Abstract
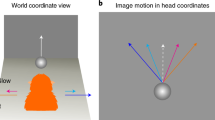
In order to direct a movement towards a visual stimulus, visualspatial information must be combined with postural information1. For example, directing gaze (eye plus head) towards a visible target requires the combination of retinal image location with eye and head position to determine the location of the target relative to the body. Similarly, world-referenced postural information is required to determine where something lies in the world. Posterior parietal neurons recorded in monkeys combine visual information with eye and head position2,3,4. A population of such cells could make up a distributed representation of target location in an extraretinal frame of reference4,5,6,7. However, previous studies have not distinguished between world-referenced and body-referenced signals4,8. Here we report that modulations of visual signals (gain fields) in two adjacent cortical fields, LIP and 7a, are referenced to the body and to the world, respectively. This segregation of spatial information is consistent with a streaming of information, with one path carrying body-referenced information for the control of gaze, and the other carrying world-referenced information for navigation and other tasks that require an absolute frame of reference.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Andersen, R. A., Snyder, L. H., Li, C. S. & Stricanne, B. Coordinate transformations in the representation of spatial information. Curr. Opin. Neurobiol. 3, 171–176 (1993).
Andersen, R. A. & Mountcastle, V. B. The influence of the angle of gaze upon the excitability of the light sensitive neurons of the posterior parietal cortex. J. Neurosci. 3, 532–548 (1983).
Anderson, R. A., Essick, G. K. & Siegel, R. M. Encoding of spatial location by posterior parietal neurons. Science 230, 456–458 (1985).
Brotchie, P. R., Andersen, R. A., Snyder, L. H. & Goodman, S. J. Head position signals used by parietal neurons to encode locations of visual stimuli. Nature 385, 232–235 (1995).
Zipser, D. & Andersen, R. A. Aback-propagation programmed network that simulates response properties of a subset of posterior parietal neurons. Nature 331, 679–684 (1988).
Salinas, E. & Abbott, L. F. Transfer of coded information from sensory to motor networks. J. Neurosci. 15, 6461–6474 (1995).
Pouget, A. & Sejnowski, T. J. Spatial representations in the parietal cortex may use basis functions. Adv. Neural Inf. Process. 7, 157–164 (1995).
Duhamel, J.-R., Bremmer, F., BenHamed, S. & Graf, W. Spatial invariance of visual receptive fields in parietal cortex neurons. Nature 389, 845–848 (1997).
Von Holst, E. & Mittelstaedt, H. Das Reafferenzprinzip (Wechselwirkungen zwischen Zentralnervensystem und Peripherie). Naturwissenschaften 37, 464–475 (1950). (In German.)
Mergner, T., Nardi, G. L., Becker, W. & Deecke, L. The role of canal–neck interaction for the perception of horizontal trunk and head rotation. Exp. Brain Res. 49, 198–208 (1983).
Roll, R., Velay, J. L. & Roll, J. P. Eye and neck proprioceptive messages contribute to the spatial coding of retinal input in visually oriented activities. Exp. Brain Res. 85, 423–431 (1991).
Karnath, H.-O., Sievering, D. & Fetter, M. The interactive contribution of neck muscle proprioception and vestibular stimulation to subjective “straight ahead” orientation in man. Exp. Brain Res. 101, 140–146 (1994).
Jones, G. M. & Milsum, J. H. Spatial and dynamic aspects of visual fixation. IEEE Trans. Biomed. Eng. 12(2), 54–62 (1965).
Robinson, D. A. in Basic Mechanisms of Ocular Motility and their Clinical Implications (eds Lennerstrand, G. & Bach-y-Rita, P.) 337–374 (Pergamon, Oxford, (1975)).
Andersen, R. A., Asanuma, C., Essick, G. & Siegel, R. M. Corticocortical connections of anatomically and physiologically defined sub-divisions within the inferior parietal lobule. J. Comp. Neurol. 296, 65–113 (1990).
Barash, S., Bracewell, R. M., Fogassi, L., Gnadt, J. W. & Andersen, R. A. Saccade-related activity in the lateral intraparietal area. I. Temporal properties. J. Neurophysiol. 66, 1095–1108 (1991).
Shibutani, H., Sakata, H. & Hyvarinen, J. Saccade and blinking evoked by microstimulation of the posterior parietal association cortex of monkey. Exp. Brain Res. 55, 1–8 (1984).
Thier, P. & Andersen, R. A. Electrical microstimulation suggests two different forms of representation of head-centered space in the intraparietal sulcus of rhesus monkeys. Proc. Natl Acad. Sci. USA 93, 4962–4967 (1996)
Snyder, L. H., Batista, A. P. & Andersen, R. A. Coding of intention in the posterior parietal cortex. Nature 386, 167–170 (1997).
Freedman, E. G. & Sparks, D. L. Activity of cells in the deeper layers of the superior colliculus of the rhesus monkey: evidence for a gaze displacement command. J. Neurophysiol. 78, 1669–1690 (1997).
Aguirre, G. K., Detre, J. A., Alsop, D. C. & D'Esposito, M. The parahippocampus subserves topographic learning in man. Cereb. Cortex 6, 823–829 (1996).
Squire, L. R. Memory and the hippocampus: a synthesis of findings from rat, monkey and humans. Psychol. Rev. 99, 195–231 (1992).
McNaughton, B. L., Barnes, C. A. & O'Keefe, J. The contributions of position, direction and velocity to single unit activity in the hippocampus of freely-moving rats. Exp. Brain Res. 52, 41–49 (1983).
Baylis, G. C. & Moore, B. O. Hippocampal lesions impair spatial response selection in the primate. Exp. Brain Res. 98, 110–118 (1994).
Rolls, E. T. & O'Mara, S. M. View-responsive neurons in the primate hippocampal complex. Hippocampus 5, 409–424 (1995).
Van Essen, D. C. & Maunsell, J. H. R. Hierarchical organization and functional streams in the visual cortex. Trends Neurosci. 6, 370–375 (1983).
Goodale, M. A. & Milner, A. D. Separate visual pathways for perception and action. Trends Neurosci. 15, 20–25 (1992).
Fredrickson, J. M., Schwarz, D. & Kornhumber, H. H. Convergence and interaction of vestibular and deep somatic afferents upon neurons in the vestibular nuclei of cat. Acta Otolaryngol. 61, 168–188 (1966).
Acknowledgements
We thank G. Roberts and B. Gillikin for technical assistance, and S. Gertmenian for editorial assistance. This work was supported by the Della Martin, McDonnell-Pew and Sloan foundations, the National Eye Institute and the Office of Naval Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Snyder, L., Grieve, K., Brotchie, P. et al. Separate body- and world-referenced representations of visual space in parietal cortex. Nature 394, 887–891 (1998). https://doi.org/10.1038/29777
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/29777
This article is cited by
-
Integration of landmark and saccade target signals in macaque frontal cortex visual responses
Communications Biology (2023)
-
Dynamic spatial coding in parietal cortex mediates tactile-motor transformation
Nature Communications (2023)
-
The role of delta and theta oscillations during ego-motion in healthy adult volunteers
Experimental Brain Research (2021)
-
Neuronal vector coding in spatial cognition
Nature Reviews Neuroscience (2020)
-
Flexible coding of object motion in multiple reference frames by parietal cortex neurons
Nature Neuroscience (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.