Abstract
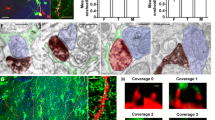
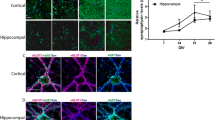
Hippocampal neurons in dissociated cell culture are one of the most extensively used model systems in the field of molecular and cellular neurobiology. Only limited data are however available on the normal time frame of synaptogenesis, synapse number and ultrastructure of excitatory synapses during early development in culture. Therefore, we analyzed the synaptic ultrastructure and morphology and the localization of presynaptic (Bassoon) and postsynaptic (ProSAP1/Shank2) marker proteins in cultures established from rat embryos at embryonic day 19, after 3, 7, 10, 14, and 21 days in culture. First excitatory synapses were identified at day 7 with a clearly defined postsynaptic density and presynaptically localized synaptic vesicles. Mature synapses on dendritic spines were seen from day 10 onward, and the number of synapses steeply increased in the third week. Fenestrated or multiple synapses were found after 14 or 21 days, respectively. So-called dense-core vesicles, responsible for the transport of proteins to the active zone of the presynaptic specialization, were seen on cultivation day 3 and 7 and could be detected in axons and especially in the presynaptic subcompartments. The expression and localization of the presynaptic protein Bassoon and of the postsynaptic molecule ProSAP1/Shank2 was found to correlate nicely with the ultrastructural results. This regular pattern of development and maturation of excitatory synapses in hippocampal culture starting from day 7 in culture should ease the comparison of synapse number and morphology of synaptic contacts in this widely used model system.






Similar content being viewed by others
Reference
Banker G, Cowan W (1977) Rat hippocampal neurons in dispersed cell culture. Brain Res 126:397–425
Banker G, Cowan W (1979) Further observations on hippocampal neurons in dispersed cell cultures. J Comp Neurol 187:469–494
Banker G, Waxman A (1988) Hippocampal neurons generate natural shapes in cell culture. In: Lasek R, Black MB (eds) Intrinsic determinants of neuronal form. Liss, New York
Bartlett WP, Banker G (1984) An electron microscopic study of the development of axons and dendrites by hippocampal neurons in culture. II Synaptic relationships. J Neurosci 4:1954–1965
Boeckers TM (2006) The postsynaptic density. Cell Tissue Res 326:409–422
Boeckers TM, Kreutz MR, Winter C, Zuschratter W, Smalla KH, Sanmarti-Vila L, Wex H, Langnaese K, Bockmann J, Garner CC, Gundelfinger ED (1999) Proline-rich synapse-associated protein-1/cortactin binding protein 1 (ProSAP1/CortBP1) is a PDZ-domain protein highly enriched in the postsynaptic density. J Neurosci 19:6506–6518
Boeckers TM, Bockmann J, Kreutz MR, Gundelfinger ED (2002) ProSAP/Shank proteins—a family of higher order organizing molecules of the postsynaptic density with an emerging role in human neurological disease. J Neurochem 81:903–910
Booher J, Sensenbrenner M (1972) Growth and cultivation of dissociated neurons and glial cells from embryonic chick, rain and human brain in flask cultures. Neurobiology 2:97–105
Bresler T, Shapira M, Boeckers T, Dresbach T, Futter M, Garner CC, Rosenblum K, Gundelfinger ED, Ziv NE (2004) Postsynaptic density assembly is fundamentally different from presynaptic active zone assembly. J Neurosci 24:1507–1520
Craig AM, Graf ER, Linhoff MW (2006) How to build a central synapse: clues from cell culture. Trends Neurosci 29:8–20
Crain B, Cotman C, Taylor D, Lynch G (1973) A quantitative electron microscopic study of synaptogenesis in the dentate gyrus of the rat. Brain Res 7:195–204
Deitch JS, Banker GA (1993) An electron microscopic analysis of hippocampal neurons developing in culture: early stages in the emergence of polarity. J Neurosci 13:4301–4315
Dieck S tom, Sanmarti-Vila L, Richter K, Kindler S, Soyke A, Wex H, Smalla KH, Kampf U, Franzer JT, Stumm M, Garner CC, Gundelfinger ED (1998) Bassoon, a novel zinc-finger CAG/glutamine-repeat protein selectively localized at the active zone of presynaptic nerve terminals. J Cell Biol 142:499–509
Dotti CG, Sullivan CA, Banker G (1988) The establishment of polarity by hippocampal neurons in culture. J Neurosci 8:1454–1468
Dresbach T, Torres V, Wittenmayer N, Altrock WD, Zamorano P, Zuschratter W, Nawrotzki R, Ziv NE, Garner CC, Gundelfinger ED (2006) Assembly of active zone precursor vesicles: obligatory trafficking of presynaptic cytomatrix proteins Bassoon and Piccolo via a trans-Golgi compartment. J Biol Chem 81:6038–6047
Fletcher TL, De Camilli P, Banker G (1994) Synaptogenesis in hippocampal cultures: evidence indicating that axons and dendrites become competent to form synapses at different stages of neuronal development. J Neurosci 14:6695–6706
Friedman HV, Bresler T, Garner CC, Ziv NE (2000) Assembly of new individual excitatory synapses: time course and temporal order of synaptic molecule recruitment. Neuron 27:57–69
Frotscher M, Zhao S, Graber W, Drakew A, Studer D (2007) New ways of looking at synapses. Histochem Cell Biol 128:91–96
Garner CC, Waites CL, Ziv NE (2006) Synapse development: still looking for the forest, still lost in the trees. Cell Tissue Res 326:249–262
Gerrow K, Romorini S, Nabi SM, Colicos MA, Sala C, El-Husseini A (2006) A preformed complex of postsynaptic proteins is involved in excitatory synapse development. Neuron 49:457–462
Goda Y, Davis GW (2003) Mechanisms of synapse assembly and disassembly. Neuron 40:243–264
Jin Y, Garner CC (2008) Molecular mechanisms of presynaptic differentiation. Annu Rev Cell Dev Biol 24:237–262
Kaech S, Banker G (2006) Culturing hippocampal neurons. Nat Protoc 1:2406–2415
Kreienkamp HJ (2008) Scaffolding proteins at the postsynaptic density: shank as the architectural framework. Handb Exp Pharmacol 186:365-380
Li Z, Sheng M (2003) Some assembly required: the development of neuronal synapses. Nat Rev Mol Cell Biol 4:833–841
Matteoli M, Coco S, Schenk U, Verderio C (2004) Vesicle turnover in developing neurons: how to build a presynaptic terminal. Trends Cell Biol 14:133–140
Naisbitt S, Kim E, Tu JC, Xiao B, Sala C, Valtschanoff J, Weinberg RJ, Worley PF, Sheng M (1999) Shank, a novel family of postsynaptic density proteins that binds to the NMDA receptor/PSD-95/GKAP complex and cortactin. Neuron 23:569–582
Papa M, Bundman MC, Greenberger V, Segal M (1995) Morphological analysis of dendritic spine development in primary cultures of hippocampal neurons. J Neurosci 15:1–11
Sensenbrenner M, Booher J, Mandel P (1972) Cultivation and growth of dissociated neurons from chick embryo cerebral cortex in the presence of different substrates. Z Zellforsch Mikrosk Anat 117:559–569
Shapira M, Zhai RG, Dresbach T, Bresler T, Torres VI, Gundelfinger ED, Ziv NE, Garner CC (2003) Unitary assembly of presynaptic active zones from piccolo-bassoon transport vesicles. Neuron 38:237–252
Siksou L, Triller A, Marty S (2009) An emerging view of presynaptic structure from electron microscopic studies. J Neurochem 108:1336–1342
Verderio C, Coco S, Pravettoni E, Bacci A, Matteoli M (1999) Synaptogenesis in hippocampal cultures. Cell Mol Life Sci 55:1448–1462
Zhai RG, Vardinon-Friedman H, Cases-Langhoff C, Becker B, Gundelfinger ED, Ziv NE, Garner CC (2001) Assembling the presynaptic active zone: a characterization of an active one precursor vesicle. Neuron 29:131–143
Ziv NE, Garner CC (2004) Cellular and molecular mechanisms of presynaptic assembly. Nat Rev Neurosci 5:385–399
Ziv NE, Smith SJ (1996) Evidence for a role of dendritic filopodia in synaptogenesis and spine formation. Neuron 17:91–102
Acknowledgements
We are grateful to Eberhard Schmidt from the Department of Electron Microscopy, University of Ulm for embedding and preparation of the sections for electron microscopy and to Renate Zienecker and Ursula Pika-Hartlaub for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the DFG (SFB 497/B8, Bo 1718/3-1, to T.M.B.) and a grant from the Graduate School for Molecular Medicine Ulm (to A.G.).
Rights and permissions
About this article
Cite this article
Grabrucker, A., Vaida, B., Bockmann, J. et al. Synaptogenesis of hippocampal neurons in primary cell culture. Cell Tissue Res 338, 333–341 (2009). https://doi.org/10.1007/s00441-009-0881-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-009-0881-z