Abstract
Rationale
Although nicotine exposure upregulates the α4β2* subtype of nicotinic acetylcholine receptors (nAChRs), the upregulation of nAChRs in non-human primates voluntarily self-administering nicotine has never been demonstrated.
Objectives
The objective of the study is to determine if short access to nicotine in a non-human primate model of nicotine self-administration is sufficient to induce nAChRs upregulation.
Methods
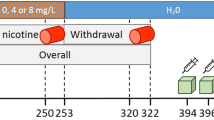
We combined a nicotine self-administration paradigm with in vivo measure of α4β2* nAChRs using 2-[18F]fluoro-A-85380 (2-FA) and positron emission tomography (PET) in six squirrel monkeys. PET measurement was performed before and after intravenous nicotine self-administration (unit dose 10 μg/kg per injection). Monkeys were trained to self-administer nicotine under a fixed-ratio (FR) schedule of reinforcement. Intermittent access (1 h daily per weekday) to nicotine was allowed for 4 weeks and levels of α4β2* nAChRs were measured 4 days later.
Results
This intermittent access was sufficient to induce upregulation of α4β2* receptors in the whole brain (31 % upregulation) and in specific brain areas (+36 % in amygdala and +62 % in putamen).
Conclusions
These results indicate that intermittent nicotine exposure is sufficient to produce change in nAChRs expression.



Similar content being viewed by others
References
Benwell ME, Balfour DJ, Anderson JM (1988) Evidence that tobacco smoking increases the density of (−)-[3H]nicotine binding sites in human brain. J Neurochem 50:1243–1247
Besson M, Granon S, Mameli-Engvall M, Cloez-Tayarani I, Maubourguet N, Cormier A, Cazala P, David V, Changeux JP, Faure P (2007) Long-term effects of chronic nicotine exposure on brain nicotinic receptors. Proc Natl Acad Sci U S A 104:8155–8160
Breese CR, Marks MJ, Logel J, Adams CE, Sullivan B, Collins AC, Leonard S (1997) Effect of smoking history on [3H]nicotine binding in human postmortem brain. J Pharmacol Exp Ther 282:7–13
Brody AL, Mukhin AG, La Charite J, Ta K, Farahi J, Sugar CA, Mamoun MS, Vellios E, Archie M, Kozman M, Phuong J, Arlorio F, Mandelkern MA (2013) Up-regulation of nicotinic acetylcholine receptors in menthol cigarette smokers. Int J Neuropsychopharmacol 16:957–966
Brody AL, Mukhin AG, Mamoun MS, Luu T, Neary M, Liang L, Shieh J, Sugar CA, Rose JE, Mandelkern MA (2014) Brain nicotinic acetylcholine receptor availability and response to smoking cessation treatment: a randomized trial. JAMA Psychiatry 71:797–805
Chefer SI, London ED, Koren AO, Pavlova OA, Kurian V, Kimes AS, Horti AG, Mukhin AG (2003) Graphical analysis of 2-[18F]FA binding to nicotinic acetylcholine receptors in rhesus monkey brain. Synapse 48:25–34
Department of Health and Human Services (1988) The Health Consequences of Smoking. Nicotine Addiction. Department of Health and Human Services (DHHS) Publication No. (CDC) 88–8406, Washington DC
Flores CM, Rogers SW, Pabreza LA, Wolfe BB, Kellar KJ (1992) A subtype of nicotinic cholinergic receptor in rat brain is composed of alpha 4 and beta 2 subunits and is up-regulated by chronic nicotine treatment. Mol Pharmacol 41:31–37
Flores CM, Davila-Garcia MI, Ulrich YM, Kellar KJ (1997) Differential regulation of neuronal nicotinic receptor binding sites following chronic nicotine administration. J Neurochem 69:2216–2219
Gergen GA, MacLean PD (1962) A Stereotaxic atlas of the Brain of the squirrel monkey (Saimiri Sciureus), U.S. Department of Health, Education, and Welfare. Public Health Service. National Institutes of Health. Bethesda, Maryland. U.S. Government Printing Office. Washington, D.C.
Goodwin AK, Hiranita T, Paule MG (2015) The reinforcing effects of nicotine in humans and nonhuman primates: a review of intravenous self-administration evidence and future directions for research. Nicotine Tob Res 17(11):1297–310
Goldberg SR, Spealman RD, Goldberg DM (1981) Persistent behavior at high rates maintained by intravenous self-administration of nicotine. Science 214:573–575
Gould RW, Duke AN, Nader MA (2014) PET studies in nonhuman primate models of cocaine abuse: translational research related to vulnerability and neuroadaptations. Neuropharmacology 84:138–151
Govind AP, Vezina P, Green WN (2009) Nicotine-induced upregulation of nicotinic receptors: underlying mechanisms and relevance to nicotine addiction. Biochem Pharmacol 78:756–765
Horti AG, Scheffel U, Koren AO, Ravert HT, Mathews WB, Musachio JL, Finley PA, London ED, Dannals RF (1998) 2-[18F]Fluoro-A-85380, an in vivo tracer for the nicotinic acetylcholine receptors. Nucl Med Biol 25:599–603
Horti AG, Kuwabara H, Holt DP, Dannals RF, Wong DF (2013) Recent PET radioligands with optimal brain kinetics for imaging nicotinic acetylcholine receptors. J Label Compd Radiopharm 56:159–166
Howell LL, Wilcox KM (2002) Functional imaging and neurochemical correlates of stimulant self-administration in primates. Psychopharmacology (Berlin) 163:352–361
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN, Holden J, Houle S, Huang SC, Ichise M, Iida H, Ito H, Kimura Y, Koeppe RA, Knudsen GM, Knuuti J, Lammertsma AA, Laruelle M, Logan J, Maguire RP, Mintun MA, Morris ED, Parsey R, Price JC, Slifstein M, Sossi V, Suhara T, Votaw JR, Wong DF, Carson RE (2007) Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27:1533–1539
Jacobs EH, Smit AB, de Vries TJ, Schoffelmeer AN (2003) Neuroadaptive effects of active versus passive drug administration in addiction research. Trends Pharmacol Sci 24:566–573
Justinova Z, Panlilio LV, Moreno-Sanz G, Redhi GH, Auber A, Secci ME, Mascia P, Bandiera T, Armirotti A, Bertorelli R, Chefer SI, Barnes C, Yasar S, Piomelli D, Goldberg SR (2015a) Effects of fatty acid amide hydrolase (FAAH) inhibitors in non-human primate models of nicotine reward and relapse. Neuropsychopharmacology 40:2185–2197
Justinova Z, Le Foll B, Redhi GH, Markou A, Goldberg SR (2015a) Differential effects of the metabotropic glutamate 2/3 receptor agonist LY379268 on nicotine versus cocaine self-administration and relapse in squirrel monkeys. Psychopharmacology (Berlin)
Kimes AS, Horti AG, London ED, Chefer SI, Contoreggi C, Ernst M, Friello P, Koren AO, Kurian V, Matochik JA, Pavlova O, Vaupel DB, Mukhin AG (2003) 2-[18F]F-A-85380: PET imaging of brain nicotinic acetylcholine receptors and whole body distribution in humans. FASEB J Off Publ Fed Am Soc Exp Biol 17:1331–1333
Kuryatov A, Luo J, Cooper J, Lindstrom J (2005) Nicotine acts as a pharmacological chaperone to up-regulate human alpha4beta2 acetylcholine receptors. Mol Pharmacol 68:1839–1851
Le Foll B, Chefer SI, Kimes AS, Shumway D, Goldberg SR, Stein EA, Mukhin AG (2007a) Validation of an extracerebral reference region approach for the quantification of brain nicotinic acetylcholine receptors in squirrel monkeys with PET and 2-18F-fluoro-A-85380. J Nucl Med 48:1492–1500
Le Foll B, Wertheim C, Goldberg SR (2007b) High reinforcing efficacy of nicotine in non-human primates. PLoS ONE 2:e230
Le Foll B, Chefer SI, Kimes AS, Shumway D, Stein EA, Mukhin AG, Goldberg SR (2009) Baseline expression of alpha4beta2* nicotinic acetylcholine receptors predicts motivation to self-administer nicotine. Biol Psychiatry 65:714–716
Lester HA, Xiao C, Srinivasan R, Son CD, Miwa J, Pantoja R, Banghart MR, Dougherty DA, Goate AM, Wang JC (2009) Nicotine is a selective pharmacological chaperone of acetylcholine receptor number and stoichiometry. Implications for drug discovery. AAPS J 11:167–177
Lomazzo E, Hussmann GP, Wolfe BB, Yasuda RP, Perry DC, Kellar KJ (2011) Effects of chronic nicotine on heteromeric neuronal nicotinic receptors in rat primary cultured neurons. J Neurochem 119:153–164
Marks MJ, Burch JB, Collins AC (1983) Effects of chronic nicotine infusion on tolerance development and nicotinic receptors. J Pharmacol Exp Ther 226:817–825
Mascia P, Pistis M, Justinova Z, Panlilio LV, Luchicchi A, Lecca S, Scherma M, Fratta W, Fadda P, Barnes C, Redhi GH, Yasar S, Le Foll B, Tanda G, Piomelli D, Goldberg SR (2011) Blockade of nicotine reward and reinstatement by activation of alpha-type peroxisome proliferator-activated receptors. Biol Psychiatry 69:633–641
Maskos U, Molles BE, Pons S, Besson M, Guiard BP, Guilloux JP, Evrard A, Cazala P, Cormier A, Mameli-Engvall M, Dufour N, Cloez-Tayarani I, Bemelmans AP, Mallet J, Gardier AM, David V, Faure P, Granon S, Changeux JP (2005) Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature 436:103–107
Mukhin AG, Kimes AS, Chefer SI, Matochik JA, Contoreggi CS, Horti AG, Vaupel DB, Pavlova O, Stein EA (2008) Greater nicotinic acetylcholine receptor density in smokers than in nonsmokers: a PET study with 2-18F-FA-85380. J Nucl Med 49:1628–1635
Perry DC, Davila-Garcia MI, Stockmeier CA, Kellar KJ (1999) Increased nicotinic receptors in brains from smokers: membrane binding and autoradiography studies. J Pharmacol Exp Ther 289:1545–1552
Picciotto MR, Mineur YS (2014) Molecules and circuits involved in nicotine addiction: The many faces of smoking. Neuropharmacology 76(Pt B):545–553
Picciotto MR, Zoli M, Rimondini R, Lena C, Marubio LM, Pich EM, Fuxe K, Changeux JP (1998) Acetylcholine receptors containing the beta2 subunit are involved in the reinforcing properties of nicotine. Nature 391:173–177
Picciotto MR, Addy NA, Mineur YS, Brunzell DH (2008) It is not “either/or”: activation and desensitization of nicotinic acetylcholine receptors both contribute to behaviors related to nicotine addiction and mood. Prog Neurobiol 84:329–342
Schwartz RD, Kellar KJ (1983) Nicotinic cholinergic receptor binding sites in the brain: regulation in vivo. Science 220:214–216
Staley JK, Krishnan-Sarin S, Cosgrove KP, Krantzler E, Frohlich E, Perry E, Dubin JA, Estok K, Brenner E, Baldwin RM, Tamagnan GD, Seibyl JP, Jatlow P, Picciotto MR, London ED, O’Malley S, van Dyck CH (2006) Human tobacco smokers in early abstinence have higher levels of beta2* nicotinic acetylcholine receptors than nonsmokers. J Neurosci 26:8707–8714
Tapper AR, McKinney SL, Nashmi R, Schwarz J, Deshpande P, Labarca C, Whiteaker P, Marks MJ, Collins AC, Lester HA (2004) Nicotine activation of alpha4* receptors: sufficient for reward, tolerance, and sensitization. Science 306:1029–1032
Valette H, Bottlaender M, Dolle F, Coulon C, Ottaviani M, Syrota A (2003) Long-lasting occupancy of central nicotinic acetylcholine receptors after smoking: a PET study in monkeys. J Neurochem 84:105–111
Valette H, Bottlaender M, Dolle F, Coulon C, Ottaviani M, Syrota A (2005) Acute effects of physostigmine and galantamine on the binding of [18F]fluoro-A-85380: a PET study in monkeys. Synapse 56:217–221
Vaupel DB, Stein EA, Mukhin AG (2007) Quantification of alpha4beta2* nicotinic receptors in the rat brain with microPET and 2-[18F]F-A-85380. Neuroimage 34:1352–1362
Wonnacott S (1990) The paradox of nicotinic acetylcholine receptor upregulation by nicotine. Trends Pharmacol Sci 11:216–219
Wullner U, Gundisch D, Herzog H, Minnerop M, Joe A, Warnecke M, Jessen F, Schutz C, Reinhardt M, Eschner W, Klockgether T, Schmaljohann J (2008) Smoking upregulates alpha4beta2* nicotinic acetylcholine receptors in the human brain. Neurosci Lett 430:34–37
Zambrano CA, Salamander RM, Collins AC, Grady SR, Marks MJ (2012) Regulation of the distribution and function of [(125)I]epibatidine binding sites by chronic nicotine in mouse embryonic neuronal cultures. J Pharmacol Exp Ther 342:245–254
Zambrano CA, Short CA, Salamander RM, Grady SR, Marks MJ (2015) Density of alpha4beta2* nAChR on the surface of neurons is modulated by chronic antagonist exposure. Pharmacol Res Perspect 3:e00111
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Financial support
This study was supported by the Intramural Research Program of the National Institute on Drug Abuse, NIH, DHHS.
Additional information
Steven R. Goldberg In memoriam
Rights and permissions
About this article
Cite this article
Le Foll, B., Chefer, S.I., Kimes, A.S. et al. Impact of short access nicotine self-administration on expression of α4β2* nicotinic acetylcholine receptors in non-human primates. Psychopharmacology 233, 1829–1835 (2016). https://doi.org/10.1007/s00213-016-4250-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4250-9