Summary
-
1.
Directionally and amplitude-controlled mechanical pulses were applied to identified sites of the tail cilia and the posterior soma membrane ofParamecium under voltage clamp.
-
2.
Lateral stimuli directed toward more distal sections of the cilia deflected the ciliary bundle as a unit. No deformations of the soma membrane were detected upon stimulus application to the ciliary shafts (Fig. 3).
-
3.
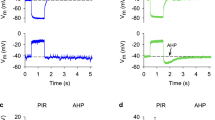
Outward receptor currents following somatic stimulation arose 3 ms after the onset of the electric driving pulse (= response time). The response times after ciliary stimulation were at least 6 ms.
-
4.
The response times rose from 6 to beyond 10 ms after shifts of the stimulus probe from proximal to more distal sections of the tail cilia.
-
5.
Maximal currents (up to 17 nA) were recorded upon stimulation of the apical posterior soma in the center of the tail. Ciliary stimulation elicited currents which had more irregular time courses of rise and decay.
-
6.
In most cells tested the current amplitudes were reduced with shifts from somatic to ciliary stimulation; the amplitudes even more decreased with stimulus displacement toward the ciliary tips. A fraction of cells was hypersensitive in that the responses to somatic and ciliary stimulation were enhanced as compared to normal cells.
-
7.
The tail cilia showed no intrinsic directional sensitivity. Minor differences in responsiveness to topographically identified lateral stimulation of the cilia are presumably related to a dorso-ventral differentiation of somatic sensitivity.
-
8.
We conclude that neither the ciliary membrane nor the axoneme and basal body play an active role in mechanotransduction. A local ciliary stimulus appears to be ‘defocused’ so that it affects an extended area of the sensitive soma membrane.
Similar content being viewed by others
References
Blake JR, Sleigh MA (1974) Mechanics of ciliary locomotion. Biol Rev 49:85–125
Corey DP, Hudspeth AJ (1979) Response latency of vertebrate hair cells. Biophys J 26:499–506
Deitmer JW (1981) Voltage and time characteristics of the potassium mechanoreceptor current in the ciliateStylonychia. J Comp Physiol 141:173–182
Erler G (1983) Sensitivity of an insect mechanoreceptor after destruction of dendritic microtubules by means of vinblastine. Cell Tissue Res 229:673–684
Flock A (1971) Sensory transduction in hair cells. In: Loewenstein WR (ed) Principles of receptor physiology. Springer, Berlin Heidelberg New York (Handbook of sensory physiology, vol I, pp 396–441)
Flock A, Cheung HC (1977) Actin filaments in sensory hairs of inner ear receptor cells. J Cell Biol 75:339–343
Hudspeth AJ, Corey DP (1977) Sensitivity, polarity, and conductance change in the response of vertebrate hair cells to controlled mechanical stimuli. Proc Natl Acad Sci USA 74:2407–2411
Hudspeth AJ, Jacobs R (1979) Stereocilia mediate transduction in vertebrate hair cells. Proc Natl Acad Sci USA 76:1506–1509
Lowenstein O, Osborne MP, Wersäll J (1964) Structure and innervation of the sensory epithelia of the labyrinth in the Thornback ray (Raja clavata). Proc R Soc London Ser B 160:1–12
Machemer H, Machemer-Röhnisch S (1983) Tail cilia ofParamecium passively transmit mechanical stimuli to the cell soma. J Submicrosc Cytol 15:281–284
Machemer H, Ogura A (1979) Ionic conductances of membranes in ciliated and deciliatedParamecium. J Physiol 296:49–60
Murakami A, Machemer H (1982) Mechanoreception and signal transmission in the lateral ciliated cells on the gill ofMytilus. J Comp Physiol 145:351–362
Naitoh Y, Eckert R (1969) Ionic mechanisms controlling behavioural responses ofParamecium to mechanical stimulation. Science 164:963–965
Naitoh Y, Eckert R (1972) Electrophysiology in the ciliate protozoa. In: Kerkut GA (ed) Experiments in physiology and biochemistry, vol V. Academic Press, New York, pp 17–38
Ogura A, Machemer H (1980) Distribution of mechanoreceptor channels in theParamecium surface membrane. J Comp Physiol 135:233–242
Ogura A, Takahashi K (1976) Artificial deciliation causes loss of calcium-dependent responses inParamecium. Nature (Lond) 264:170–172
Peyer JE de, Machemer H (1977) Membrane excitability inStylonychia: properties of the two-peak regenerative Ca response. J Comp Physiol 121:15–32
Pitelka DR (1968) Fibrillar systems in protozoa. In: Chen TT (ed) Research in protozoology. Pergamon Press, Oxford New York, vol III, pp 280–388
Rohde K (1958) Der periodische Wechsel der Bewegungsformen vonParamecium caudatum und seine Abhängigkeit von äußeren und inneren Faktoren. Z Wiss Zool 161:277–345
Satir P (1968) Studies on cilia. III. Further studies on the cilium tip and a sliding filament model of ciliary motility. J Cell Biol 39:77–94
Stommel EW, Stephens RE, Alkon DL (1980) Motile statocyst cilia transmit rather than directly transduce mechanical stimuli. J Cell Biol 87:652–662
Thurm U (1968) Steps in the transducer process of mechanoreceptors. Symp Zool Soc Lond 23:199–216
Thurm U (1982) Mechanoelektrische Transduktion. In: Hoppe W, Lohmann W, Markl H, Ziegler H (Hrsg) Biophysik, 2. Aufl. Springer, Berlin Heidelberg New York, pp 691–696
Thurm U, Erler G, Gödde J, Kastrup H, Keil T, Völker W, Vohwinkel B (1983) Cilia specialized for mechanoreception. J Submicrosc Cytol 15:151–155
Author information
Authors and Affiliations
Additional information
Dedicated to Karl G. Grell, on the occasion of his 70th birthday
Rights and permissions
About this article
Cite this article
Machemer-Röhnisch, S., Machemer, H. Receptor current following controlled stimulation of immobile tail cilia inParamecium caudatum . J. Comp. Physiol. 154, 263–271 (1984). https://doi.org/10.1007/BF00604992
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00604992