Abstract
Rationale
Nicotine intravenous self-administration (IVSA) in rats has been conducted using a variety of methodological procedures with equally variable results.
Objectives
Here, we addressed the importance of the type of response operandum and prior instrumental training with a natural reinforcer on nicotine IVSA and reinstatement.
Methods
Rats were tested for spontaneous acquisition of IVSA using either nose poke (NP) or lever press (LVR) operandum. A dose-response test was then conducted, followed by extinction and cue- and nicotine-induced reinstatement.
Results
The use of the NP operandum resulted in markedly higher levels of IVSA across acquisition and across dose-response testing compared with the LVR group. Whereas both groups reinstated following a nicotine prime, only the LVR group demonstrated cue-induced reinstatement. As a positive control, the experiment was repeated with cocaine as the reinforcer: equivalent levels of IVSA were observed across all tests, irrespective of operandum. When rats self-administering nicotine received instrumental training with a sucrose reinforcer prior to IVSA, a facilitated acquisition of IVSA was observed in both LVR and NP groups to a similar extent (the effect of operandum remained), but had little effect on responding thereafter. During reinstatement testing, both groups now displayed cue- and nicotine-induced reinstatement, but this was also evident in saline control animals that had never received nicotine.
Conclusions
These results suggest that, unlike cocaine, an increased physical response requirement can decrease nicotine intake. It also indicates that operandum and prior sucrose training may influence the role that visual cues play in nicotine dependence.





Similar content being viewed by others
References
Andreoli M, Tessari M, Pilla M, Valerio E, Hagan JJ, Heidbreder CA (2003) Selective antagonism at dopamine D3 receptors prevents nicotine-triggered relapse to nicotine-seeking behavior. Neuropsychopharmacology 28:1272–1280
Bespalov A, Lebedev A, Panchenko G, Zvartau E (1999) Effects of abused drugs on thresholds and breaking points of intracranial self-stimulation in rats. Eur Neuropsychopharmacol 9:377–383
Bespalov AY, Dravolina OA, Sukhanov I, Zakharova E, Blokhina E, Zvartau E, Danysz W, van Heeke G, Markou A (2005) Metabotropic glutamate receptor (mGluR5) antagonist MPEP attenuated cue- and schedule-induced reinstatement of nicotine self-administration behavior in rats. Neuropharmacology 49(Suppl 1):167–178
Bongiovanni M, See RE (2008) A comparison of the effects of different operant training experiences and dietary restriction on the reinstatement of cocaine-seeking in rats. Pharmacol Biochem Behav 89:227–233
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2001) Cue dependency of nicotine self-administration and smoking. Pharmacol Biochem Behav 70:515–530
Caggiula AR, Donny EC, Palmatier MI, Liu X, Chaudhri N, Sved AF (2009) The role of nicotine in smoking: a dual-reinforcement model. Nebr Symp Motiv 55:91–109
Caine SB, Negus SS, Mello NK (1999) Method for training operant responding and evaluating cocaine self-administration behavior in mutant mice. Psychopharmacology (Berl) 147:22–24
Clemens KJ, Caille S, Stinus L, Cador M (2009) The addition of five minor tobacco alkaloids increases nicotine-induced hyperactivity, sensitization and intravenous self-administration in rats. Int J Neuropsychopharmacol 12(10):1355–1366
David V, Polis I, McDonald J, Gold LH (2001) Intravenous self-administration of heroin/cocaine combinations (speedball) using nose-poke or lever-press operant responding in mice. Behav Pharmacol 12:25–34
De Vries TJ, de Vries W, Janssen MC, Schoffelmeer AN (2005) Suppression of conditioned nicotine and sucrose seeking by the cannabinoid-1 receptor antagonist SR141716A. Behav Brain Res 161:164–168
Deiana S, Fattore L, Spano MS, Cossu G, Porcu E, Fadda P, Fratta W (2007) Strain and schedule-dependent differences in the acquisition, maintenance and extinction of intravenous cannabinoid self-administration in rats. Neuropharmacology 52:646–654
Diergaarde L, de Vries W, Raaso H, Schoffelmeer AN, De Vries TJ (2008a) Contextual renewal of nicotine seeking in rats and its suppression by the cannabinoid-1 receptor antagonist Rimonabant (SR141716A). Neuropharmacology 55:712–716
Diergaarde L, Pattij T, Poortvliet I, Hogenboom F, de Vries W, Schoffelmeer AN, De Vries TJ (2008b) Impulsive choice and impulsive action predict vulnerability to distinct stages of nicotine seeking in rats. Biol Psychiatry 63:301–308
Donny EC, Caggiula AR, Knopf S, Brown C (1995) Nicotine self-administration in rats. Psychopharmacology (Berl) 122:390–394
Donny EC, Chaudhri N, Caggiula AR, Evans-Martin FF, Booth S, Gharib MA, Clements LA, Sved AF (2003) Operant responding for a visual reinforcer in rats is enhanced by noncontingent nicotine: implications for nicotine self-administration and reinforcement. Psychopharmacology (Berl) 169:68–76
Dravolina OA, Zakharova ES, Shekunova EV, Zvartau EE, Danysz W, Bespalov AY (2007) mGlu1 receptor blockade attenuates cue- and nicotine-induced reinstatement of extinguished nicotine self-administration behavior in rats. Neuropharmacology 52:263–269
Fattore L, Spano MS, Cossu G, Scherma M, Fratta W, Fadda P (2009) Baclofen prevents drug-induced reinstatement of extinguished nicotine-seeking behaviour and nicotine place preference in rodents. Eur Neuropsychopharmacol 19:487–498
Forget B, Coen KM, Le Foll B (2009) Inhibition of fatty acid amide hydrolase reduces reinstatement of nicotine seeking but not break point for nicotine self-administration-comparison with CB(1) receptor blockade. Psychopharmacology (Berl) 205:613–624
Gallus S, Schiaffino A, La Vecchia C, Townsend J, Fernandez E (2006) Price and cigarette consumption in Europe. Tob Control 15:114–119
Goeders JE, Murnane KS, Banks ML, Fantegrossi WE (2009) Escalation of food-maintained responding and sensitivity to the locomotor stimulant effects of cocaine in mice. Pharmacol Biochem Behav 93:67–74
Guillem K, Vouillac C, Azar MR, Parsons LH, Koob GF, Cador M, Stinus L (2005) Monoamine oxidase inhibition dramatically increases the motivation to self-administer nicotine in rats. J Neurosci 25:8593–8600
Henningfield JE, Benowitz NL, Slade J, Houston TP, Davis RM, Deitchman SD (1998) Reducing the addictiveness of cigarettes. Council on Scientific Affairs, American Medical Association. Tob Control 7:281–293
Hughes JR, Goldstein MG, Hurt RD, Shiffman S (1999) Recent advances in the pharmacotherapy of smoking. Jama 281:72–76
LeSage MG, Burroughs D, Dufek M, Keyler DE, Pentel PR (2004) Reinstatement of nicotine self-administration in rats by presentation of nicotine-paired stimuli, but not nicotine priming. Pharmacol Biochem Behav 79:507–513
Liu X, Caggiula AR, Yee SK, Nobuta H, Sved AF, Pechnick RN, Poland RE (2007a) Mecamylamine attenuates cue-induced reinstatement of nicotine-seeking behavior in rats. Neuropsychopharmacology 32:710–718
Liu X, Palmatier MI, Caggiula AR, Donny EC, Sved AF (2007b) Reinforcement enhancing effect of nicotine and its attenuation by nicotinic antagonists in rats. Psychopharmacology (Berl) 194:463–473
Liu X, Caggiula AR, Palmatier MI, Donny EC, Sved AF (2008) Cue-induced reinstatement of nicotine-seeking behavior in rats: effect of bupropion, persistence over repeated tests, and its dependence on training dose. Psychopharmacology (Berl) 196:365–375
MacKillop J, Murphy JG, Ray LA, Eisenberg DT, Lisman SA, Lum JK, Wilson DS (2008) Further validation of a cigarette purchase task for assessing the relative reinforcing efficacy of nicotine in college smokers. Exp Clin Psychopharmacol 16:57–65
Manzardo AM, Stein L, Belluzzi JD (2002) Rats prefer cocaine over nicotine in a two-lever self-administration choice test. Brain Res 924:10–19
Panlilio LV, Goldberg SR (2007) Self-administration of drugs in animals and humans as a model and an investigative tool. Addiction 102:1863–1870
Paterson NE, Froestl W, Markou A (2005) Repeated administration of the GABAB receptor agonist CGP44532 decreased nicotine self-administration, and acute administration decreased cue-induced reinstatement of nicotine-seeking in rats. Neuropsychopharmacology 30:119–128
Raiff BR, Dallery J (2009) Responding maintained by primary reinforcing visual stimuli is increased by nicotine administration in rats. Behav Process 82:95–99
Rose JE (2006) Nicotine and nonnicotine factors in cigarette addiction. Psychopharmacology (Berl) 184:274–285
Samaha AN, Yau WY, Yang P, Robinson TE (2005) Rapid delivery of nicotine promotes behavioral sensitization and alters its neurobiological impact. Biol Psychiatry 57:351–360
Shaham Y, Adamson LK, Grocki S, Corrigall WA (1997) Reinstatement and spontaneous recovery of nicotine seeking in rats. Psychopharmacology (Berl) 130:396–403
Shoaib M, Schindler CW, Goldberg SR (1997) Nicotine self-administration in rats: strain and nicotine pre-exposure effects on acquisition. Psychopharmacology (Berl) 129:35–43
Shram MJ, Funk D, Li Z, Le AD (2008) Nicotine self-administration, extinction responding and reinstatement in adolescent and adult male rats: evidence against a biological vulnerability to nicotine addiction during adolescence. Neuropsychopharmacology 33:739–748
Sorge RE, Clarke PB (2009) Rats self-administer intravenous nicotine delivered in a novel smoking-relevant procedure: effects of dopamine antagonists. J Pharmacol Exp Ther 330:633–640
Stolerman IP, Jarvis MJ (1995) The scientific case that nicotine is addictive. Psychopharmacology (Berl) 117:2–10, discussion 14–20
Substance Abuse and Mental Health Services Administration (2009) Results from the 2008 National Survey on Drug Use and Health: National Findings (Office of Applied Studies, NSDUH Series H-36, HHS Publication No. SMA 09-4434). Rockville, MD
West R, McEwen A, Bolling K, Owen L (2001) Smoking cessation and smoking patterns in the general population: a 1-year follow-up. Addiction 96:891–902
Acknowledgments
The authors are solely responsible for the scientific content of this paper. This research and KJC were supported by funding from the Association pour la Recherche sur les Nicotianées (ARN), Centre National de la Recherche Scientifique, Universities Bordeaux 1 and 2 and Region Aquitaine, France. The authors would also like to thank Anne Fayoux and Stephane Lelgouach for animal care, Marie-Hélène Bruyères for administrative assistance and Dr. Nathan Holmes for comments on the manuscript.
Disclosure/conflict of interest
The authors have no known conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research and KJC were supported by funding from the Association pour la Recherche sur les Nicotianées (ARN), France.
Electronic supplementary material
Below is the link to the electronic supplementary material.
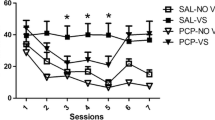
Supplementary Fig. 1
Extinction of responding in rats previously lever pressing (LVR) or nose-poking (NP) for A) nicotine, B) cocaine, C) saline, D) nicotine in rats previously trained with sucrose and E) saline in rats previously trained with sucrose. The first data point (FR-5) represents the mean responding across the last 3 days of responding on a FR-5 schedule. n = 4-10 rats per group. (GIF 86.2 kb)
Rights and permissions
About this article
Cite this article
Clemens, K.J., Caillé, S. & Cador, M. The effects of response operandum and prior food training on intravenous nicotine self-administration in rats. Psychopharmacology 211, 43–54 (2010). https://doi.org/10.1007/s00213-010-1866-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-010-1866-z