Abstract
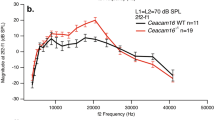
Mutations in genes encoding tectorial membrane (TM) proteins are a significant cause of human hereditary hearing loss (Hildebrand et al. 2011), and several mouse models have been developed to study the functional significance of this accessory structure in the mammalian cochlea. In this study, we use otoacoustic emissions (OAE), signals obtained from the ear canal that provide a measure of cochlear function, to characterize a mouse in which the TM is detached from the spiral limbus due to an absence of otoancorin (Otoa, Lukashkin et al. 2012). Our results demonstrate that spontaneous emissions (SOAE), sounds produced in the cochlea without stimulation, increase dramatically in mice with detached TMs even though their hearing sensitivity is reduced. This behavior is unusual because wild-type (WT) controls are rarely spontaneous emitters. SOAEs in mice lacking Otoa predominate around 7 kHz, which is much lower than in either WT animals when they generate SOAEs or in mutant mice in which the TM protein Ceacam16 is absent (Cheatham et al. 2014). Although both mutants lack Hensen’s stripe, loss of this TM feature is only observed in regions coding frequencies greater than ~15 kHz in WT mice so its loss cannot explain the low-frequency, de novo SOAEs observed in mice lacking Otoa. The fact that ~80 % of mice lacking Otoa produce SOAEs even when they generate smaller distortion product OAEs suggests that the active process is still functioning in these mutants but the system(s) involved have become less stable due to alterations in TM structure.




Similar content being viewed by others
References
Carvalho S, Mom T, Gilain L, Avan P (2004) Frequency specificity of distortion-product otoacoustic emissions produced by high-level tones despite inefficient cochlear electromechanical feedback. J Acous Soc Am 116:1639
Cheatham MA, Goodyear RJ, Homma K, Legan PK, Korchagina J, Naskar S, Siegel JH, Dallos P, Zheng J, Richardson GP (2014) Loss of the tectorial membrane protein CEACAM16 enhances spontaneous, stimulus-frequency, and transiently evoked otoacoustic emissions. J Neurosci 34:10325–10338
Crawford AC, Fettiplace R (1985) The mechanical properties of ciliary bundles of turtle cochlear hair cells. J Physiol 364:359–379
Dallos P, Billone MC, Durrant JD, Wang C, Raynor S (1972) Cochlear inner and outer hair cells: functional differences. Science 177:356–358
Dallos P, Wu X, Cheatham MA, Gao J, Zheng J, Anderson CT, Jia S, Wang X, Cheng WH, Sengupta S, He DZ, Zuo J (2008) Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron 58:1–7
Gold T (1948) Hearing II: the physical basis of the action of the cochlea. Proc R Soc B 135:492–498
Hildebrand MS et al (2011) DFNA8/12 caused by TECTA mutations is the most identified subtype of nonsyndromic autosomal dominant hearing loss. Hum Mutat 32:825–834
Hudspeth A (1997) Mechanical amplification of stimuli by hair cells. Curr Opin Neurobiol 7:480–486
Hudspeth AJ (2014) Integrating the active process of hair cells with cochlear function. Nat Rev Neurosci 15:600–614
Jaramillo F, Markin VS, Hudspeth AJ (1993) Auditory illusions and the single hair cell. Nature 364:527–529
Jones GP, Elliott SJ, Russell IJ, Lukashkin AN (2015) Modified protein expression in the tectorial membrane of the cochlea reveals roles for the striated sheet matrix. Biophys J 108:203–210
Kammerer R, Ruttiger L, Riesenberg R, Schauble C, Krupar R, Kamp A, Sunami K, Eisenried A, Hennenberg M, Grunert F, Bress A, Battaglia S, Schrewe H, Knipper M, Schneider MR, Zimmermann W (2012) Loss of mammal-specific tectorial membrane component carcinoembryonic antigen cell adhesion molecule 16 (CEACAM16) leads to hearing impairment at low and high frequencies. J Biol Chem 287:21584–21598
Kemp DT (1978) Stimulated acoustic emissions from within the human auditory system. J Acoust Soc Am 64:1386–1391
Legan PK, Rau A, Keen JN, Richardson GP (1997) The mouse tectorins. Modular matrix proteins of the inner ear homologous to components of the sperm-egg adhesion system. J Biol Chem 272:8791–8801
Legan PK, Lukashkina VA, Goodyear RJ, Kössl M, Russell IJ, Richardson GP (2000) A targeted deletion in alpha-tectorin reveals that the tectorial membrane is required for the gain and timing of cochlear feedback. Neuron 28:273–285
Legan PK, Goodyear RJ, Morin M, Mencia A, Pollard H, Olavarrieta L, Korchagina J, Modamio-Hoybjor S, Mayo F, Moreno F, Moreno-Pelayo MA, Richardson GP (2014) Three deaf mice: mouse models for TECTA-based human hereditary deafness reveal domain-specific structural phenotypes in the tectorial membrane. Hum Mol Genet 23:2551–2568
Liberman MC, Zuo J, Guinan JJ Jr (2004) Otoacoustic emissions without somatic motility: can stereocilia mechanics drive the mammalian cochlea? J Acoust Soc Am 116:1649–1655
Lim DJ (1971) Vestibular sensory organs. A scanning electron microscopic investigation. Arch Otolaryngol 94:69–76
Lukashkin AN, Richardson GP, Russell IJ (2010) Multiple roles for the tectorial membrane in the active cochlea. Hear Res 266:26–35
Lukashkin AN, Legan PK, Weddell TD, Lukashkina VA, Goodyear RJ, Welstead LJ, Petit C, Russell IJ, Richardson GP (2012) A mouse model for human deafness DFNB22 reveals that hearing impairment is due to a loss of inner hair cell stimulation. Proc Natl Acad Sci U S A 109:19351–19356
Maoileidigh DO, Hudspeth AJ (2013) Effects of cochlear loading on the motility of active outer hair cells. PNAS 110:5474–5479
Martin P, Bozovic D, Choe Y, Hudspeth AJ (2003) Spontaneous oscillation by hair bundles of the bullfrog’s sacculus. J Neurosci 23:4533–4548
Mom T, Bonfils P, Gilain L, Avan P (2001) Origin of cubic difference tones generated by high-intensity stimuli: effect of ischemia and auditory fatigue on the gerbil cochlea. J Acoust Soc Am 110:1477–1488
Moreno-Pelayo MA, Goodyear RJ, Mencia A, Modamio-Hoybjor S, Legan PK, Olavarrieta L, Moreno F, Richardson GP (2008) Characterization of a spontaneous, recessive, missense mutation arising in the Tecta gene. J Assoc Res Otolaryngol 9:202–214
Müller M, von Hunerbein K, Hoidis S, Smolders JW (2005) A physiological place-frequency map of the cochlea in the CBA/J mouse. Hear Res 202:63–73
Neely ST, Kim DO (1986) A model for active elements in cochlear biomechanics. J Acoust Soc Am 79:1472–1480
Neely S, Liu Z (1994) EMAV: otoacoustic emission averager. Technical Memo No 17, Boy’s Town National Research Hospital, Omaha NE.
Neely ST, Stevenson R (1992) SysRes. In: Tech Memo No. 1. Omaha NE: Boystown National Research Hospital.
Pearce M, Richter CP, Cheatham MA (2001) A reconsideration of sound calibration in the mouse. J Neurosci Methods 106:57–67
Probst R, Lonsbury-Martin BL, Martin GK (1991) A review of otoacoustic emissions. J Acoust Soc Am 89:2027–2067
Prodanovic S, Gracewski S, Nam JH (2015) Power dissipation in the subtectorial space of the mammalian cochlea is modulated by inner hair cell stereocilia. Biophys J 108:479–488
Richardson GP, Lukashkin AN, Russell IJ (2008) The tectorial membrane: one slice of a complex cochlear sandwich. Curr Opin Otolaryngol Head Neck Surg 16:458–464
Ruggero MA, Rich NC, Freyman R (1983) Spontaneous and impulsively evoked otoacoustic emissions: indicators of cochlear pathology? Hear Res 10:283–300
Salvi JD, O’Maoileidigh D, Fabella BA, Tobin M, Hudspeth AJ (2015) Control of a hair bundle’s mechanosensory function by its mechanical load. Proc Natl Acad Sci U S A 112:E1000–E1009
Strimbu CE, Kao A, Tokuda J, Ramunno-Johnson D, Bozovic D (2010) Dynamic state and evoked motility in coupled hair bundles of the bullfrog sacculus. Hear Res 265:38–45
Zheng J, Miller KK, Yang T, Hildebrand MS, Shearer AE, DeLuca AP, Scheetz TE, Drummond J, Scherer SE, Legan PK, Goodyear RJ, Richardson GP, Cheatham MA, Smith RJ, Dallos P (2011) Carcinoembryonic antigen-related cell adhesion molecule 16 interacts with alpha-tectorin and is mutated in autosomal dominant hearing loss (DFNA4). Proc Natl Acad Sci U S A 108:4218–4223
Acknowledgments
Work was supported by the National Institute on Deafness and Other Communicative Disorders, the National Institutes of Health (Grant DC000089 to M.A.C.), and the Wellcome Trust (Grant 087377 to G.P.R.). We thank Jonathan H. Siegel for wise counsel and technical expertise, as well as Elaine Cheshire Thompson and Trent Nicol for assistance with the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Cheatham, M.A., Ahmad, A., Zhou, Y. et al. Increased Spontaneous Otoacoustic Emissions in Mice with a Detached Tectorial Membrane. JARO 17, 81–88 (2016). https://doi.org/10.1007/s10162-015-0551-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10162-015-0551-7