Summary
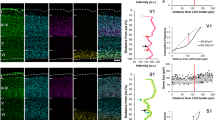
The origin and laminar arrangement of the homolateral and callosal projections to the anterior (AAF), primary (AI), posterior (PAF) and secondary (AII) auditory cortical areas were studied in the cat by means of electrophysiological recording and WGA-HRP tracing techniques. The transcallosal projections to AAF, AI, PAF and AII were principally homotypic since the major source of input was their corresponding area in the contralateral cortex. Heterotypic transcallosal projections to AAF and AI were seen, originating from the contralateral AI and AAF, respectively. PAF received heterotypic commissural projections from the opposite ventroposterior auditory cortical field (VPAF). Heterotypic callosal inputs to AII were rare, originating from AAF and AI. The neurons of origin of the transcallosal connections were located mainly in layers II and III (70–92%), and less frequently in deep layers (V and VI, 8–30%). Single unit recordings provided evidence that both homotypic and heterotypic transcallosal projections connect corresponding frequency regions of the two hemispheres. The regional distribution of the anterogradely labeled terminals indicated that the homotypic and heterotypic auditory transcallosal projections are reciprocal. The present data suggest that the transcallosal auditory interconnections are segregated in 3 major parallel components (AAF-AI, PAF-VPAF and AII), maintaining a segregation between parallel functional channels already established for the thalamocortical auditory interconnections. For the intrahemispheric connections, the analysis of the retrograde tracing data revealed that AAF and AI receive projections from the homolateral cortical areas PAF, VPAF and AII, whose neurons of origin were located mainly in their deep (V and VI) cortical layers. The reciprocal interconnections between the homolateral AAF and AI did not show a preferential laminar arrangement since the neurons of origin were distributed almost evenly in both superficial (II and III) and deep (V and VI) cortical layers. On the contrary, PAF received inputs from the homolateral cortical fields AAF, AI, AII and VPAF, originating predominantly from their superficial (II and III) layers. The homolateral projections reaching AII originated mainly from the superficial layers of AAF and AI, but from the deep layers of VPAF and PAF. The laminar distribution of anterogradely labeled terminal fields, when they were dense enough for a confident identification, was systematically related to the laminar arrangement of neurons of origin of the reciprocal projection: a projection originating from deep layers was associated with a reciprocal projection terminating mainly in layer IV, whereas a projection originating from superficial layers was associated with a reciprocal projection terminating predominantly outside layer IV. This laminar distribution indicates that the homolateral auditory cortical interconnections have a feed-forward/feed-back organization, corresponding to a hierarchical arrangement of the auditory cortical areas, according to criteria previously established in the visual system of primates. The principal auditory cortical areas could be ranked into 4 distinct hierarchical levels. The tonotopically organized areas AAF and AI represent the lowest level. The second level corresponds to the non-tonotopically organized area AII. Higher, the tonotopically organized areas VPAF and PAF occupy the third and fourth hierarchical levels, respectively.
Similar content being viewed by others
Abbreviations
- AAF:
-
anterior auditory cortical area
- AI:
-
primary auditory cortical area
- AII:
-
secondary auditory cortical area
- BF:
-
best frequency
- C:
-
cerebral cortex
- CA:
-
caudate nucleus
- CL:
-
claustrum
- D:
-
dorsal nucleus of the dorsal division of the MGB
- ea:
-
anterior ectosylvian sulcus
- ep:
-
posterior ectosylviansulcus
- IC:
-
internal capsule
- LGN:
-
lateral geniculate nucleus
- LV:
-
pars lateralis of the ventral division of the MGB
- LVe:
-
lateral ventricule
- M:
-
pars magnocellularis of the medial division of the MGB
- MGB:
-
medial geniculate body
- MGBv:
-
ventral division of the MGB
- OT:
-
optic tract
- OV:
-
pars ovoidea of the ventral division of the MGB
- PAF:
-
posterior auditory cortical area
- PH:
-
parahippocampal cortex
- PO:
-
lateral part of the posterior group of thalamic nuclei
- PU:
-
putamen
- RE:
-
reticular complex of thalamus
- rs:
-
rhinal sulcus
- SG:
-
suprageniculate nucleus of the dorsal division of the MGB
- ss:
-
suprasylvian sulcus
- TMB:
-
tetrametylbenzidine
- VBX:
-
ventrobasal complex
- VLa:
-
ventrolateral complex
- VL:
-
ventro-lateral nucleus of the ventral division of the MGB
- WGA-HRP:
-
wheat germ agglutinin conjugated to horse-radish peroxidase
- WM:
-
white matter
- VPAF:
-
ventro-posterior auditory cortical area
References
Andersen RA, Knight PL, Merzenich MM (1980a) The thalamocortical and corticothalamic connections of AI, AII, and the anterior auditory field (AAF) in the cat: evidence for two largely segregated systems of connections. J Comp Neurol 194: 663–701
Andersen RA, Snyder RL, Merzenich MM (1980b) The topographic organization of corticocollicular projections from physiologically identified loci in the AI, AII, and anterior auditory cortical fields of the cat. J Comp Neurol 191: 479–494
Andersen RA, Roth GL, Aitkin LM, Merzenich MM (1980c) The efferent projections of the central nucleus and the pericentral nucleus of the inferior colliculus in the cat. J Comp Neurol 194: 649–662
Blum PS, Abraham LD, Gilman S (1979) Vestibular, auditory and somatic input to the posterior thalamus of the cat. Exp Brain Res 34: 1–9
Calford MB (1983) The parcellation of the medial geniculate body of the cat defined by the auditory response properties of single units. J Neurosci 3: 2350–2364
Calford MB, Aitkin LM (1983) Ascending projections to the medial geniculate body of the cat: Evidence for multiple parallel auditory pathways through thalamus. J Neurosci 3: 2365–2380
Code RA, Winer JA (1985) Commissural neurons in layer III of cat primary auditory cortex (AI): Pyramidal and nonpyramidal cell input. J Comp Neurol 242: 485–510
Code RA, Winer JA (1986) Columnar organization and reciprocity of commissural connections in cat primary auditory cortex (AI). Hearing Res 23: 205–222
Coogan TA, Burkhalter A (1990) Conserved patterns of corticocortical connections define areal hierarchy in rat visual cortex. Exp Brain Res 80: 49–53
Diamond IT, Jones EG and Powell TPS (1968) Interhemispheric fiber connections of the auditory cortex of the cat. Brain Res 11: 177–193
Fitzpatrick KA, Imig TJ (1980) Auditory cortico-cortical connections in the owl monkey. J Comp Neurol 192: 589–610
Galaburda AM, Pandya DN (1983) The intrinsic architectonic and connectional organization of the superior temporal region of the rhesus monkey. J Comp Neurol 221: 169–184
Hubel DH, Livingstone MS (1987) Segregation of form, color, and stereopsis in primate area 18. J Neurosci 7: 3378–3415
Imig TJ, Adrian HO (1977) Binaural columns in the primary field (AI) of cat auditory cortex. Brain Res 138: 241–257
Imig TJ, Brugge JF (1978) Sources and terminations of callosal axons related to binaural and frequency maps in primary auditory cortex of the cat. J Comp Neurol 182: 637–660
Imig TJ, Reale RA (1980) Patterns of corticocortical connections related to tonotopic maps in cat auditory cortex. J Comp Neurol 192: 293–332
Imig TJ, Reale RA (1981) Ipsilateral corticocortical projections related to binaural columns in cat primary auditory cortex. J Comp Neurol 203: 1–14
Imig TJ, Morel A (1985) Tonotopic organization in lateral part of posterior group of thalamic nuclei in the cat. J Neurophysiol 53: 836–851
Imig TJ, Morel A, Dale Kauer C (1982) Covariations of distributions of callosal cell bodies and callosal axon terminals in layer III of cat primary auditory cortex (AI). Brain Res 251: 157–159
Kelly JB, Wong D (1981) Laminar connections of the cat's auditory cortex. Brain Res 212: 1–15
Khorevin VI (1978) Responses of units in the magnocellular part of the medial geniculate body to acoustic and somatosensory stimulation. Neurophysiology 10: 89–94
Khorevin VI (1980) Interaction between responses evoked by acoustic and somatosensory stimuli in neurons of the magnocellular part of the medial geniculate body. Neurophysiology 12: 241–245
Knight PL (1977) Representation of the cochlea within the anterior auditory field (AAF) of the cat. Brain Res 130: 447–467
Kudo M, Niimi K (1978) Ascending projections of the inferior colliculus onto the medial geniculate body in the cat studied by anterograde and retrograde tracing techniques. Brain Res 155: 113–117
Kudo M, Niimi K (1980) Ascending projections of the inferior colliculus in the cat: an autoradiographic study. J Comp Neurol 191: 545–556
Livingstone MS, Hubel DH (1987a) Connections between layer 4B of area 17 and the thick cytochrome oxidase stripes of area 18 in the squirrel monkey. J Neurosci 7: 3371–3377
Livingstone MS, Hubel DH (1987b) Psychophysical evidence for separate channels for the perception of form, color, movement, and depth. J Neurosci 7: 3416–3468
Love JA, Scott JW (1969) Some response characteristics of cells of the magnocellular division of the medial geniculate body of the cat. Can J Physiol Pharmacol 47: 881–888
Luethke LE, Krubitzer LA, Kaas JH (1988) Cortical connections of electrophysiologically and architectonically defined subdivisions of auditory cortex in squirrels. J Comp Neurol 268: 181–203
Merzenich MM, Knight PL, Roth GL (1975) Representation of cochlea within primary auditory cortex in the cat. J Neurophysiol 38: 231–249
Mesulam MM (1982) Principles of horseradish peroxidase neurohistochemistry and their applications for tracing neural pathways. In: Mesulam MM, ed Tracing neural connections with HRP, IBRO Press, Wiley pp 1–151
Mitani A, Itoh K, Mizuno N (1987) Distribution and size of thalamic neurons projecting to layer I of the auditory cortical fields of the cat compared to those projecting to layer IV. J Comp Neurol 257: 105–121
Morel A, Imig TJ (1987) Thalamic projections to fields A, AI, P and VP in cat auditory cortex. J Comp Neurol 265: 119–144
Morel A, Rouiller E, de Ribaupierre Y, de Ribaupierre F (1987) Tonotopic organization in the medial geniculate body (MGB) of lightly anesthetized cats. Exp Brain Res 69: 24–42
Morest DK (1964) The neuronal architecture of the medial geniculate body of the cat. J Anat 98: 611–630
Morest DK (1965) The lateral tegmental system of the midbrain and the medial geniculate body: a study with Golgi and Nauta methods in cat. J Anat 99: 611–634
Niimi K, Matsuoka H (1979) Thalamocortical organization of the auditory system in the cat: studies by retrograde axonal transport of HRP. Adv Anat Embryol Cell Biol 57: 1–56
Niimi K, Ono K, Kusunose M (1984) Projections of the medial geniculate nucleus to layer 1 of the auditory cortex in the cat traced with horseradish peroxidase. Neurosci Lett 45: 223–228
Phillips DP, Irvine DRF (1981) Responses of single neurons in physiologically defined area AI of cat cerebral cortex: sensitivity to interaural intensity difference. Hearing Res 4: 299–307
Phillips DP, Irvine DRF (1982) Properties of single neurons in the anterior auditory field (AAF) of cat cerebral cortex. Brain Res 248: 237–244
Phillips DP, Orman SS (1984) Responses of single neurons in posterior field of cat auditory cortex to tonal stimulation. J Neurophysiol 51: 147–163
Poggio GF, Mountcastle VB (1960) A study of the functional contributions of the lemniscal and spinothalamic systems to somatic sensibility. Bull Johns Hopkins Hosp 106: 266–316
Reale RA, Imig TJ (1980) Tonotopic organization of auditory cortex in the cat. J Comp Neurol 192: 265–291
Rodrigues-Dagaeff C, Simm G, de Ribaupierre Y, Villa A, de Ribaupierre F, Rouiller EM (1989) Functional organization of the ventral division of the medial geniculate body of the cat: evidences for a rostro-caudal gradient of response properties and cortical projections. Hearing Res 39: 103–126
Rose JE (1949) The cellular structure of the auditory region of the cat. J Comp Neurol 91: 409–439
Rouiller E, de Ribaupierre Y, de Ribaupierre F (1979) Phase-locked responses to low-frequency tones in the medial geniculate body. Hearing Res 1: 213–226
Rouiller E, de Ribaupierre F (1982) Neurons sensitive to narrow ranges of repetitive acoustic transients in the medial geniculate body of the cat. Exp Brain Res 4: 323–326
Rouiller EM, de Ribaupierre F (1985) Origin of afferents to physiologically defined regions of the medial geniculate body of the cat: ventral and dorsal divisions. Hearing Res 19: 97–114
Rouiller EM, Rodrigues-Dagaeff C, Simm G, de Ribaupierre Y, Villa A, de Ribaupierre F (1989a) The functional organization of the medial division of the medial geniculate body of cat: tonotopic organization, spatial distribution of response properties and cortical connections. Hearing Res 39: 127–142
Rouiller EM, Hornung JP, de Ribaupierre F (1989b) Extrathalamic ascending projections to physiologically identified fields of the cat auditory cortex. Hearing Res 40: 233–246
Rouiller EM, Innocenti GM, de Ribaupierre F (1990) Interconnections of the auditory cortical fields of the cat with the cingulate and parahippocampal cortices. Exp Brain Res 80: 501–511
Rüttgers K, Ashoff A, Friauf E (1990) Commissural connections between the auditory cortices of the rat. Brain Res 509: 71–79
Schreiner CE, Cynader MS (1984) Basic functional organization of second auditory cortical field (AII) of the cat. J Neurophysiol 51: 1284–1305
Schreiner CE, Urbas JV (1988) Representation of amplitude modulation in the auditory cortex of the cat: II Comparison between cortical fields. Hearing Res 32: 49–64
Suga N (1984) Neural mechanisms of complex-sound processing for echolocation. Trends Neurosci 7: 20–27
Toros-Morel A, de Ribaupierre F, Rouiller E (1981) Coding properties of the different nuclei of the cat's medial geniculate body. In: Syka J, Aitkin L (eds) Neuronal mechanisms of hearing, Plenum Press, New York London, pp 239–243
van Essen DC (1985) Functional organization of primate visual cortex. In: Peters A, Jones EG, (eds) Cerebral cortex, volume 3, visual cortex. Plenum Press, New York London, pp 259–329
van Essen DC, Maunsell JHR (1983) Hierarchical organization and functional streams in the visual cortex. Trends Neurosci 6: 370–375
Vaughan D (1983) Thalamic and callosal connections of the rat auditory cortex. Brain Res 260: 181–189
Wepsic JG (1966) Multimodal sensory activation of cells in the magnocellular medial geniculate nucleus. Exp Neurol 15: 299–318
Winer JA (1984a) Anatomy of layer IV in cat primary auditory cortex (AI). J Comp Neurol 224: 535–567
Winer JA (1984b) The pyramidal neurons in layer III of cat primary auditory cortex (AI). J Comp Neurol 229: 476–496
Winer JA (1984c) The non-pyramidal cells in layer III of cat primary auditory cortex (AI). J Comp Neurol 229: 512–530
Winer JA (1985) Structure of layer II in cat primary auditory cortex (AI). J Comp Neurol 238: 10–37
Winguth SD, Winer JA (1986) Corticocortical connections of cat primary auditory cortex (AI): Laminar organization and identification of supragranular neurons projecting to area AIL. J Comp Neurol 248: 36–56
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rouiller, E.M., Simm, G.M., Villa, A.E.P. et al. Auditory corticocortical interconnections in the cat: evidence for parallel and hierarchical arrangement of the auditory cortical areas. Exp Brain Res 86, 483–505 (1991). https://doi.org/10.1007/BF00230523
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230523