Summary
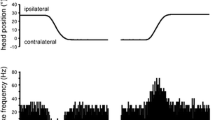
Neuronal activity in the flocculus of alert Rhesus monkeys was recorded during vestibular stimulation (rotation of the monkey about a vertical axis in complete darkness), optokinetic stimulation (rotation of the visual surround around the stationary monkey), combined visual-vestibular stimulation (rotation of the monkey inside the stationary surround in the light), and conflicting visual-vestibular stimulation (rotation of the monkey together with the visual surround in the same direction). The input to the flocculus was recorded as non-Purkinje cell (non-P-cell) activity. Ninety per cent of the non-P-cells which were modulated during our stimulation paradigms carry information similar to that in the neurons of vestibular nuclei. This suggests that the main mossy fiber input to the flocculus originates in the vestibular nuclei. A second input of unknown origin conveys visual information about retinal slip. Thus, part of the flocculus — as further discussed elsewhere (Waespe and Henn 1981) — may be specialized to subserve visual-vestibular interaction to improve the nystagmus response.
Similar content being viewed by others
References
Alley K (1977) Anatomical basis for interaction between cerebellar flocculus and brainstem. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons. Developments in neuroscience, vol 1. Elsevier, Amsterdam New York, pp 109–117
Baker J, Gibson A, Glickstein M, Stein J (1976) Visual cells in the pontine nuclei of the cat. J Physiol (Lond) 255: 415–433
Baker R, Berthoz A (1975) Is the prepositus hypoglossi. nucleus the source of another vestibulo-ocular pathway? Brain Res 86: 121–127
Baloh RW, Jenkins HA, Honrubia V, Yee RD, Leu CGY (1979) Visual-vestibular interaction and cerebellar atrophy. Neurology (Minneap) 29: 116–119
Bell CC, Grimm RJ (1969) Discharge properties of Purkinje cells recorded on single and double microelectrodes. J Neurophysiol 32: 1044–1055
Brodal A (1972) Vestibulocerebellar input in the cat: Anatomy. Basic aspects of central vestibular mechanisms. Prog Brain Res 37: 315–327
Brodal A, Høivik B (1964) Site and mode of termination of primary Vestibulocerebellar fibers in the cat. Arch Ital Biol 102: 1–21
Buettner UW, Büttner U (1979) Vestibular nuclei activity in the alert monkey during suppression of vestibular and optokinetic nystagmus. Exp Brain Res 37: 581–593
Büttner U, Waespe W (1981) Vestibular nerve activity in the alert monkey during vestibular and optokinetic nystagmus. Exp Brain Res 41: 310–315
Carpenter MC, Stein BM, Peter P (1972) Primary vestibulocerebellar fibers in the monkey. Distribution of fibers arising from distinctive cell groups in the vestibular ganglia. Am J Anat 135: 221–250
Cohen B, Matsuo V, Raphan Th (1977) Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic afternystagmus. J Physiol (Lond) 270: 321–344
Dichgans J, Jung R (1975) Oculomotor abnormalities due to cerebellar lesions. In: Lennerstrand G, Bach-y-Rita P (eds) Basic mechanisms of ocular motility and their clinical implications. Pergamon Press, Oxford New York, pp 281–298
Duensing F, Schaefer KP (1958) Die Aktivität einzelner Neurone im Bereiche der Vestibulariskerne bei Horizontalbeschleunigungen unter besonderer Berücksichtigung des vestibulären Nystagmus. Arch Psychiatr Nervenkr 198: 224–252
Eccles JC, Ito M, Szentágothai J (1967) The cerebellum as a neuronal machine. Springer, Berlin Heidelberg New York
Estanol B, Romero R, Corvera J (1979) Effects of cerebellectomy on eye movements in man. Arch Neurol 36: 281–284
Ghelarducci B, Ito M, Yagi N (1975) Impulse discharges from flocculus Purkinje cells of alert rabbits during visual stimulation combined with horizontal head rotation. Brain Res 87: 66–72
Gresty M, Baker R (1976) Neurons with visual receptive field, eye movement and neck displacement sensitivity within and around the nucleus prepositus in the alert cat. Exp Brain Res 24: 429–433
Hassul M, Daniels PD, Kimm J (1976) Effects of bilateral flocculectomy on the vestibulo-ocular reflex in the chinchilla. Brain Res 118: 339–343
Ito M (1972) Neural design of the cerebellar motor control system. Brain Res 40: 81–84
Ito M (1979) Adaptive modification of the vestibulo-ocular reflex in rabbits affected by visual input and its possible neuronal mechanisms. Reflex control of posture and movements. Prog Brain Res 50: 757–761
Ito M, Shiida T, Yagi N, Yamamoto M (1974) Visual influence on rabbit horizontal vestibulo-ocular reflex presumably effected via the cerebellar flocculus. Brain Res 65: 170–174
Kato I, Kawasaki T, Aoyagi M, Sato Y, Mizukoshi K (1979) Loss of visual suppression of caloric nystagmus in cats. Acta Otolaryngol (Stockh) 87: 489–505
Kawasaki T, Kato I, Aoyagi M, Sato Y, Koike Y, Ino H (1978) On the role of the brain stem in the visual suppression of caloric nystagmus. In: Hood JD (ed) Vestibular mechanisms in health and disease. Academic Press, London New York, pp 247–253
Keller EL (1976) Behavior of horizontal semicircular canal afferents in alert monkey during vestibular and optokinetic stimulation. Exp Brain Res 24: 459–471
Keller EL, Precht W (1979) Visual-vestibular responses in vestibular nuclear neurons in the intact and cerebellectomized alert cat. Neuroscience 4: 1599–1613
Kotchabhakdi N, Walberg F (1978) Cerebellar afferent projections from the vestibular nuclei in the cat. An experimental study with the method of retrograde axonal transport of horseradish peroxidase. Exp Brain Res 31: 591–604
Kotchabhakdi N, Hoddevik GH, Walberg F (1978) Cerebellar afferent projections from the perihypoglossal nuclei. An experimental study with the method of retrograde axonal transport of horseradish peroxidase. Exp Brain Res 31: 13–29
Kubo T, Matsunaga T, Hayashi Y (1978) Convergence of visual and vestibular inputs in pontine reticular formation of the rabbit. Brain Res 147: 177–182
Lisberger SG, Fuchs AF (1978a) Role of primate flocculus during rapid behavioral modification of vestibuloocular reflex. I. Purkinje cell activity during visually guided horizontal smooth-pursuit eye movements and passive head rotation. J Neurophysiol 41: 733–763
Lisberger SG, Fuchs AF (1978b) Role of primate flocculus during rapid behavioral modification of vestibuloocular reflex. II. Mossy fiber firing patterns during horizontal head rotation and eye movements. J Neurophysiol 41: 764–777
Louie AW, Kimm J (1976) The response of 8th nerve fibers to horizontal sinusoidal oscillation in the alert monkey. Exp Brain Res 24: 447–457
Maekawa K, Takeda T (1975) Mossy fiber responses evoked in the cerebellar flocculus of rabbits by stimulation of the optic pathway. Brain Res 98: 590–595
Miles FA, Fuller JH (1975) Visual tracking and the primate flocculus. Science 189: 1000–1002
Noda H, Suzuki DA (1979) Processing of the eye movement signals in the flocculus of the monkey. J Physiol (Lond) 294: 349–364
Noda H, Asoh R, Shibagaki M (1977) Floccular unit activity associated with eye movements and fixation. In: Baker R, Berthoz A (eds) Control of gaze by brain stem neurons, Developments in neuroscience, vol 1. Elsevier, Amsterdam New York, pp 371–380
Precht W (1978) Neuronal operations in the vestibular system. Studies of brain function, vol 2. Springer, Berlin Heidelberg New York
Precht W, Llinás R (1969) Functional organization of the vestibular afferents to the cerebellar cortex of the frog and cat. Exp Brain Res 9: 30–52
Reutern von GM, Dichgans J (1977) Augenbewegungsstörungen als cerebelläre Symptome bei Kleinhirnbrückenwinkeltumoren. Arch Psychiatr Nervenkr 223: 117–130
Shinoda Y, Yoshida K (1975) Neural pathways from the vestibular labyrinths to the flocculus in the cat. Exp Brain Res 22: 97–111
Simpson JI, Soodak RE, Hess R (1979) The accessory optic system and its relation to the vestibulocerebellum. Reflex control of posture and movement. Prog Brain Res 50: 715–724
Takemori S, Cohen B (1974) Loss of visual suppression of vestibular nystagmus after flocculus lesions. Brain Res 72: 213–224
Thach WT (1968) Discharge of Purkinje and cerebellar nuclear neurons during rapidly alternating arm movements in the monkey. J Neurophysiol 31: 785–797
Waespe W, Kenn V (1977a) Neuronal activity in the vestibular nuclei of the alert monkey during vestibular and optokinetic stimulation. Exp Brain Res 27: 523–538
Waespe W, Henn V (1977b) Vestibular nuclei activity during optokinetic after-nystagmus (OKAN) in the alert monkey. Exp Brain Res 30: 323–330
Waespe W, Henn V (1978) Conflicting visual-vestibular stimulation and vestibular nucleus activity in alert monkeys. Exp Brain Res 33: 203–211
Waespe W, Henn V (1979a) The velocity response of vestibular nucleus neurons during vestibular, visual and combined angular acceleration. Exp Brain Res 37: 337–347
Waespe W, Henn V (1979b) Motion information in the vestibular nucleus of alert monkeys. Visual and vestibular input vs. optomotor output. Reflex control of posture and movement. Prog Brain Res 50: 683–693
Waespe W, Henn V (1981) Visual-vestibular interaction in the flocculus of the alert monkey. II. Purkinje cell activity. Exp Brain Res 43: 349–360
Waespe W, Büttner U, Henn V (1981) Input-output activity of the primate flocculus during visual-vestibular interaction. Annals New York Academy of Sciences (in press)
Westheimer G, Blair SM (1974) Unit activity in accessory optic system in alert monkeys. Invest Ophthalmol Vis Sci 13: 533–534
Winfield JA, Hendrickson A, Kimm J (1978) Anatomical evidence that the medial terminal nucleus of the accessory optic tract in mammals provides a visual mossy fiber input to the flocculus. Brain Res 151: 175–182
Wurtz RH (1969) Visual receptive fields of striate cortex neurons in awake monkeys. J Neurophysiol 32: 27–742
Zee DS, Yee RD, Cogan DG, Robinson DA (1976) Ocular motor abnormalities in hereditary cerebellar ataxia. Brain 99: 207–234
Author information
Authors and Affiliations
Additional information
Supported by a grant from the Swiss National Foundation for Scientific Research 3.343-2.78
Rights and permissions
About this article
Cite this article
Waespe, W., Büttner, U. & Henn, V. Visual-vestibular interaction in the flocculus of the alert monkey. Exp Brain Res 43, 337–348 (1981). https://doi.org/10.1007/BF00238376
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00238376