach H-A et al (2002) Reduced fear expression after lesions of the ventral hippocampus. Proc Natl Acad Sci 99:10825–10830. https://doi.org/10.1073/pnas.152112399
Knapska E, Macias M, Mikosz M et al (2012) Functional anatomy of neural circuits regulating fear and extinction. Proc Natl Acad Sci 109:17093–17098. https://doi.org/10.1073/pnas.1202087109
Kobayashi I, Boarts JM, Delahanty DL (2007) Polysomnographically measured sleep abnormalities in PTSD: a meta-analytic review. Psychophysiology 44:660–669. https://doi.org/10.1111/j.1469-8986.2007.537.x
Korotkova T, Ponomarenko A, Monaghan CK et al (2018) Reconciling the different faces of hippocampal theta: the role of theta oscillations in cognitive, emotional and innate behaviors. Neurosci Biobehav Rev 85:65–80. https://doi.org/10.1016/j.neubiorev.2017.09.004
Kramis R, Vanderwolf CH, Bland BH (1975) Two types of hippocampal rhythmical slow activity in both the rabbit and the rat : relations to behavior and effects of atropine, diethyl ether, urethane, and pentobarbital. 49:58–85
Kudrimoti HS, Barnes CA, McNaughton BL (1999) Reactivation of hippocampal cell assemblies: effects of behavioral state, experience, and EEG dynamics. J Neurosci 19:4090–4101
Ledoux JE (2000) Emotion circuits in the brain. Annu Rev Neurosci 23:155–184
Lee MG, Chrobak JJ, Sik A et al (1994) Hippocampal theta activity following selective lesion of the septal cholinergic system. Neurosci 62:1033–1047
Lesting J, Geiger M, Narayanan RT, et al (2011a) Impaired extinction of fear and maintained amygdala-hippocampal theta synchrony in a mouse model of temporal lobe epilepsy. Epilepsia 52:337–346. https://doi.org/10.1111/j.1528-1167.2010.02758.x
Lesting J, Narayanan RT, Kluge C et al (2011b) Patterns of coupled theta activity in amygdala-hippocampal-prefrontal cortical circuits during fear extinction. PLoS One 6. https://doi.org/10.1371/journal.pone.0021714
Lesting J, Daldrup T, Narayanan V et al (2013) Directional theta coherence in prefrontal cortical to amygdalo-hippocampal pathways signals fear extinction. PLoS One 8:17–19. https://doi.org/10.1371/journal.pone.0077707
Li WJ, Yu H, Yang JM et al (2010) Anxiolytic effect of music exposure on BDNFMet/Mettransgenic mice. Brain Res 1347:71–79. https://doi.org/10.1016/j.brainres.2010.05.080
Likhtik E, Stujenske JM, A Topiwala M et al (2014) Prefrontal entrainment of amygdala activity signals safety in learned fear and innate anxiety. Nat Neurosci 17:106–113. https://doi.org/10.1038/nn.3582
Liu X, Ramirez S, Pang PT et al (2012) Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature 484:381–385. https://doi.org/10.1038/nature11028
Maggio N, Segal M (2011) Persistent changes in ability to express long-term potentiation/depression in the rat hippocampus after juvenile/adult stress. Biol Psychiatry 69:748–753. https://doi.org/10.1016/j.biopsych.2010.11.026
Maingret N, Girardeau G, Todorova R et al (2016) Hippocampo-cortical coupling mediates memory consolidation during sleep. Nat Neurosci 19:959–964. https://doi.org/10.1038/nn.4304 http://www.nature.com/neuro/journal/v19/n7/abs/nn.4304.html#supplementary-information
Mann EO, Suckling JM, Hajos N et al (2005) Perisomatic feedback inhibition underlies cholinergically induced fast network oscillations in the rat hippocampus in vitro. Neuron 45:105–117. https://doi.org/10.1016/j.neuron.2004.12.016
Marek R, Jin J, Goode TD et al (2018) Hippocampus-driven feed-forward inhibition of the prefrontal cortex mediates relapse of extinguished fear. Nat Neurosci 21:1–9. https://doi.org/10.1038/s41593-018-0073-9
Maren S, Holmes A (2015) Stress and fear extinction. Neuropsychopharmacology 41:1–22. https://doi.org/10.1038/npp.2015.180
Maren S, Holt W (2000) The hippocampus and contextual memory retrieval in Pavlovian conditioning. Behav Brain Res 110:97–108. https://doi.org/10.1016/S0166-4328(99)00188-6
Maren S, Quirk GJ (2004) Neuronal signalling of fear memory. Nat Rev Neurosci 5:844–852. https://doi.org/10.1038/nrn1535
Maren S, Phan KL, Liberzon I (2013) The contextual brain: implications for fear conditioning, extinction and psychopathology. Nat Rev Neurosci 14:417–428. https://doi.org/10.1038/nrn3492
Maroun M, Richter-Levin G (2003) Exposure to acute stress blocks the induction of long-term potentiation of the amygdala-prefrontal cortex pathway in vivo. J Neurosci 23:4406–4409
Marrosu F, Portas C, Mascia MS et al (1995) Microdialysis measurement of cortical and hippocampal acetylcholine-release during sleep-wake cycle in freely moving cats. Brain Res 671:329–332. https://doi.org/10.1016/0006-8993(94)01399-3
McGaugh JL (2000) Neuroscience - memory - a century of consolidation. Science 287:248–251. https://doi.org/10.1126/science.287.5451.248
Mizunuma M, Norimoto H, Tao K et al (2014) Unbalanced excitability underlies offline reactivation of behaviorally activated neurons. Nat Neurosci 17:503–505. https://doi.org/10.1038/nn.3674
Montgomery SM, Buzsáki G (2007) Gamma oscillations dynamically couple hippocampal CA3 and CA1 regions during memory task performance. Proc Natl Acad Sci U S A 104:14495–14500. https://doi.org/10.1073/pnas.0701826104
Myers KM, Davis M (2007) Mechanisms of fear extinction. Mol Psychiatry 12:120–150. https://doi.org/10.1038/sj.mp.4001939
Naber PA, Witter MP (1998) Subicular efferents are organized mostly as parallel projections: a double-labeling, retrograde-tracing study in the rat. J Comp Neurol 393:284–297. https://doi.org/10.1002/(SICI)1096-9861(19980413)393:3<284::AID-CNE2>3.0.CO;2-Y
Nader K, Schafe GE, LeDoux JE (2000) The labile nature of consolidation theory. Nat Rev Neurosci 1:216–219. https://doi.org/10.1038/35044580
Narayanan RT, Seidenbecher T, Kluge C et al (2007) Dissociated theta phase synchronization in amygdalo-hippocampal circuits during various stages of fear memory. Eur J Neurosci 25:1823–1831. https://doi.org/10.1111/j.1460-9568.2007.05437.x
Neske GT (2015) The slow oscillation in cortical and thalamic networks: mechanisms and functions. Front Neural Circuits 9(88). https://doi.org/10.3389/fncir.2015.00088
Neylan TC, Lenoci M, Maglione ML et al (2003) Delta sleep response to metyrapone in post-traumatic stress disorder. Neuropsychopharmacology 28:1666–1676. https://doi.org/10.1038/sj.npp.1300215
Nikolić D, Fries P, Singer W (2013) Gamma oscillations: precise temporal coordination without a metronome. Trends Cogn Sci 17:54–55. https://doi.org/10.1016/j.tics.2012.12.003
Ognjanovski N, Maruyama D, Lashner N et al (2014) CA1 hippocampal network activity changes during sleep-dependent memory consolidation. Front Syst Neurosci 8:1–11. https://doi.org/10.3389/fnsys.2014.00061
Ognjanovski N, Schaeffer S, Wu J et al (2017) Parvalbumin-expressing interneurons coordinate hippocampal network dynamics required for memory consolidation. Nat Commun 8:15039. https://doi.org/10.1038/ncomms15039
Ognjanovski N, Broussard C, Zochowski M, Aton SJ (2018) Hippocampal network oscillations rescue memory consolidation deficits caused by sleep loss. Cereb Cortex:1–13. https://doi.org/10.1093/cercor/bhy174
Olsen RHJ, Marzulla T, Raber J (2014) Impairment in extinction of contextual and cued fear following post-training whole-body irradiation. Front Behav Neurosci 8:1–13. https://doi.org/10.3389/fnbeh.2014.00231
Padilla-Coreano N, Bolkan SS, Pierce GM et al (2016) Direct ventral hippocampal-prefrontal input is required for anxiety-related neural activity and behavior. Neuron 89:857–866. https://doi.org/10.1016/j.neuron.2016.01.011
Papadimitriou GN, Linkowski P (2005) Sleep disturbance in anxiety disorders. Int Rev Psychiatry 17:229–236. https://doi.org/10.1080/09540260500104524
Pape HC, Narayanan RT, Smid J et al (2005) Theta activity in neurons and networks of the amygdala related to long-term fear memory. Hippocampus 15:874–880. https://doi.org/10.1002/hipo.20120
Parikh V, Kozak R, Martinez V, Sarter M (2007) Prefrontal acetylcholine release controls cue detection on multiple timescales. Neuron 56:141–154. https://doi.org/10.1016/j.neuron.2007.08.025
Patel J, Fujisawa S, Berényi A et al (2012) Traveling theta waves along the entire septotemporal axis of the hippocampus. Neuron 75:410–417. https://doi.org/10.1016/j.neuron.2012.07.015
Patel D, Anilkumar S, Chattarji S, Buwalda B (2018) Repeated social stress leads to contrasting patterns of structural plasticity in the amygdala and hippocampus. Behav Brain Res 347:314–324. https://doi.org/10.1016/j.bbr.2018.03.034
Pellow S, Chopin P, File SE, Briley M (1985) Validation of open : closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Methods 14:149–167. https://doi.org/10.1016/0165-0270(85)90031-7
Petsche H, Stumpf C, Gogolak G (1962) The significance of the rabbit’s septum as a relay station between the midbrain and the hippocampus. I. The control of hippocampus arousal activity by the septum cells. Electroencephalogr Clin Neurophysiol 14:202–211
Phelps EA (2004) Human emotion and memory: interactions of the amygdala and hippocampal complex. Curr Opin Neurobiol 14:198–202. https://doi.org/10.1016/j.conb.2004.03.015
Pikkarainen M, Rönkkö S, Savander V et al (1999) Projections from the lateral, basal, and accessory basal nuclei of the amygdala to the entorhinal cortex in the macaque monkey. J Comp Neurol 403:229–260. https://doi.org/10.1002/hipo.1099
Popa D, Duvarci S, Popescu AT et al (2010) Coherent amygdalocortical theta promotes fear memory consolidation during paradoxical sleep. Proc Natl Acad Sci 107:6516–6519. https://doi.org/10.1073/pnas.0913016107
Prasad JA, Chudasama Y (2013) Viral tracing identifies parallel disynaptic pathways to the hippocampus. J Neurosci 33:8494–8503. https://doi.org/10.1523/JNEUROSCI.5072-12.2013
Quirk GJ, Mueller D (2008) Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 33:56–72. https://doi.org/10.1038/sj.npp.1301555
Ramirez S, Liu X, Lin P-A et al (2013) Creating a false memory in the hippocampus. Science (80- ) 341:387–391. https://doi.org/10.1126/science.1239073
Raza SA, Albrecht A, Çalışkan G et al (2017) HIPP neurons in the dentate gyrus mediate the cholinergic modulation of background context memory salience. Nat Commun 8:189. https://doi.org/10.1038/s41467-017-00205-3
Reijmers LG, Perkins BL, Matsuo N, Mayford M (2007) Localization of a stable neural correlate of associative memory. Science (80- ) 317:1230–1233. https://doi.org/10.1126/science.1143839
Royer S, Sirota A, Patel J, Buzsáki G (2012) Distinct representations and theta dynamics in dorsal and ventral hippocampus. 76:211–220. https://doi.org/10.1523/JNEUROSCI.4681-09.2010
Sadowski JHLP, Jones MW, Mellor JR et al (2016) Sharp-wave ripples orchestrate the induction of synaptic plasticity during reactivation of place cell firing patterns in the hippocampus. Cell Rep 14:1916–1929. https://doi.org/10.1016/j.celrep.2016.01.061
Sampath D, Sabitha KR, Hegde P et al (2014) A study on fear memory retrieval and REM sleep in maternal separation and isolation stressed rats. Behav Brain Res 273:144–154. https://doi.org/10.1016/j.bbr.2014.07.034
Sangha S, Narayanan RT, Bergado-Acosta JR et al (2009) Deficiency of the 65 kDa isoform of glutamic acid decarboxylase impairs extinction of cued but not contextual fear memory. J Neurosci 29:15713–15720. https://doi.org/10.1523/JNEUROSCI.2620-09.2009
Satpute AB, Mumford JA, Naliboff BD, Poldrack RA (2012) Human anterior and posterior hippocampus respond distinctly to state and trait anxiety. Emotion 12:58–68. https://doi.org/10.1037/a0026517
Schlingloff D, Kali S, Freund TF et al (2014) Mechanisms of sharp wave initiation and ripple generation. J Neurosci 34:11385–11398. https://doi.org/10.1523/JNEUROSCI.0867-14.2014
Segal M, Richter-Levin G, Maggio N (2010) Stress-induced dynamic routing of hippocampal connectivity: a hypothesis. Hippocampus 20:1332–1338. https://doi.org/10.1002/hipo.20751
Seidenbecher T, Laxmi TR, Stork O, Pape HC (2003) Amygdalar and hippocampal theta rhythm synchronization during fear memory retrieval. Science (80- ) 301:846–850. https://doi.org/10.1126/science.1085818
Senn V, Wolff SBE, Herry C et al (2014) Long-range connectivity defines behavioral specificity of amygdala neurons. Neuron 81:428–437. https://doi.org/10.1016/j.neuron.2013.11.006
Shors TJ, Seib TB, Levine S, Thompson RF (1989) Inescapable versus escapable shock modulates long-term potentiation in the rat hippocampus. Science 244:224–226. https://doi.org/10.1126/science.2704997
Siapas AG, Lubenov EV, Wilson MA (2005) Prefrontal phase locking to hippocampal theta oscillations. Neuron 46:141–151. https://doi.org/10.1016/j.neuron.2005.02.028
Siegle JS, Pritschett DL, Moore CI (2014) Gamma-range synchronization of fast-spiking interneurons can enhance detection of tactile stimuli. Nat Neurosci 17:1371–1379. https://doi.org/10.1038/nn.3797
Sirota A, Csicsvari J, Buhl D, Buzsáki G (2003) Communication between neocortex and hippocampus during sleep in rodents. Proc Natl Acad Sci U S A 100:2065–2069. https://doi.org/10.1073/pnas.0437938100
Sohal VS, Zhang F, Yizhar O, Deisseroth K (2009) Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459:1–5. https://doi.org/10.1038/nature07991
Soliman F, Glatt CE, Bath KG et al (2010) A genetic variant BDNF polymorphism alters extinction learning in both mouse and human. Science 327:863–866
Sotres-Bayon F, Sierra-Mercado D, Pardilla-Delgado E, Quirk GJ (2012) Gating of fear in prelimbic cortex by hippocampal and amygdala inputs. Neuron 76:804–812. https://doi.org/10.1016/j.neuron.2012.09.028
Stark E, Roux L, Eichler R et al (2014) Pyramidal cell-interneuron interactions underlie hippocampal ripple oscillations. Neuron 83:467–480. https://doi.org/10.1016/j.neuron.2014.06.023
Strange BA, Witter MP, Lein ES, Moser EI (2014) Functional organization of the hippocampal longitudinal axis. Nat Rev Neurosci 15:655–669. https://doi.org/10.1038/nrn3785
Stujenske JM, Likhtik E, Topiwala MA, Gordon JA (2014) Fear and safety engage competing patterns of theta-gamma coupling in the basolateral amygdala. Neuron 83:919–933. https://doi.org/10.1016/j.neuron.2014.07.026
Sylvers P, Lilienfeld SO, LaPrairie JL (2011) Differences between trait fear and trait anxiety: implications for psychopathology. Clin Psychol Rev 31:122–137. https://doi.org/10.1016/j.cpr.2010.08.004
Takahashi K, Kayama Y, Lin JS, Sakai K (2010) Locus coeruleus neuronal activity during the sleep-waking cycle in mice. Neuroscience 169:1115–1126. https://doi.org/10.1016/j.neuroscience.2010.06.009
Totty MS, Chesney LA, Geist PA, Datta S (2017) Sleep-dependent oscillatory synchronization: a role in fear memory consolidation. Front Neural Circuits 11:1–13. https://doi.org/10.3389/fncir.2017.00049
Tovote P, Fadok JP, Lüthi A (2015) Neuronal circuits for fear and anxiety. Nat Rev Neurosci 16:317–331. https://doi.org/10.1038/nrn3945
Tronson NC, Taylor JR (2007) Molecular mechanisms of memory reconsolidation. Nat Rev Neurosci 8:262–275. https://doi.org/10.1038/nrn2090
Tronson NC, Schrick C, Guzman YF et al (2009) Segregated populations of hippocampal principal CA1 neurons mediating conditioning and extinction of contextual fear. J Neurosci 29:3387–3394. https://doi.org/10.1523/JNEUROSCI.5619-08.2009
Unal G, Joshi A, Viney TJ et al (2015) Synaptic targets of medial septal projections in the hippocampus and extrahippocampal cortices of the mouse. J Neurosci 35:15812–15826. https://doi.org/10.1523/JNEUROSCI.2639-15.2015
Van Groen T, Wyss JM (1990) Extrinsic projections from area CA1 of the rat hippocampus: olfactory, cortical, subcortical, and bilateral hippocampal formation projections. J Comp Neurol 302:515–528. https://doi.org/10.1002/cne.903020308
Vandecasteele M, Varga V, Berényi A et al (2014) Optogenetic activation of septal cholinergic neurons suppresses sharp wave ripples and enhances theta oscillations in the hippocampus. Proc Natl Acad Sci U S A 111:13535–13540. https://doi.org/10.1073/pnas.1411233111
Vanderwolf C (1969) Hippocampal electrical activity and voluntary movement in the rat. Electroencephalogr Clin Neurophysiol 26:407–418
VanElzakker MB, Kathryn Dahlgren M, Caroline Davis F et al (2014) From Pavlov to PTSD: the extinction of conditioned fear in rodents, humans, and anxiety disorders. Neurobiol Learn Mem 113:3–18. https://doi.org/10.1016/j.nlm.2013.11.014
Varela C, Kumar S, Yang JY, Wilson MA (2014) Anatomical substrates for direct interactions between hippocampus, medial prefrontal cortex, and the thalamic nucleus reuniens. Brain Struct Funct 219:911–929. https://doi.org/10.1007/s00429-013-0543-5
Viriyopase A, Memmesheimer R-M, Gielen S (2016) Cooperation and competition of gamma oscillation mechanisms. J Neurophysiol. https://doi.org/10.1152/jn.00493.2015
Wang X, Buzsáki G (1996) Gamma oscillation by synaptic inhibition in a hippocampal interneuronal network model. J Neurosci 16:6402–6413
Watanabe Y, Gould E, McEwen BS (1992) Stress induces atrophy of apical dendrites of hippocampus CA3 pyramidal neurons. Brain Res 588:341–344
Whittington MA, Traub RD, Kopell N et al (2000) Inhibition-based rhythms: experimental and mathematical observations on network dynamics. Int J Psychophysiol 38:315–336. https://doi.org/10.1016/S0167-8760(00)00173-2
Wilson MA, McNaughton BL (1994) Reactivation of hippocampal ensemble memories during sleep. Science (80- ) 265:676–679. https://doi.org/10.1126/science.8036517
Winkelmann A, Maggio N, Eller J et al (2014) Changes in neural network homeostasis trigger neuropsychiatric symptoms. J Clin Invest 124. https://doi.org/10.1172/JCI71472
Wójtowicz AM, Van Den Boom L, Chakrabarty A et al (2009) Monoamines block kainate- and carbachol-induced. γ-oscillations but augment stimulus-induced γ-oscillations in rat hippocampus in vitro. Hippocampus 19:273–288. https://doi.org/10.1002/hipo.20508
Wolansky T, Clement EA, Peters SR et al (2006) Hippocampal slow oscillation: a novel EEG state and its coordination with ongoing neocortical activity. J Neurosci 26:6213–6229. https://doi.org/10.1523/JNEUROSCI.5594-05.2006
Woodward SH, Murburg MM, Bliwise DL (2000) PTSD-related hyperarousal assessed during sleep. Physiol Behav 70:197–203. https://doi.org/10.1016/S0031-9384(00)00271-7
Xia F, Richards BA, Tran MM et al (2017) Parvalbumin-positive interneurons mediate neocortical-hippocampal interactions that are necessary for memory consolidation. Elife 6:1–25. https://doi.org/10.7554/eLife.27868
Xu C, Krabbe S, Gründemann J et al (2016) Distinct hippocampal pathways mediate dissociable roles of context in memory retrieval. Cell 167:961–972.e16. https://doi.org/10.1016/j.cell.2016.09.051
Ylinen A, Bragin A, Nádasdy Z et al (1995) Sharp wave-associated high-frequency oscillation (200 Hz) in the intact hippocampus: network and intracellular mechanisms. J Neurosci 15:30–46. https://doi.org/10.1038/17605
Yu H, Wang Y, Pattwell S et al (2009) Variant BDNF Val66Met polymorphism affects extinction of conditioned aversive memory. J Neurosci 29:4056–4064. https://doi.org/10.1523/JNEUROSCI.5539-08.2009
Zeitlin R, Patel S, Solomon R et al (2012) Cotinine enhances the extinction of contextual fear memory and reduces anxiety after fear conditioning. Behav Brain Res 228:284–293. https://doi.org/10.1016/j.bbr.2011.11.023
Zhang H, Lin S-C, Nicolelis MAL (2010) Spatiotemporal coupling between hippocampal acetylcholine release and theta oscillations in vivo. J Neurosci 30:13431–13440. https://doi.org/10.1523/JNEUROSCI.1144-10.2010
Zheng J, Anderson KL, Leal SL et al (2017) Amygdala-hippocampal dynamics during salient information processing. Nat Commun 8:14413. https://doi.org/10.1038/ncomms14413
Funding
This research was funded by grants, from the German Research Foundation (CRC779 TPB5 and STO488/6) and by the federal state of Saxony-Anhalt and the “European Regional Development Fund” (ERDF 2007–2013), Vorhaben: Centre for Behavioural Brain Sciences (CBBS) to Oliver Stork. Gürsel Çalışkan is funded by CBBS ScienceCampus financed by the Leibniz Association (SAS-2015-LIN-LWC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
This article belongs to a Special Issue on Psychopharmacology of Extinction.
Glossary
- Coherence
-
is measure of synchrony between two LFP patterns based on changes in both amplitude and phase.
- Correlation
-
is measure of synchrony between two LFP patterns based on their phase similarity as a product of time. This measure is independent of amplitude changes in time and rather focuses on waveform similarity.
- Power (Volt2/Hz)
-
is a measure that gives information about the strength of LFP signal at a certain frequency range. In extracellular physiology, to determine the frequency of main oscillation power, scientists convert the LFP signal into a range of frequencies (Power Spectrum) as a function of their power in the LFP signal.
- Synchrony
-
is a measure to determine the functional communication between two regions in the brain by measuring the similarity of simultaneously recorded LFP oscillation patterns based on their fluctuations in phase and/or amplitude. The synchrony of underlying cellular firing can also be detected by measuring whether cells fire at a particular phase of oscillations recorded locally or in a remote brain region.
Rights and permissions
About this article
Cite this article
Çalışkan, G., Stork, O. Hippocampal network oscillations at the interplay between innate anxiety and learned fear. Psychopharmacology 236, 321–338 (2019). https://doi.org/10.1007/s00213-018-5109-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-5109-z